
Neurotrophic keratopathy is a common problem that can significantly affect quality of life and lead to permanent loss of vision. Poor corneal sensation can decrease the blink reflex and limbal stem cell function and can impair corneal epithelial integrity.1,2 Corneal scarring and ulceration ensue, leading to vision loss and even perforation and loss of the eye.
Standard treatment consists of aggressive lubrication, application of bandage contact lenses, and eyelid tarsorrhaphy to help maintain wetting of the ocular surface. Amniotic membrane grafts, cyanoacrylate glue, and lamellar corneal transplantation are used in cases of corneal thinning or perforation.3,4 A neurotrophic cornea and accompanying persistent epithelial defects remain difficult to treat. Medical therapy is one area of exploration (see New Medical Therapies). Others include surgical techniques that directly address the underlying problem of the denervated cornea.
NEW MEDICAL THERAPIES
Cenegermin (Oxervate, Dompe Farmaceutici) is specifically indicated for the treatment of neurotrophic keratitis. This recombinant replacement of human nerve growth factor can activate and stimulate dormant or immature nerves to grow new axons and make appropriate connections. In two multicenter randomized controlled trials, 70% of corneas treated with cenegermin showed complete corneal healing after 8 weeks compared to 28% of corneas treated with placebo.1,2
Other topical biologics are currently under investigation for the treatment of dry eye disease and neurotrophic cornea:
• Blood-derived autologous serum eye drops provide growth factors that can aid epithelial healing, but cost and difficulties in preparation may be prohibitive.3
• Caciol20 (RGTA) is a new matrix therapy agent that provides binding sites for growth factors to promote regeneration of the epithelium.1,4
1. Dua HS, Said DG, Messmer EM, et al. Neurotrophic keratopathy. Prog Retin Eye Res. 2018;66:107-131.
2. McDonald MB. Topical recombinant human nerve growth factor an innovation in ocular surface disease treatment. Ocular Surgery News. Published October 10, 2018. https://bit.ly/2UHYUug. Accessed December 13, 2018.
3. Drew VJ, Tseng C, Seghatchian J, Burnouf T. Reflections on dry eye syndrome treatment: therapeutic role of blood products. Front Med (Lausanne). 2018;5:33.
4. Aifa A. Gueudry J. Portmann A, et al. Topical treatment with a new matrix therapy agent (RGTA) for the treatment of corneal neurotrophic ulcers. Invest Ophthalmol Vis Sci. 2012;53(13);8181-8185.
SURGICAL REINNERVATION
Corneal neurotization restores sensory function by grafting a healthy donor nerve segment to the denervated cornea. In several independent series, patients who have undergone the procedure have recovered corneal sensation, as determined by Cochet-Bonnet esthesiometry, within 6 months after the procedure.3-7 Some patients with nonhealing corneal epithelial defects were able to achieve full epithelialization after corneal neurotization (Figure 1), and many of them reported improved quality of life and ocular comfort. More important, treatment of the neurotrophic cornea subsequently allowed a number of patients to undergo vision-restoring keratoplasty.
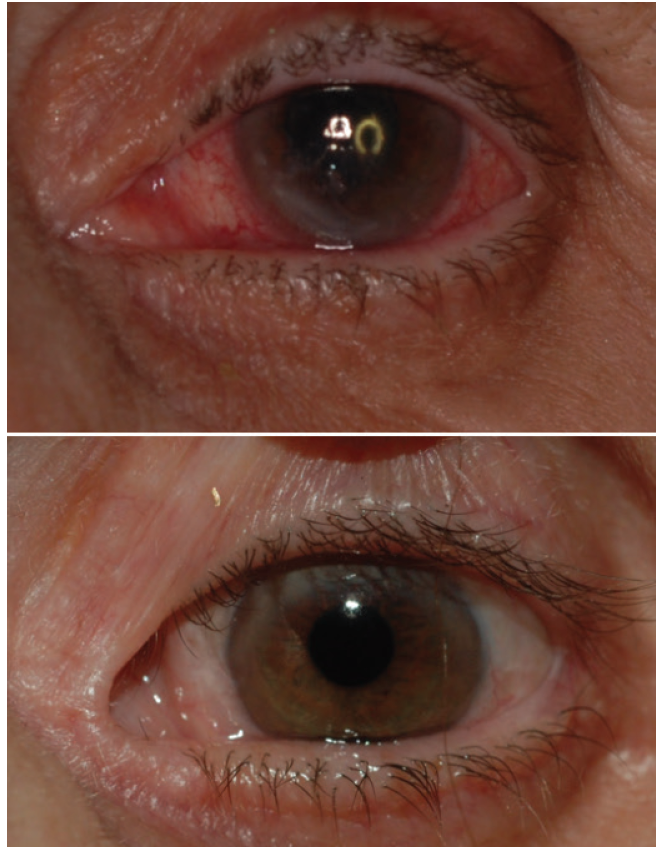
Figure 1. Before (top) and 2 years after (bottom) a corneal neurotization procedure. Note the relief of conjunctival injection, stromal haze, and corneal epithelial irregularity.
Courtesy of Ilya Leyngold, MD
Corneal sensation is supplied by the terminal endings of the frontal nerve, the largest branch of the ophthalmic division of the trigeminal nerve. Acquired corneal anesthesia is most commonly caused by postherpetic neuropathy, but it can also result from surgery for posterior fossa tumor. Congenital causes can be isolated or related to other syndromes.3,4
Terzis and colleagues first described harvesting the intact contralateral supraorbital and supratrochlear nerves to directly provide sensory innervation to the neurotrophic cornea in 2008.5 Although successful in improving corneal sensibility, visual acuity, and overall corneal health, the procedure required a large bicoronal incision, resulting in a large scar and a risk of alopecia, facial nerve injury, and subgaleal hematoma.3,5
More recently, less invasive techniques have been developed that achieve similar improvement in corneal function. Ilya Leyngold, MD, an oculoplastic surgeon and assistant professor of ophthalmology at Duke University School of Medicine in Durham, North Carolina, performs corneal neurotization with an endoscope and accesses either the ipsilateral or contralateral supraorbital and supratrochlear nerves. This minimally invasive method requires the creation of two small incisions behind the hairline and one small eyelid incision versus the large bicoronal scalp incision originally described.
“I came across the idea of using this less invasive approach while performing endoscopic brow lifts, where I encounter the supraorbital and supratrochlear nerves all the time, and decided to do some cadaver work and found it very feasible,” Dr. Leyngold told me.
In cases in which there is bilateral corneal anesthesia and ophthalmic nerve damage, using a donor nerve graft can allow coaptation to other nerves, such as the maxillary or even the mandibular division of the trigeminal nerve. Asim Ali, MD, a pediatric cornea specialist at the Hospital for Sick Children in Toronto, has described using the sural nerve, which normally supplies sensation to the lateral aspect of the foot and ankle, to act as a graft for neurotization.3,4
“This limits scarring and morbidity compared to the original bicoronal approach, allows more rapid return of sensation, and can address both unilateral and bilateral disease as well as congenital and acquired causes of corneal anesthesia,” Dr. Ali explained to me. “We conduct sensory input from any functioning regional sensory nerve and direct that sensation into the affected cornea via the nerve graft.”
In addition to restoring corneal sensation, Dr. Ali and colleagues have used sural nerve graft segments to reinnervate the cheek and lips in a 16-year-old girl and to treat facial paralysis in a 9-year-old boy.3
Acellular nerve allograft (Avance, AxoGen) is another option that Dr. Leyngold and associates have used in order to avoid donor site morbidity altogether.7 The allograft “acts as a scaffold” and “provides a conduit for the nerve to regenerate, bridging the distal nerve discontinuity” with target tissue, he said. Dr. Ali cautioned, however, that, although nerve allografts “reduce the complexity of the operation, [they] may limit the overall axonal growth, as observed in some of our basic science studies. Further, the grafts that are currently available are not quite long enough to cover the distance from the donor nerve to the corneal stroma in some cases.”
In all these surgical methods, the nerve fascicles are tunneled toward the diseased cornea, and the endings are tacked around the limbus (Figure 2), resulting in nerve regeneration.8,9 That said, there is more involved than just physically extending an intact nerve or graft. Chemical neuromediators play a role.
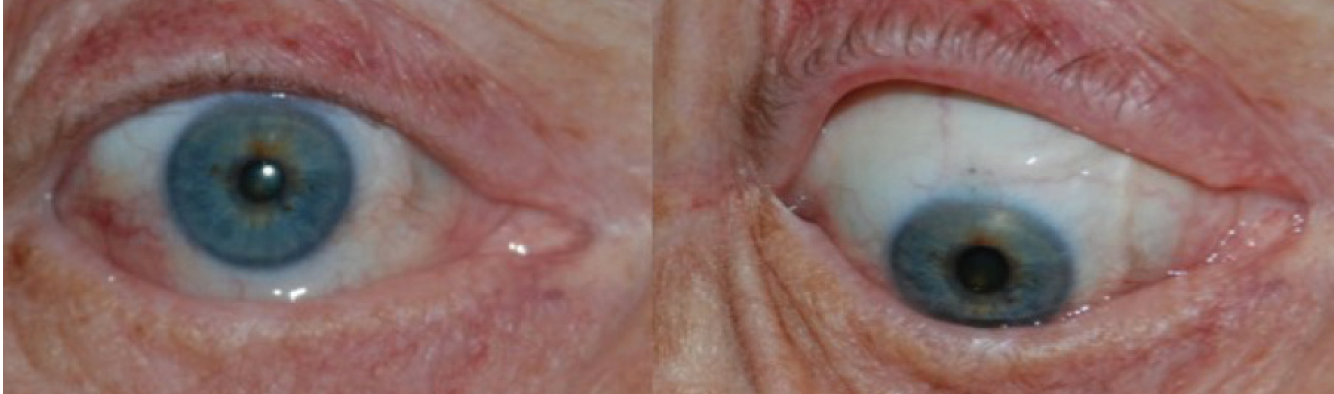
Figure 2. Appearance of cornea and donor nerve fascicles 3 months after corneal neurotization procedure.
Courtesy of Ilya Leyngold, MD
“Neuropeptides, in addition to the direct nerve transfer, play a great role in resensitizing the cornea, inducing new axonal sprouting,” Dr. Leyngold said.
LIMITATIONS AND FUTURE CHALLENGES
As with any procedure using a patient’s own donor nerves, hypoesthesia at the donor site is a side effect. Patients have also reported that mechanical stimulation of their cornea after surgery can feel as if they are being touched at the respective cutaneous territory of the donor nerve.3,4,7 Fortunately, neural adaptation takes place in most cases, restoring sensation at the skin territory of the donor site and appropriate perception of stimulation at the cornea within a number of months postoperatively.
“We discuss with patients the possibility of dysesthesia, although it hasn’t been a significant problem in our experience,” Dr. Ali said.
Both he and Dr. Leyngold acknowledged that the learning curve for the surgical procedure is steep, requiring collaboration between oculoplastic surgeons and corneal specialists. For instance, Dr. Ali works with his plastic and reconstructive surgery colleagues in a team approach to corneal neurotization. His colleagues harvest the sural nerve grafts and coapt them to the donor nerves, while Dr. Ali secures the nerve fascicles around the eye into corneoscleral tunnels. Dr. Leyngold said he hopes that, as these partnerships develop and other centers adopt the procedure, it will allow neurotrophic patients to be treated at an earlier stage in their disease and thus preserve vision or allow restorative procedures to be performed more expeditiously.
1. Garcia-Hirschfeld J, Lopez-Briones LG, Belmonte C. Neurotrophic influences on corneal epithelial cells. Exp Eye Res. 1994;59(5):597-605.
2. Ueno H, Ferrari G, Hattori T, et al. Dependence of corneal stem/progenitor cells on ocular surface innervation. Invest Ophthalmol Vis Sci. 2012;53(2):867-872.
3. Elbaz U, Bains R, Zuker RM, et al. Restoration of corneal sensation with regional nerve transfers and nerve grafts: a new approach to a difficult problem. JAMA Ophthalmol. 2014;132(1):1289-1295.
4. Bains RD, Elbaz U, Zuker RM, et al. Corneal neurotization from the supratrochlear nerve with sural nerve grafts: a minimally invasive approach. Plast Resconstr Surg. 2015;135(2):397e-400e.
5. Terzis JK, Dryer MM, Bodner BI. Corneal neurotization: a novel technique for the anesthetic cornea. Cornea. 2010;29(7):812-819.
6. Leyngold I, Weller C, Leyngold M, Tabor M. Endoscopic corneal neurotization: technique and initial experience. Ophthalmic Plast Reconstr Surg. 2018;34(1):82-85.
7. Leyngold IM, Yen MT, Tian J, et al. Minimally invasive corneal neurotization with acellular nerve allograft: surgical technique and clinical outcomes [published online ahead of print July 24, 2018]. Ophthalmic Plast Reconstr Surg.
8. Fung S, Catapano J, Elbaz U, et al. In vivo confocal microscopy reveals corneal reinnervation after treatment of neurotrophic keratopathy with corneal neurotization. Cornea. 2018;37(1):109-112.
9. Ting DS, Figueiredo GS, Henein C, et al. Corneal neurotization for neurotrophic keratopathy: clinical outcomes and in vivo confocal microscopic and histopathological findings. Cornea. 2018;37(5):641-646.



