CASE PRESENTATION
A 27-year-old white woman presented for a refractive surgery consultation. The patient had a history of high hyperopia and high regular astigmatism in both eyes as well as mild amblyopia in the right eye, for which she received treatment as a child in the form of patch occlusion. She complained of poor vision at distance and near that was getting progressively worse despite her use of spectacles and contact lenses. Additionally, she stated that she had become contact lens intolerant and was no longer comfortable driving because of the visual distortion caused by her glasses. The patient said that poor vision was affecting her quality of life and was a major source of stress for her.
An ocular examination was unremarkable other than some conjunctival hyperemia and mild corneal pannus in each eye. BCVA was +10.25 -6.25 x 174º = 20/30+2 OD and +7.25 -5.00 x 005º = 20/20- OS. Keratometry readings obtained with the OPD-Scan III (Nidek) were 38.35/44.29 D @ 86º (5.94 D) OD and 38.93/43.89 D @ 100º (4.96 D) OS (Figure 1). Keratometry readings obtained with the Lenstar (Haag-Streit) were 38.24/44.45 @ 85º (6.32 D) OD and 39.04/44.63 @ 99º (5.54 D) OS.
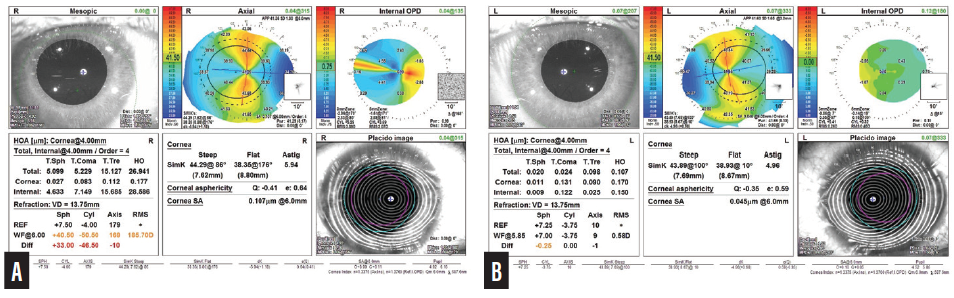
Figure 1. Preoperative measurements with the OPD-Scan III for the right (A) and left (B) eyes.
Axial length measurements obtained with the Lenstar were 21.07 mm OD and 21.55 mm OS (Figure 2). Central corneal thickness was 493 µm OD and 501 µm OS. Anterior chamber depth was 3.13 mm OD and 3.10 mm OS.
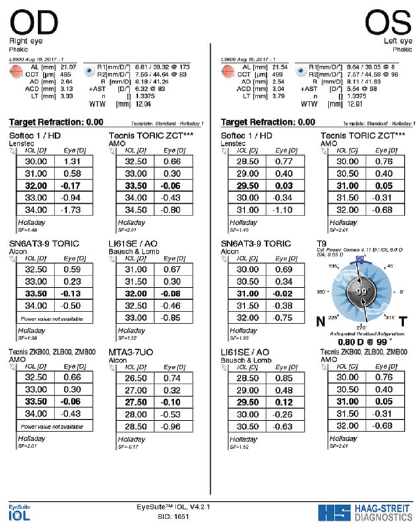
Figure 2. Preoperative measurements with the Lenstar.
IOL power calculations using the Holladay I formula recommended a Tecnis Toric 1-Piece IOL (model ZCT600, Johnson & Johnson Vision) with a power of 33.50 D for the right eye, leaving a predicted spherical equivalent of -0.06 D and 2.20 D of residual astigmatism. IOL power calculations using the same formula recommended a Tecnis Toric 1-Piece IOL with a power of 31.50 D for the left eye, leaving a predicted spherical equivalent of -0.31 D and 1.60 D of residual astigmatism (Figure 3). As a reference for understanding, calculations using the Donnenfeld nomogram called for two 75º corneal arcuate incisions in the right eye and two 55º arcuate incisions in the left eye to correct the residual astigmatism.
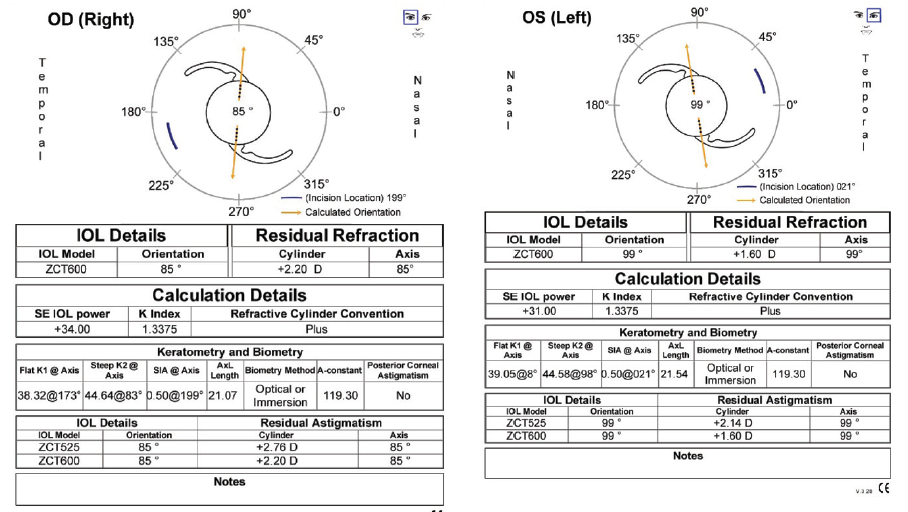
Figure 3. Calculations for the Tecnis Toric 1-Piece IOL.
How would you proceed?
—Case prepared by Gary Wörtz, MD

ARTHUR B. CUMMINGS, MB CHB, FCS(SA), MMED(OPHTH), FRCS(EDIN)
Young patients with high hyperopia present the biggest challenge in my practice for three reasons:
No. 1: They really need treatment because they cannot see well at any distance.
No. 2: The solution to their problem often is not obvious.
No. 3: These patients frequently also have amblyopia, making the risk-benefit ratio a little less attractive than in typical cases.
I will address each of the four tools at my disposal in turn, starting with LASIK. The cornea of the left eye is flat enough to allow the steepening required, but treatment of the right eye remains outside the scope of LASIK in my experience. I also do not have access to the cycloplegic values.
The aqueous depth is insufficient in each eye to allow placement of a Visian ICL (STAAR Surgical). Otherwise, implantation of this phakic IOL would be my unequivocal first choice.
Refractive lens exchange (RLE) would be an excellent strategy if the patient were presbyopic, but she is not. I have performed RLE even for patients with high hyperopia who are in their 20s like this patient, but typically only when the implications are even more significant (eg, a job requirement for joining the police force).
The best current option may be a new procedure such as lenticular implantation keratoplasty or placement of an allogenic lenticule (TransForm Corneal Allograft, Allotex). The general idea is to create a LASIK flap and add corneal tissue to achieve the correct refraction. If the result is unsatisfactory, the lenticule can be removed. These procedures, however, are neither mainstream globally nor available in the United States.
My advice is therefore to have the patient continue wearing spectacles and possibly try a different brand of contact lenses such as a rigid gas permeable, hybrid, or mini-scleral model and to review her situation in 1 to 2 years. The rate of change in ophthalmology is so fast that a suitable bridging solution may become available that can help her until RLE is a suitable option. If she is unsuccessful with contact lenses and is adamant about wanting freedom from spectacles, the only viable option for her is RLE with a presbyopia-correcting IOL.

PREEYA K. GUPTA, MD
Absolute presbyopia can be difficult for young patients to tolerate, but this patient is already having difficulty seeing at near and far because of accommodative stress and blur. She should be counseled about the changes she will experience at near after IOL surgery.
Preoperatively, it is vital that the patient understand that she will likely have a residual refractive error requiring a PRK enhancement in each eye. I would attempt to correct as much hyperopia and astigmatism as possible with a toric IOL and laser arcuate incisions. (I typically do not make incisions greater than 45º because they may behave less predictably.) She likely retains some level of refractive amblyopia and will probably be able to tolerate the residual cylinder.
The keys to success here are the informed consent and setting realistic expectations. RLE can be a life-changing procedure for patients who have an extreme refractive error if they have appropriate expectations.


RAPHAEL PENATTI, MD; AND GEORGE O. WARING IV, MD, FACS
In the United States, the most suitable option for this patient is RLE. We would make her fully aware of the procedure’s risks, benefits, and limitations and take all normal precautions for lens surgery on a short eye, potential inability to achieve 20/20 visual acuity with amblyopia, and possible need for spectacles for some activities after surgery.
Because of the short axial length, we would use the Barrett universal formula and Hill-RBF calculator for IOL selection. To attempt to preserve functional reading vision, our preference would be a toric presbyopia-correcting IOL. In the United States, both a toric extended depth of focus IOL and a toric trifocal IOL are available in the required spherical power with up to approximately 2.50 D of astigmatic correction at the corneal plane, which would leave approximately 1.00 D of residual corneal astigmatism in each eye after we account for posterior corneal astigmatism and the effect of drift over time in a young patient, so we would aim to leave some residual with-the-rule astigmatism. Our recommendation would be RLE assisted by a femtosecond laser and combined with laser arcuate incisions at the 9-mm optical zone to address residual astigmatism. Preoperatively, we would counsel the patient regarding the likelihood that she will need an enhancement once she achieves refractive stability.

WHAT I DID: GARY WÖRTZ, MD
Given the high hyperopia and high regular astigmatism, I felt that the only reasonable option for this patient was RLE with a toric IOL. The highest-powered toric IOL available in the United States, however, corrects only about 4.00 D of corneal astigmatism, so I knew that this strategy would leave residual astigmatism. Moreover, because the preoperative measurements were unusual, I knew that the IOL power calculations were going to be difficult. Another consideration was the loss of accommodation after surgery. Given the patient’s inability to function in her environment with glasses and her contact lens intolerance, I felt that the pros of surgery outweighed the cons.
The patient and I had a long discussion about her probable need for additional corrective surgery such as PRK to fine-tune her result. We also discussed her amblyopia and that a 20/20 outcome was likely unattainable in either eye. Additionally, I explained that she would need reading glasses postoperatively. This patient was desperate for help, and her expectations were very reasonable, which made me feel comfortable about offering treatment. We discussed monovision, but after considering her history of amblyopia and lack of prior experience with monovision, we elected to proceed with distance correction only.
I used the Catalys Precision Laser System (Johnson & Johnson Vision) to create a 4.8-mm capsulotomy centered via the scanned capsule method and two 60º corneal arcuate incisions at 265º and 85º (9 mm in length and 80% depth) in the right eye. Although the Donnenfeld nomogram called for two 75º arcs, I am not comfortable making corneal arcuate incisions beyond 60º after experiencing some poor results with larger arcs. I implanted a 34.00 D Tecnis Toric IOL at the 85º axis (predicted postoperative refraction of -0.43 D). Because I knew the result would likely require refinement with PRK, I targeted a refractive result on the minus side instead of plano.
One week later, I operated on the left eye. I used the Catalys Precision Laser System to create a 4.8-mm capsulotomy and two 50º corneal arcuate incisions at 99º and 279º, again by the scanned capsule method. Based on the difference in preoperative keratometry readings obtained with two devices, I reduced the arcs from the 55º recommended by the Donnenfeld nomogram to 50º. I implanted a 30.50 D Tecnis Toric IOL at the 99º axis (predicted postoperative refraction of +0.40 D). Because the postoperative result in the patient’s first eye was slightly more myopic than expected, I felt comfortable adjusting the target closer to plano.
The postoperative results were better than expected. UCVA was 20/30 OD and 20/20 OS. BCVA was -0.50 -0.50 x 045º = 20/20-3 OD and +0.25 -0.50 x 130º = 20/20- OS. Having been turned away by many other ophthalmologists, the patient was overjoyed with her outcome. She is able to drive and work, and she wears reading glasses for near tasks.
Based on my success to date with this approach, I was not too concerned about the possibility that imprecision in the arcuate incisions would change the axis of astigmatism and thus render the toric IOL ineffective. In eyes with this degree of astigmatism, the key is to debulk the astigmatism to the best of our ability. I should note that this case predated US availability of an extended depth of focus or toric multifocal IOL.



