Patients With Corneal Ectasia
Neel S. Vaidya, MD, MPH, and Parag A. Majmudar, MD


Several important factors must be considered during the cataract surgery evaluation for patients with corneal ectasia. The patient’s complaints and symptoms must be evaluated carefully to ensure that the cataract is significant and the symptoms are not related to another pathology. It can be tricky, however, to discern the etiology of symptoms because the symptoms commonly attributed to cataracts—glare, halos, and difficulty with night driving—can also be attributed to the higher-order aberrations caused by corneal ectasia.
DETERMINE WHEN THE SYMPTOMS DEVELOPED
Understanding the timeline for the development of symptoms is key. Did the patient begin experiencing symptoms a few months ago, or was it years ago? A rigid gas permeable (RGP) contact lens overrefraction can help differentiate the visual impact of the ectasia from that of a developing cataract. If the density of the cataract is moderate to severe, however, a contact lens overrefraction may not improve the visual acuity. Additionally, if the ectasia is severe and the patient’s visual complaints preceded cataract formation, it may be necessary to consider corneal transplantation (penetrating keratoplasty or deep anterior lamellar keratoplasty) in addition to cataract surgery.
Ensuring the cornea is stable with sequential corneal imaging studies, such as topography or Pentacam (Oculus Optikgeräte), over time is crucial. In general, keratoconus is unlikely to progress in the age group of patients who present for a cataract surgery evaluation. With that said, ectasia after refractive surgery and pellucid marginal degeneration tend to be less predictable. Confirming corneal stability is therefore crucial. The ectasia should be addressed before cataract surgery if the cornea is found to be unstable.
CONTACT LENS HISTORY AND USE
Another key component of the preoperative assessment is contact lens history. The irregular astigmatism present in eyes with corneal ectasia is difficult to correct surgically (ie, with a toric IOL). Patients are likely to continue to require contact lenses after cataract surgery to achieve optimal vision. Contact lens wear must be discontinued for days to weeks before the evaluation to achieve reliable biometry measurements. The duration of discontinuation depends on the type of contact lens (ie, soft vs RGP).
If a patient cannot function without contact lenses, surgery can be staged in such a way that allows them to continue to wear a contact lens on one eye while the other eye is being evaluated. If the patient has never worn contact lenses, performing a contact lens trial before surgery can help determine if they will be able to wear a contact lens comfortably after surgery. If a patient routinely wears RGP or scleral contact lenses and is a candidate for a toric IOL, it is important to explain that they will not be able to wear RGP and scleral contact lenses postoperatively.
IOL SELECTION
IOL selection can be the most challenging aspect of cataract surgery on patients with corneal ectasia. Our preference is a monofocal nontoric IOL with a target of approximately 3.00 D of myopia for a calculated refractive outcome. The main reason is that most patients with ectasia are myopic before they develop a cataract and rely on some degree of uncorrected near vision. If an emmetropic endpoint is chosen, then, given the often substantial magnitude of astigmatism present in these eyes, neither uncorrected distance nor near visual acuity will be useful for the patient.
Toric IOLs should be implanted cautiously in this population. Given the irregular nature of the astigmatism, outcomes tend to be less predictable. Intraoperative aberrometry such as Optiwave Refractive Analysis (Alcon) may help to orient the toric IOL to the correct axis. It is harder, however, for the device to capture accurate biometric data in these cases, so aberrometry readings may be unreliable.
Multifocal and extended depth of focus IOLs should be avoided in these patients.
SURGICAL TECHNIQUE
Surgery may be performed in the manner that is most comfortable to the ophthalmologist. No significant alterations in standard technique are required. Severe ectasia and corneal scarring may affect visualization during the typical maneuvers of cataract surgery.
Regardless of IOL selection, it is important to set realistic expectations for patients with corneal ectasia. We explain that they will still require correction with glasses or contact lenses postoperatively to achieve optimal visual acuity and that their BCVA may not be 20/20 after surgery owing to the presence of ectasia.
CONCLUSION
With an appropriate preoperative evaluation and a thorough discussion with the patient to set realistic goals and expectations, cataract surgery can significantly improve the vision and quality of life of patients with corneal ectasia.
Cataract Surgery in the Presence of Fuchs Dystrophy
Kenneth A. Beckman, MD, FACS

Fuchs dystrophy is a common condition that necessitates complex planning for cataract surgery. As with many other coexisting corneal conditions, there are three questions that I ask myself when approaching surgery:
No. 1: Is the visibility through the cornea adequate for me to perform cataract surgery?
No. 2: Can the cornea withstand cataract surgery? If so, what precautions should be taken to protect the cornea?
No. 3: Should cataract surgery be combined with endothelial keratoplasty (EK), or should the procedures be staged?
COMBINED OR STAGED PROCEDURES
Sequence. If corneal clarity permits cataract surgery, the surgeon has the option of combining it with EK or staging the procedures. In general, when EK is performed in combination with cataract surgery, the cataract is removed first.
I prefer to stage cataract surgery and EK whenever possible. I perform the cataract procedure first to give the patient an opportunity to avoid EK if the cornea survives. The recovery period after EK is much longer than after cataract surgery, and the former may require a lifetime of maintenance. Because the decision to proceed with subsequent EK can be made within weeks of cataract surgery, this staging sequence does not cause the patient a significant delay.
Proceeding with EK at the time of cataract surgery guarantees the patient an extended recovery time and a lifetime of maintenance. If it is inevitable that the cornea will decompensate because of cataract surgery or if the patient’s other eye experienced decompensation after cataract surgery, then I typically proceed with a combined procedure.
Risk assessment. Pachymetry may be an indicator that the cornea is at risk of decompensation after cataract surgery. It is not always reliable, however, because some corneas may be thick without having significant edema. Some may also have visually disabling dense guttata without edema. If the guttata are dense enough even without edema, the patient may still benefit from EK. Also, if there is significant asymmetry in corneal thickness between the two eyes, then I may opt for combined surgery.
It may be useful to assess the endothelial cells with specular microscopy. I, however, do not rely on this because there is no clear cutoff as to when EK is necessary based on endothelial cell counts alone.
STANDALONE SURGERY
EK alone. If the cornea is so cloudy that there is not a clear view to remove the cataract first or if there is no cataract at all, then EK may be performed alone. In my experience, the patient may rapidly develop cataracts after EK. I prefer to wait about 6 months after EK before proceeding with cataract surgery; this can be frustrating to the patient. This type of timing, however, often is not possible. Performing cataract surgery after EK carries the risk of damaging the graft and leading to graft failure, regardless of when the cataract is done.
Cataract surgery alone. I take several precautions if cataract surgery is to be performed alone. The use of topical hypertonics pre- and postoperatively may help decrease corneal edema. One week before surgery, patients typically begin administering topical steroids four times daily.
During surgery, a dispersive OVD is instilled to help protect the endothelium. Attempts to minimize phaco energy are also made. The use of trypan blue dye may improve visualization through the guttata for the capsulorhexis, although the cataract may not be particularly dense.
After surgery, steroids may be needed for an extended period. The dose may be higher than with standard surgery. Usually, the patient’s need for EK can be determined within a few weeks.
IOL CONSIDERATIONS
If combined surgery or EK after cataract surgery is planned, I account for the hyperopic shift that may occur after DSEK. Some surgeons have reported a significant shift, but I have found that targeting a postoperative refractive error of approximately -0.75 D is usually sufficient. If I do not expect the patient to need EK, then I select a lens power in my usual fashion. For DMEK, the hyperopic shift is not a concern.
Presbyopia-correcting IOLs should be used with caution. Dense guttata may cause significant postoperative aberrations and intolerance of the IOL. If combined surgery is planned, I hesitate to use presbyopia-correcting IOLs because of these potential aberrations. Additionally, the final refractive error is less predictable. I prefer aspheric monofocal IOLs for these patients. I also avoid hydrophilic acrylic lenses in these patients as they may opacify after EK due to the air bubble. I have seen several patients develop opacification after air bubble placement and require an IOL exchange. If EK is performed before cataract surgery and the cornea is stable and clear, some surgeons may elect to implant an extended depth of focus IOL.
CONCLUSION
Cataract surgery on a patient with Fuchs dystrophy requires significant preoperative planning and intraoperative precautions, and the postoperative care required may be more extensive than after standard cataract surgery. Preoperative counseling and careful decision-making are crucial to attaining the best outcomes.
Patients With a History of Radial Keratotomy
Joseph G. Christenbury, MD

Patients with a history of radial keratotomy (RK) often experience diurnal fluctuations in vision as the cornea steepens and the refraction becomes myopic.1 A link between myopia and an earlier onset of cataracts is well established,2 so it is not surprising that patients who underwent RK for the treatment of high myopia tend to develop nuclear sclerotic cataracts early in life. Before proceeding to cataract surgery, it is important for ophthalmologists to describe the risks and benefits of the procedure—including unique considerations in this population such as less reliable IOL power calculations (for more on this topic, see “IOL Calculations in Patients With Keratoconus”). Reasonable patient expectations are essential for postoperative satisfaction.
PREOPERATIVE COUNSELING
Some patients who have undergone RK are not candidates for a presbyopia-correcting IOL. The RK cuts in the cornea may cause irregular astigmatism or coma, which can negatively affect the optics of a multifocal or extended depth of focus IOL. Obtaining an accurate measurement of preoperative corneal power and predicting effective lens position can be challenging, and errors can induce an undesirable postoperative refractive surprise.3 Postoperatively, dry eye can cause vision to fluctuate and exacerbate the natural fluctuations that RK patients experience owing to their corneal irregularity. I inform them preoperatively that they may need to wear glasses when reading or to see at distance, and I caution them that a scleral or RGP contact lens may be necessary after surgery.
CORNEAL CONSIDERATIONS
If the eye has eight or fewer RK incisions, it may be possible to create a clear corneal incision that does not intersect with an RK incision. If the eye has more than eight RK cuts, however, it is generally safer to construct a scleral tunnel wound. Ophthalmologists should be prepared for a longer surgical duration in the latter scenario.
Corneal pathology such as epithelial basement membrane dystrophy and Salzmann nodular degeneration should be addressed surgically before the cataract procedure. These corneal pathologies often affect the central cornea, causing irregular astigmatism (Figure 1).
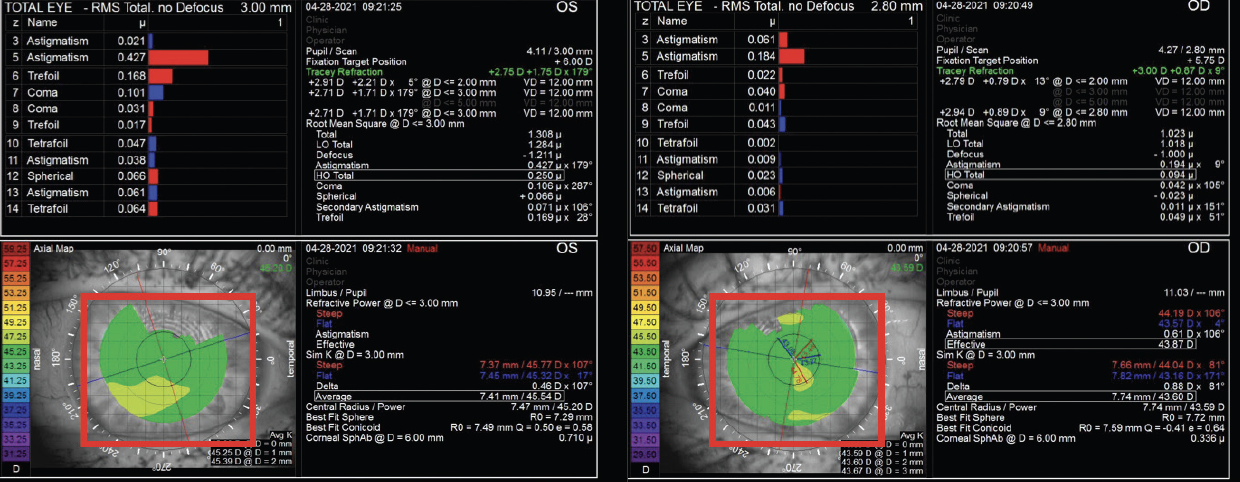
Figure 1. Preoperative corneal topography of a patient with significant epithelial basement membrane dystrophy shows irregular astigmatism in both the right and left eyes.
My preference is to perform a superficial keratectomy in the OR with a light diamond burr for polishing. A bandage contact lens is placed at the conclusion of surgery. Depending on the patient’s age and whether dry eye disease is present, I may place a Prokera device (BioTissue) instead to accelerate surface healing. I wait 2 months after superficial keratectomy before obtaining IOL measurements to promote adequate epithelial remodeling (Figure 2).
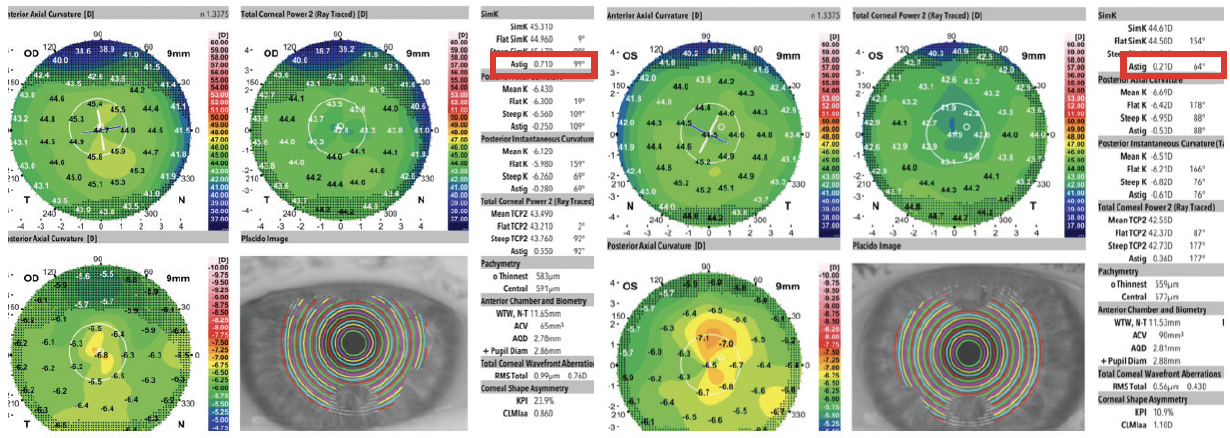
Figure 2. Two months after bilateral superficial keratectomy with a diamond burr and postoperative healing with a Prokera device, corneal topography demonstrates regular corneal astigmatism in both eyes.
Figures courtesy of Joseph G. Christenbury, MD
Addressing ocular surface pathology can set patients up for successful cataract surgery, expand their IOL options, and increase their opportunity for spectacle independence.
SURGICAL PLANNING
I review both topography (Atlas Corneal Topography System, Carl Zeiss Meditec) and Scheimpflug tomography (Galilei, Ziemer Ophthalmic Systems) for corneal irregularities such as ectasia and irregular astigmatism. Veracity Surgical (Carl Zeiss Meditec) integrates biometry (IOLMaster 700, Carl Zeiss Meditec) and topography measurements to create a surgical plan. I also regularly consult the ASCRS online calculator for prior RK. I tend to use the Barrett True K value and generally target a postoperative refraction of -0.25 to -0.50 D.
CONCLUSION
Patients with a history of RK can achieve excellent vision after cataract surgery. It is to be hoped that advances in IOL technology and power calculations allow them to achieve a greater degree of spectacle independence in the future.
1. Schanzlin DJ, Santos VR, Waring GO 3rd, et al. Diurnal change in refraction, corneal curvature, visual acuity, and intraocular pressure after radial keratotomy in the PERK Study. Ophthalmology. 1986;93(2):167-175.
2. Younan C, Mitchell P, Cumming RG, Rochtchina E, Wang JJ. Myopia and incident cataract and cataract surgery: the Blue Mountains Eye Study. Invest Ophthalmol Vis Sci. 2002;43(12):3625-3632.
3. Wang L, Koch DD. Intraocular lens power calculations in eyes with previous corneal refractive surgery: review and expert opinion. Ophthalmology. 2021;128(11):e121-e131.


