Patients judge the success of their lens surgery by the improvement in their functional vision and quality of life. Their judgement is based on their binocular visual perception and not on the vision in each eye. As surgeons, our aim should be to create a binocular visual percept compatible with excellent functional vision and meet the following criteria:
No. 1. A seamless, continuous range of vision from infinity to 40 cm;
No. 2. The highest possible contrast sensitivity and quality of vision; and
No. 3. Minimal, if any, dysphotopsias.
The binocular visual percept is generated when the two retinal images merge and a single context-appropriate visual percept is generated by the visual cortex and other cortical areas of visual processing. We have a limited understanding of these processes but are familiar with neural adaptation after IOL implantation.1 IOLs allow us to generate retinal images based on their optical design. The challenge is to generate a pair of retinal images that, after cortical processing and neural adaptation, achieve the three objectives described earlier.
WHAT IS BINOCULAR IOL CUSTOMIZATION?
Binocular customization is the process of choosing an IOL for each eye to produce the best binocular vision in terms of range of vision, contrast sensitivity, and dysphotopsias. The aim is to modulate the retinal image in each eye so that binocular functional vision is optimized. Binocular customization should not be confused with a mix-and-match strategy in which every patient always ends up with different IOLs.
A common strategy is to choose the same IOL for both eyes out of a mistaken belief that the patient will not perceive a difference in vision between eyes. In reality, interocular differences in residual defocus, torus, higher-order aberrations, pupil size, angle kappa, and ocular dominance can alter the retinal image even though the same IOL was implanted in both eyes. Almost all patients with the same IOL in both eyes can easily identify interocular differences in vision.
Always using the same IOL in both eyes ignores the possibility of modulating the retinal image in each eye to generate an optimal binocular vision percept. Selecting an IOL for each eye on the basis of range and quality of vision as well as dysphotopsia is a better strategy because it gives us the option of leveraging IOL optical design and/or refractive targeting to modulate the retinal image in each eye.
Binocular customization based on IOL profile. All presbyopia-correcting IOLs sacrifice quality of vision and induce dysphotopsia in exchange for a proportional extended range of vision. High-add multifocal IOLs provide the widest range of vision but have the poorest contrast sensitivity and induce the most dysphotopsias. Low-add multifocal IOLs compromise contrast sensitivity less and induce less dysphotopsia compared to high-add multifocal IOLs. Extended depth of focus IOLs provide a narrower range of vision than multifocals but provide comparatively improved contrast sensitivity and produce fewer dysphotopsias. New-generation monofocal IOLs, which some refer to as monofocal plus IOLs, provide a wider range of vision than standard monofocals but not as wide as extended depth of focus and multifocal IOLs. Monofocal plus IOLs, however, have, nearly identical contrast sensitivity, quality-of-vision, and dysphotopsia profiles as aspheric monofocal IOLs.
When a multifocal IOL is implanted bilaterally, the visual cortex does not obtain a full-contrast and dysphotopsia-free image from either eye. This reduces the cortical potential to generate high-contrast binocular perception. In contrast, the visual cortex of a patient who has a monofocal or monofocal plus IOL in one eye and a multifocal IOL in the other eye receives a high-contrast image from the eye with the monofocal IOL and a reduced-contrast image from the eye with the multifocal IOL. As a result, the visual cortex has the potential to process the two images to the higher-contrast image. A seminal study by Lida et al demonstrated that binocular contrast sensitivity was better than the contrast sensitivity in the multifocal eye of patients implanted with a monofocal IOL in their dominant eye and a multifocal IOL in their nondominant eye. They also reported an excellent range and quality of binocular vision in these patients.2
Binocular customization based on refractive targeting. Another way to modulate the retinal images is by customizing the refractive target in each eye using standard monovision (myopic anisometropia of 1.50–2.00 D), mini-monovision (myopic anisometropia of 1.00–1.50 D), and nanovision. The degree of myopic anisometropia is proportional to binocular range of vision. Nanovision, on the other hand, induces a minimal amount of anisometropia within the physiologic range of 1.00 D. Nanovision is thus consistent with binocular fusion where binocular contrast is higher than uniocular contrast.3
A key advantage of modulating the retinal image with refractive target customization is that reversal is easily achieved by wearing spectacles when needed. For example, spectacles may be worn at night to suppress the halos and glare induced by the myopia in the near eye.
Refractive target and IOL design strategies can be combined in a binocular IOL customization process to produce the widest range of binocular vision with the least amount of dysphotopsias and the least compromise to quality of vision.
FUNCTIONAL VISION TOOL
My colleagues and I are developing a software system to help surgeons implement binocular customization in their practices. The Panfocal-VA is a functional vision tool that can objectively assess patient needs in terms of their focal range, quality of vision, and dysphotopsias.
The Panfocal-VA is used preoperatively to choose the IOL for the first eye and again 1 to 2 weeks after surgery to assess the patient’s vision. The IOL for the second eye is then chosen based on the Panfocal-VA assessment and the patient’s subjective feedback. The goal is to provide the widest range of vision with the least compromise to quality of vision while minimizing dysphotopsias.
The following is my basic algorithm for binocular IOL customization with the Panfocal-VA. If the patient is happy with all aspects of their vision in the first eye, the same IOL and refractive target are used for the second eye. If they wish to improve their range of vision for near, a full-add multifocal IOL is selected. If they desire sharper distance vision or are struggling with halos and glare, a new-generation monofocal IOL is chosen.
Recently, a 53-year-old man with hyperopia and poor vision at all distances desired better spectacle-free vision after refractive lens surgery. A key consideration was his desire for spectacle-free near vision. A 25.50 D Tecnis Synergy IOL (Johnson & Johnson Vision) was implanted in the first eye (Figure 1). Postoperatively, his uncorrected distance visual acuity was 6/6, and his uncorrected near visual acuity was N5 (Figure 2A). The patient was able to perform all near and intermediate vision tasks without wearing spectacles, but he wished that the sharpness and quality of his distance vision were better. A Tecnis Eyhance IOL (Johnson & Johnson Vision) was implanted in the second eye. After surgery, the patient’s uncorrected binocular vision was 6/5 for distance and intermediate, 6/6 for near in photopic conditions, and 6/7.5 for near in mesopic conditions. He noticed the change in the quality of his distance vision after surgery on his second eye and was pleased with the final outcome (Figure 2B).
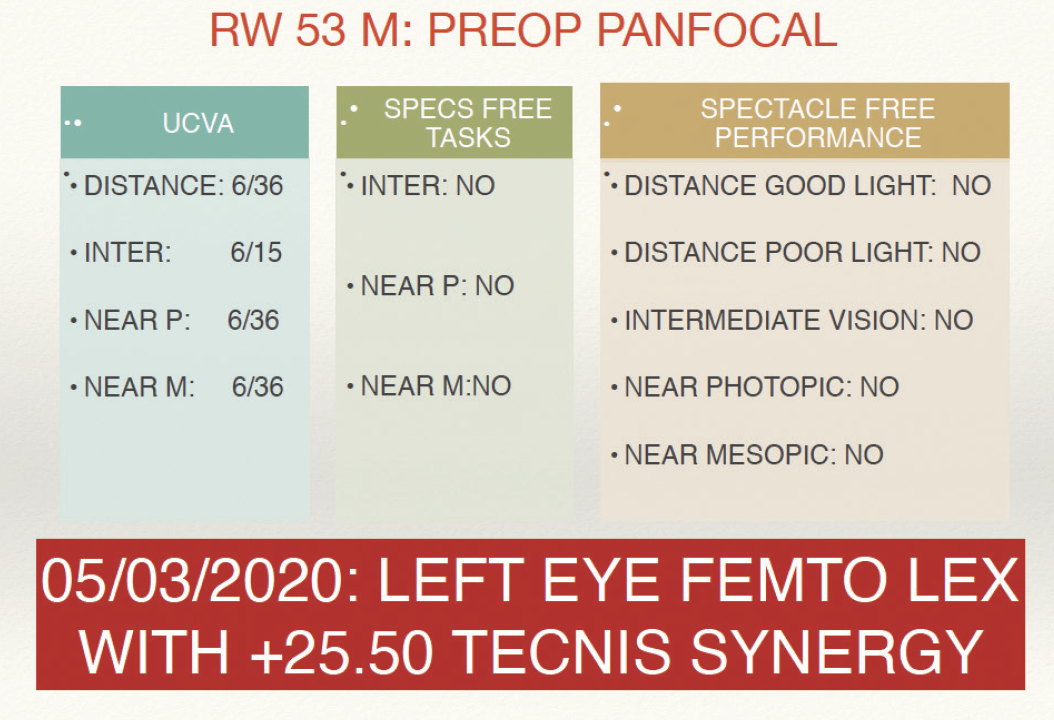
Figure 1. Panfocal-VA testing indicated that a 25.50 D Tecnis Synergy IOL was the appropriate choice for the patient’s first eye.

Figure 2. An assessment with the Panfocal-VA after cataract surgery on the first (A) and second (B) eyes.
CONCLUSION
I believe that binocular IOL customization is crucial to achieving excellent functional visual outcomes. It requires the surgeon to think in terms of generating an excellent binocular vision profile instead of following the dogma of implanting the same IOL in both eyes.
1. Zhang L, Lin D, Wang Y. Comparison of visual neuroadaptations after multifocal and monofocal intraocular lens implantation. Front Neurosci. 2021;15:648863.
2. Iida Y, Shimizu K, Ito M. Pseudophakic monovision using monofocal and multifocal intraocular lenses: hybrid monovision. J Cataract Refract Surg. 2011;37(11):2001-2005.
3. Pardhan S, Gilchrist J. The effect of monocular defocus on binocular contrast sensitivity. Ophthal Physiol Opt. 1990;10:33-36.



