

Astigmatism is one of the main factors affecting vision quality. Around 30% of cataract surgery candidates have greater than 1.00 D of corneal astigmatism.1,2 In a large, cross-sectional study in Western China, the amount of corneal astigmatism was between 0.25 and 1.25 D in 53.71% of eyes, 1.25 D or higher in 39.06% of eyes, and lower than 0.25 D in 7.23% of eyes.3 Based on these statistics, we believe that toric IOLs should be implanted in at least 30% and up to 80% of patients undergoing refractive cataract surgery if the cost is not a problem and if keratometry readings are accurate.
In 2020, 73% of the IOLs implanted in our practice were toric lenses, but optimizing vision quality with these IOLs can be challenging. In this article, we share our experience and how our approach has evolved since we began using toric IOL technology in 2010 (see The Evolution of Our Experience With Toric IOLs below).
THE EVOLUTION OF OUR EXPERIENCE WITH TORIC IOLS
2010: The Beginning
In 2010, we were using limbal relaxing incisions (LRIs) created with a diamond knife to correct astigmatism in cataract surgery patients. This technique could not address high amounts of astigmatism, but it was fairly effective for low amounts. Our journey with toric IOLs began when we started proposing a copayment for toric IOL implantation to our patients with high astigmatism while continuing to treat others with LRIs. Clinical efficiency decreased owing to the time required to mark the horizontal axis of the seated patient, and toric IOL power calculations were performed based only on anterior keratometry.
2012: Posterior Corneal Astigmatism
In his 2012 Charles D. Kelman Lecture at the 2012 ASCRS Annual Meeting, Douglas D. Koch, MD, reminded us of the contribution of posterior corneal astigmatism (PCA) to total corneal power and its importance in optimizing IOL power calculations.1,2 Even though this idea had been reported in the literature, PCA was not used in daily practice at that time owing to a lack of technology to measure it.
After hearing Dr. Koch’s lecture, we decided to use the Pentacam (Oculus Optikgeräte) to perform Scheimpflug imaging to analyze our patients’ PCA. We analyzed 146 eyes and found a mean PCA of -0.53 D (±0.05) at 86°. This was consistent with the literature. We voluntarily left +0.50 D at 90° in patients who were 70 years old or younger so that we could perform calculations knowing the total corneal power. This individual measurement enabled us to achieve good postoperative refractive astigmatism results.
2014: Image-Guided Surgery
In 2014, we began using the Callisto eye (Carl Zeiss Meditec) surgical guidance system with an external data injection system. Our prospective analysis of 146 eyes found that, although this device did not provide greater accuracy than manual marking (91% vs 90.5% of toric IOLs between 0° and 10°), it improved clinical workflow and increased patient comfort by reducing procedural time by 3 minutes per eye.
At this time, the volume of toric IOLs we implanted had started to increase, but we still used LRIs to correct astigmatism in many cases.
2016: More Toric IOLs
I (G.L.) was invited by Graham D. Barrett, MB BCh SAf, FRACO, FRACS, to speak about multifocality at the 2016 Australasian Society of Cataract and Refractive Surgeons Annual Conference (Figure). While I was there, several of my Australian colleagues were surprised by how often I performed LRIs. They encouraged me to use more toric IOLs instead. My response was that it was a matter of cost; there is better reimbursement in Australia than in France where I practice. The question was whether patients would agree to pay out of pocket for a better quality of vision.
In 2017, we conducted a retrospective study of 407 eyes that received a toric IOL (AT Torbi 709MP, Carl Zeiss Meditec) in our practice in 2016. Mean preoperative total corneal astigmatism was 1.24 D (standard deviation [SD] ±0.74), and mean postoperative refractive astigmatism was 0.17 D (SD ±0.29). The Figure shows the cumulative percentage of the postoperative refractive cylinder (unpublished data, 2017).

Figure. Raphaël Ervinckx of Beaver-Visitec International (left); Roberto Bellucci, MD, FEBOS-CR (center, left); Dr. Barrett (center, right); and Dr. Lesieur (right) at the 2016 Australasian Society of Cataract and Refractive Surgeons Annual Conference.
After flying home and considering the matter for a few days, I (G.L.) decided to stop performing LRIs when a suitably powered toric IOL was available. The number of toric IOLs implanted in our practice increased, and in October 2016, we purchased an IOLMaster 700 (Carl Zeiss Meditec) in an effort to provide better results.
1. Koch DD, Ali SF, Weikert MP, Shirayama M, Jenkins R, Wang L. Contribution of posterior corneal astigmatism to total corneal astigmatism. J Cataract Refract Surg. 2012;38(12):2080-2087.
2. Koch D, Jenkins R, Weikert M, Yeu E, Wang L. Correcting astigmatism with toric intraocular lenses: Effect of posterior corneal astigmatism. J Cataract Refract Surg. 2013;39:1803-1809.
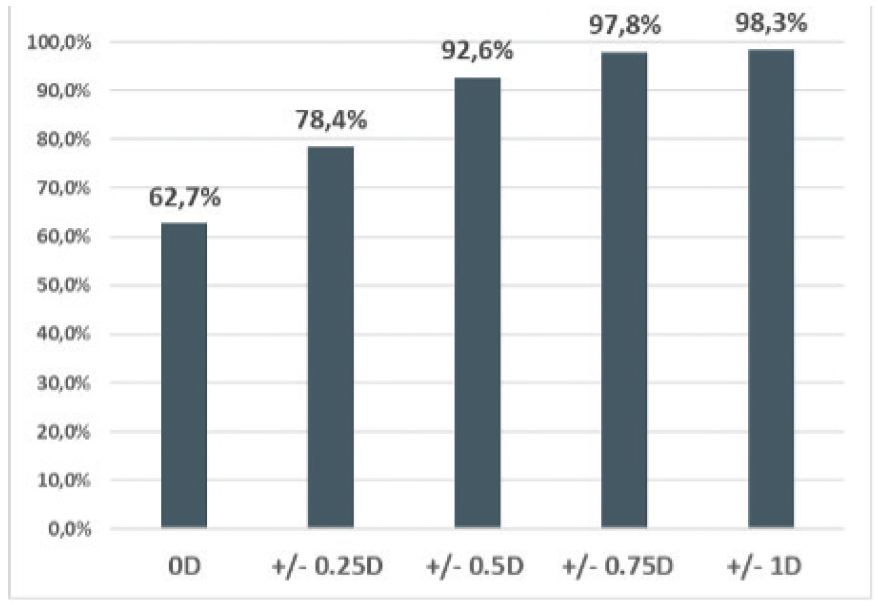
Figure. The cumulative percentage of the postoperative refractive cylinder in 407 eyes implanted with the AT Torbi 709MP at the Centre IRIDIS in 2016.
The mean results were good, but we were concerned about postoperative rotation of the toric IOL in the capsular bag, which reduced the amount of astigmatism corrected. In some instances, we had to realign the toric IOL. Based on the data, we wanted to look for a way to stabilize toric IOLs, so we decided to consider the correlation between the capsular bag size and the white-to-white (WTW) distance.
The WTW Hypothesis
Promising findings. After a 1-year analysis of the biometric parameters of the eyes in which toric IOLs had rotated the most, we realized that rotation occurred as frequently in long eyes as it did in short eyes. Erb-Eigner et al4 reported a correlation between the WTW corneal diameter and capsular bag size, so we decided to implant a larger-diameter 11.4-mm toric IOL (Ankoris, Beaver-Visitec International) in eyes with a larger WTW distance (≥ 12 mm). In eyes with a WTW distance that was less than 12 mm, we implanted an 11-mm–diameter toric IOL (AT Torbi 709MP, Carl Zeiss Meditec).
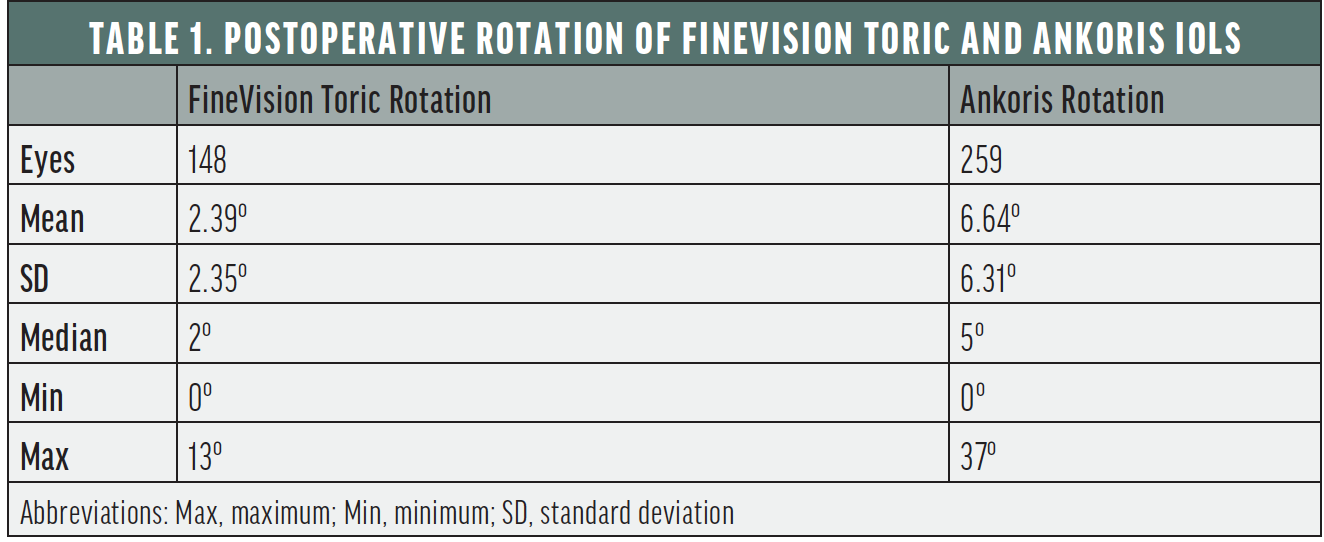
The results of our analysis suggested that WTW measurements could be correlated with postoperative rotation of a toric IOL. Mean rotation was 3.7° (±5.3°) with the AT Torbi 709MP and 6.7° (±5.4°) with the Ankoris.
During this time, we also initiated a retrospective comparative analysis of the stability of two IOLs of the same design and material (26% hydrophilic acrylic toric IOL) but with different optics—a multifocal (FineVision Toric POD-FT, Beaver-Visitec International) and a monofocal (Ankoris). A swept-source OCT feature on the IOLMaster 700 (Carl Zeiss Meditec) allowed us to obtain accurate and repeatable measurements of the total keratometry.
Table 1 shows the rotation values of the two IOLs in our analysis (unpublished data, 2019). We found a marked reduction in rotation by an average 4.25º with the multifocal toric. Based on these data, we concluded that IOLs with a nonpolished diffractive zone are generally more stable in the bag, which supported findings reported by Vandekerckhove in 2018.5
Invalidated hypotheses. In a 2020 article, we reported better absolute astigmatism prediction error results when true keratometry was measured rather than estimated.6
To confirm our WTW hypothesis, we used an 11-mm diameter toric (AT Torbi 709-MP) in eyes with a large WTW corneal diameter (≥ 12 mm) and a larger-diameter 11.4-mm toric (Ankoris) in eyes with a small WTW corneal diameter (< 12 mm). We implanted 45 Ankoris IOLs in eyes with a small WTW corneal diameter and found that the lens rotated to a greater degree in these eyes than when it was implanted in eyes with a large WTW corneal diameter (Table 2). We therefore concluded that WTW was not a good predictor of postoperative IOL rotation, and our hypothesis was invalidated (unpublished data, 2020).

We set out to find another reason for IOL rotation. A retrospective analysis of a large sample of eyes implanted with a toric IOL in our practice showed no correlation between postoperative rotation and preoperative biometric parameters or between postoperative rotation and axial length (unpublished data, 2020).
A Successful Approach
At the end of 2020, we began implanting an unpolished extended depth of focus toric IOL (Lucidis MT, Swiss Advanced Vision) that is available in two diameters, 10.8 mm (Lucidis 108MT) and 12.4 mm (Lucidis 124MT). We hoped that the large diameter and unpolished surface of these IOLs would optimize their stability.
We noted some rotation and decentration with the small-diameter 10.8-mm IOL, so we began using the larger-diameter 12.4-mm lens and achieved impressive results. We found a median rotation of 2º at 3 months postoperatively with this IOL (n = 113). In 50% of eyes analyzed, this IOL rotated by 2º or less and never more than 11º.7
We found that the large diameter of the 12.4-mm IOL caused anterior capsular contraction syndrome in some eyes, so we enlarged the diameter of the capsulorhexis to 5.5 mm and opted for the 10.8-mm IOL in short eyes (axial length < 21.5 mm) and eyes with a WTW corneal diameter of less than 11 mm.
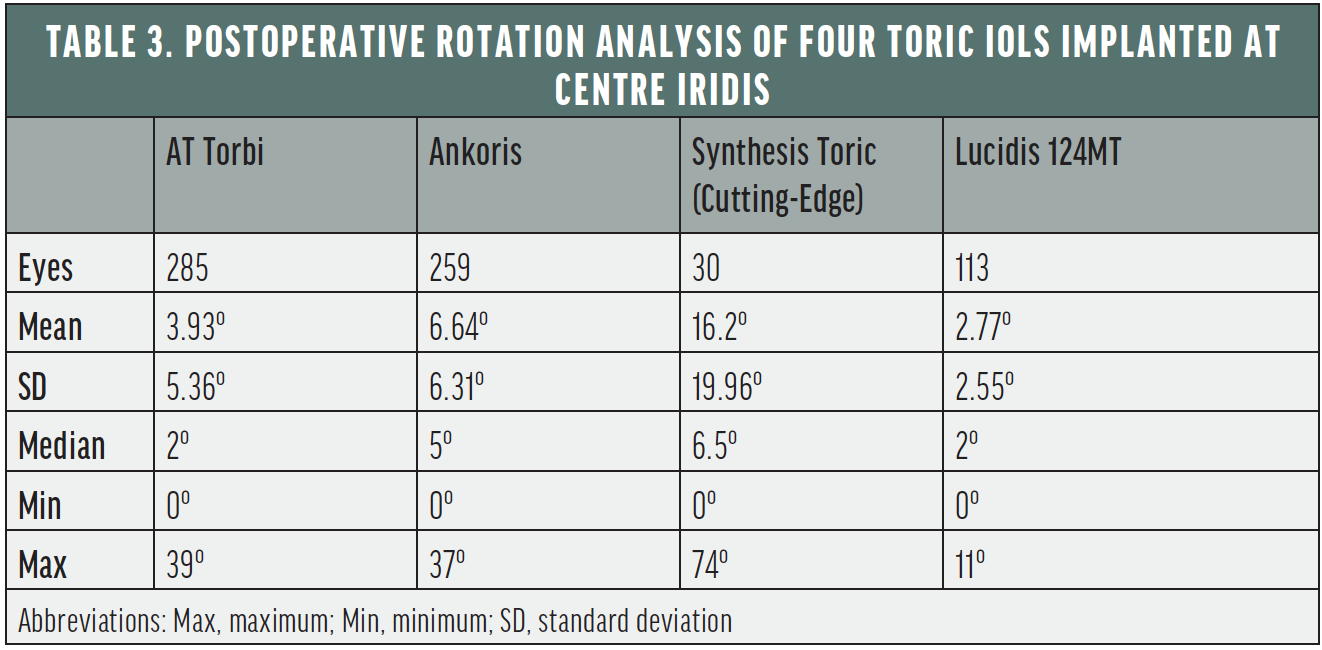
Table 3 compares the postoperative rotation of the four toric IOLs we analyzed over the years.
Conclusion
Evolutions in technology, total keratometry measurement, and patient care (including optimizing the corneal surface before biometry) provide the opportunity to expand the pool of candidates for toric IOLs to up to 80% of patients. Unfortunately, the toric IOLs available today cannot meet the visual needs of all of these patients because postoperative rotation remains a key issue.
Finding a truly stable IOL is no easy task, yet it is crucial to correct astigmatism. Years of analysis, successes, and mistakes have shown that the diameter of the IOL and an unpolished surface can increase IOL adhesion to the capsular bag and, therefore, reduce postoperative rotation. Postoperative rotation, however, can still occur with these factors accounted for. Developing a smart and simple IOL that can completely correct presbyopia and toricity is the next frontier.
1. Hoffmann PC, Hütz WW. Analysis of biometry and prevalence data for corneal astigmatism in 23,239 eyes. J Cataract Refract Surg. 2010;36(9):1479-1485.
2. Khan MI, Muhtaseb M. Prevalence of corneal astigmatism in patients having routine cataract surgery at a teaching hospital in the United Kingdom. J Cataract Refract Surg. 2011;37(10):1751-1755.
3. Ma W, Zuo C, Chen W, et al. Prevalence of corneal astigmatism in patients before cataract surgery in Western China. J Ophthalmol. 2020;6.
4. Erb-Eigner K, Hirnschall N, Hackl C, Schmidt C, Asbach P, Findl O. Predicting lens diameter: ocular biometry with high-resolution MRI. Invest Ophthalmol Vis Sci. 2015;56(11):6847-6854.
5. Vandekerckhove K. Rotational stability of monofocal and trifocal intraocular lenses with identical design and material but different surface treatment. J Refract Surg. 2018;34(2):84-91.
6. Lesieur G. Microincision cataract surgery with implantation of a bitoric intraocular lens using and enhanced program for intra ocular lens power calculation. Eur J Ophthalmol. 2020;30(6):1308-1313.
7. Lesieur G. Refractive and visual outcomes of a new extended depth of focus (EDOF) intraocular lens. Paper presented at: 39th Congress of the European Society of Cataract and Refractive Surgery; October 8-12, 2021; Amsterdam, Netherlands.


