

Cataract surgeons commonly encounter patients with concomitant cataracts and glaucoma. When cataract surgery is planned for these patients, it is important for the surgeon to keep both conditions in mind. This article lists five pearls that every cataract surgeon should know about glaucoma. We hope to provide tips to help the surgeon successfully manage both conditions.
Pearl #1. PREOPERATIVE EVALUATION TAKES EXTRA TIME
Glaucoma is the most common cause of irreversible blindness worldwide.1 As such, one must always be on the lookout for possible undiagnosed glaucoma. Additionally, because the symptoms of glaucoma and cataract can often overlap, it is important to evaluate each disease process prior to surgery. Glaucoma, like cataract, exists on a spectrum. Understanding this will allow you to provide an accurate diagnosis, conduct proper surgical planning, and set realistic patient expectations regarding outcomes for both diseases.
If a patient presents for cataract surgery with preexisting glaucoma, the stage and type of glaucoma should be noted, as this can affect surgical planning. For instance, patients with pseudoexfoliative glaucoma may be at higher risk for cataract surgical complications, and those with mild to moderate glaucoma might benefit from an additional microinvasive glaucoma surgery (MIGS) procedure performed at the time of cataract surgery. (More on MIGS below and in the article by Magda Rau, MD, Tips for MIGS.) A complete examination should be conducted, including gonioscopy and assessment of any visual fields and OCT imaging available.
In patients who have had previous glaucoma surgery, the surgeon should anticipate any alterations to his or her usual surgical technique. This may include altering one’s standard incision site to avoid a functioning bleb, employing pupil expansion techniques, and avoiding contact with existing intraocular devices such as a drainage shunt.
The surgeon must also take a little extra time to discuss with the patient that the two diseases interact. This helps set the stage for any management that is needed following surgery, and it helps the patient understand that cataract surgery can affect his or her glaucoma and vice versa. Patients should know that visual recovery can take a bit longer in glaucoma patients. These patients are also at increased risk for IOP spikes immediately following surgery, and for prolonged IOP elevation following surgery. (More on IOP spikes below.) It should also be emphasized that the patient may need more frequent follow-up and perhaps the addition of medications or other treatment if his or her IOP is not adequately controlled on the antiglaucoma regimen the patient was using before surgery.
Pearl #2. IOL CHOICE DEPENDS ON GLAUCOMA STATUS
When deciding what IOL to implant following cataract removal, one must consider how glaucoma can affect vision and the limitations of the current range of IOL implants available. Understanding this will help to guide IOL selection.
Patients with glaucoma can have decreased contrast sensitivity. In pseudoexfoliative glaucoma, there is increased risk of zonular weakness and capsular contraction that can lead to decentration of the implant. The use of topical medications can lead to ocular surface disease, and this may need to be optimized prior to IOL power calculation and lens selection. If a filtering bleb is present, there may be induced astigmatism. All of these factors can weigh on IOL choice.
Modern options for IOL selection include aspheric IOLs, astigmatism-correcting toric IOLs, diffractive or refractive multifocal IOLs, accommodating IOLs, and others. Additionally, there are multiple design platforms supporting these lens types. All of this must be taken into consideration when selecting a lens.
Aspheric IOLs help to improve contrast sensitivity while also attempting to decrease higher-order aberrations (HOAs). However, if these lenses become decentered, they can instead induce HOAs. In patients with loose zonules, other alternatives should be considered.
Corneal astigmatism can be addressed at the time of surgery with toric IOLs or limbal relaxing incisions (LRIs). Again, one must consider the possibility of lens decentration or movement, as rotation of a toric IOL can reduce its effect. A rotation of 30° from the desired axis will give the lens zero astigmatic effect, and greater rotation can induce additional astigmatism. Patients who have an existing bleb may have induced with-the-rule astigmatism. If the bleb has been of long standing and is stable, correction of the induced astigmatism should be considered.
Patients who want to be less dependent on spectacles also have options. Multifocal IOLs can help patients achieve good quality vision at both distance and near, although glasses may still be needed for certain activities. Multifocal IOLs do have downsides, as patients may experience glare and halos with these lenses. Decreased contrast sensitivity is another characteristic of these lenses, and, in glaucoma patients who may already have compromised contrast sensitivity, these may not be the best choice. One small study suggested that glaucoma patients may benefit from multifocal lens implantation, but this remains controversial.2
Accommodating IOLs provide another option, as their optic designs do not cause a decrease in contrast sensitivity. However, in the setting of zonular instability, accommodating IOLs should be avoided.
Lastly, mini-monovision using standard IOLs can be considered. This can be achieved by targeting plano in the dominant eye and -0.75 to -1.25 D in the nondominant eye, depending on the near vision requirements of the patient. This will give patients good distance and intermediate vision, although they will still have to rely on glasses for near work or small print. In patients with significant visual field loss, however, only one eye may be providing adequate peripheral vision, and this must be taken into account.
Pearl #3. FEMTOSECOND LASER-assisted cataract SURGERY IS SAFE
IOP can increase both during and after cataract surgery, and patients with a diagnosis of glaucoma are more sensitive to these changes. During phacoemulsification, IOP can be significantly elevated to about 75 mm Hg when the bottle height is at 100 cm, although it may be slightly lower due to wound leaks and other factors during surgery.
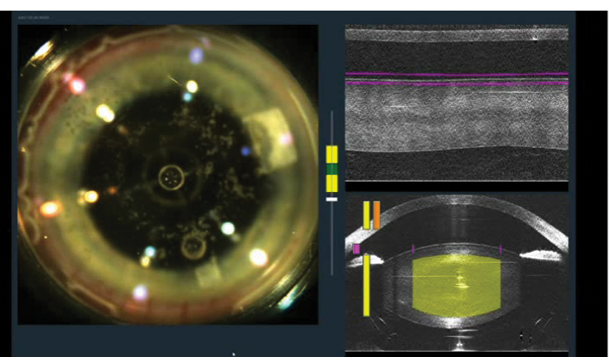
Figure 1. Incisions are created with the femtosecond laser in a patient who is undergoing combined cataract and glaucoma surgery.
Initially there were concerns about IOP elevation during the femtosecond laser portion of laser-assisted cataract surgery (LACS). However, the safety of the procedure, including intraoperative IOP levels, has now been studied thoroughly. On average, IOP increase from baseline is about 10 to 16 mm Hg during the laser procedure, and IOP returns to baseline shortly thereafter. This transient IOP rise, for about 30 to 90 seconds depending on surgeon preferences and platforms, is well tolerated in both glaucomatous and nonglaucomatous eyes.3-5 All things considered, patients with glaucoma should not be denied the opportunity of femtosecond LACS (Figure 1; http://eyetube.net/?v=sutha). This is also an excellent time to consider performing laser LRIs in a patient with astigmatism.
Pearl #4. COMBINING CATARACT SURGERY AND MIGS CAN LOWER IOP
Cataract surgery has been shown to provide a small (1 to 2 mm Hg) decrease in IOP.6,7 Although this may be beneficial, the change may not be sufficient to provide adequate IOP control. MIGS techniques are quickly becoming a mainstay for cataract surgeons. In general, these procedures can be performed at the same time as cataract surgery, spare the conjunctiva, have excellent safety profiles, and can help lower IOP more than cataract surgery alone. They are also astigmatism-neutral and should not affect the refractive outcome of cataract surgery.
These procedures are changing the way glaucoma patients are managed by providing additional IOP control for those who do not need a more invasive approach. Ideal patients include those with mild to moderate glaucoma who will be undergoing cataract surgery, although the indications continue to expand.
Watch it Now
Dr. Berdahl highlights the use of a femtosecond laser to assist in a cataract procedure and finishes with implantation of the iStent.
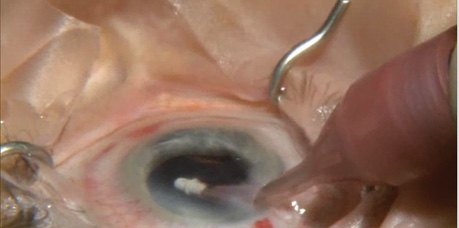
Multiple MIGS options are available, and many more are in development. Two devices currently available are the Trabectome (Neomedix) and the iStent (Glaukos). Endocyclophotocoagulation with the E2 laser and endoscopy system (Endo Optiks) is another possibility. Other devices under development include the Hydrus microstent (Ivantis), the CyPass microstent (Transcend Medical), and the AqueSys microfistula implant (AqueSys).
In patients who have advanced glaucoma and inadequate IOP, a more invasive procedure such as trabeculectomy or tube shunt implantation can be considered, but the safety of MIGS approaches makes these the preferred options. Thorough discussion of these techniques and their timing are beyond the scope of this article.
In addition to offering therapeutic options, MIGS procedures can also provide quality of life improvement, as patients may become less dependent on topical medications. With so many MIGS options available or in development, patients and surgeons have considerable flexibility to address both cataract and glaucoma.
Pearl #5. BEWARE OF POSTOPERATIVE IOP SPIKES
Postoperative management following cataract surgery in glaucoma patients may be different from management after standard procedures. A few things to keep in mind are that IOP rise occurs within the first 8 to 12 hours after surgery in normal eyes, and it may be higher and more prolonged in glaucoma patients. Glaucoma patients also are more likely to experience postoperative IOP spikes.8
Many surgeons add some form of IOP-lowering medication to their postoperative treatment regimen. This usually takes the form of a topical beta-blocker given at the completion of surgery to help limit or prevent IOP spikes.9 The patient should also continue his or her previous glaucoma treatment regimen to maintain IOP control.
Should a postoperative IOP spike occur, we suggest beginning topical treatment in the office with a fixed combination of timolol and dorzolamide or timolol and brimonidine to lower IOP. The patient can then continue this regimen for 1 week or longer until his or her IOP is controlled. Medication choice may vary based on what is available and on patient tolerance. Some surgeons may also choose to taper the use of topical steroids after cataract surgery to prevent steroid-induced IOP elevation because glaucoma patients are more sensitive to steroid-induced IOP response. The risk of this is very low, however.10 This applies only for patients who have not had previous glaucoma surgery.
In patients who have had previous glaucoma surgery, the surgeon should aim to reduce any inflammation in order to avoid complications with a functioning bleb. Even with the cleanest technique, blebs are at increased risk of failing following cataract surgery.11 This may require an increase in frequency of or a prolonged duration of topical steroids after cataract surgery to prevent bleb failure.
As noted above, glaucoma patients should be followed closely, as almost a third of patients may need additional medications or procedures to keep IOP under control.12 Having a discussion of this likelihood with patients prior to surgery will help to prevent surprises in the postoperative period.
CONCLUSION
Patients with concomitant cataract and glaucoma will continue to be a growing part of the cataract surgeon’s practice. Because patient presentations will vary, it is important to remember the five things described above in order to provide the best possible individual outcomes, keeping both you and your patients happy. n
1. Resnikoff S, Pascolini D, Etyaále D, et al. Global data on visual impairment in the year 2002. Bull World Health Organization. 2004;82:844-851.
2. Kamath GG, Prasad S, Danson A, Phillips RP. Visual outcome with the array multifocal intraocular lens in patients with concurrent eye disease. J Cataract Refract Surg. 2000;26(4):576-581.
3. Donaldson K, Braga-Mele R, Cabot F, et al. Femtosecond laser-assisted cataract surgery. J Cataract Refract Surg. 2013;39:1753-1763.
4. Schultz T, Conrad-Hengerer I, Hengerer FH, Dick HB. Intraocular pressure variation during femtosecond laser–assisted cataract surgery using a fluid-filled interface. J Cataract Refract Surg. 2013;39:22-27.
5. Kerr NM, Abell FG, Vote BJ, Toh T’. Intraocular pressure during femtosecond laser pretreatment of cataract. J Cataract Refract Surg. 2013;39:339-342.
6. Shingleton BJ, Pasternack JJ, Hung JW, et al. Three and five year changes in intraocular pressures after clear corneal phacoemulsification in open-angle glaucoma patients, glaucoma suspects, and normal patients. J Glaucoma. 2006;15(6):494-498.
7. Rao A. Diurnal curve after phacoemulsification in patients with pseudoexfoliation syndrome and cataract. Semin Ophthalmol. 2012;27(1-2):1-5.
8. Tranos P, Bhar G, Little B. Postoperative intraocular pressure spikes: the need to treat. Eye (Lond). 2004;18(7):673-679.
9. Kanellopoulos AJ, Perry HD, Donnenfeld ED. Timolol gel versus acetazolamide in the prophylaxis of ocular hypertension after phacoemulsification. J Cataract Refract Surg. 1997;23(7):1070-1074.
10. Chang DF, Tan JJ, Tripodis Y. Risk factors for steroid response among cataract patients. J Cataract Refract Surg. 2011;37(4):675-681.
11. Husain R, Liang S, Foster PJ, et al. Cataract surgery after trabeculectomy: the effect on trabeculectomy function. Arch Ophthalmol. 2012;130(2):165-170.
12. Slabaugh MA, Bojikian KD, Moore DB, Chen PP. The effect of phacoemulsification on intraocular pressure in medically controlled open-angle glaucoma patients. Am J Ophthalmol. 2014;157(1):26-31. doi: 10.1016/j.ajo.2013.08.023.
John P. Berdahl, MD
• Vance Thompson Vision, Sioux Falls, South Dakota
• johnberdahl@gmail.com
• Financial disclosure: Lecture fees (Alcon, Allergan, Glaukos), Consultant (Alcon, Allergan, Avedro, Bausch + Lomb, Clarvista Medical, Envisia, Equinox, Glaukos, Omega Ophthalmic, Ocular Surgical Data, Vittamed), Equity Owner (Equinox, Omega Ophthalmic, Ocular Surgical Data)
Michael D. Greenwood, MD
• Vance Thompson Vision, Sioux Falls, South Dakota
• migreenw@gmail.com
• Financial disclosure: None

