
In recent years, a number of microstent devices for glaucoma have been developed, and their use is generally classified under the heading of microinvasive glaucoma surgery (MIGS). These devices include ab externo approaches that aim at subconjunctival filtration, such as the Ex-Press Glaucoma Filtration Device (Alcon), which relies on the same mechanisms of function as trabeculectomy but in a more controlled form. Aiming also at the subconjunctival space, but from an ab interno approach, is the Xen Gel Stent (AqueSys). Other implants, including the Hydrus Microstent (Ivantis) and the first- and second-generation iStent Trabecular Micro-Bypass Stent (Glaukos) target the Schlemm canal, while the third-generation iStent and the CyPass microstent (Transcend Medical) aim ab interno at the suprachoroidal space. The InnFocus MicroShunt (previously the Midi-Arrow; InnFocus) renews the concept of subconjunctival filtration with an ab externo drainage device but using a simple tube in a minimally invasive fashion.
IOP-lowering medications are still the first line treatment in glaucoma management. However, in 5% to 10% of patients they are not well tolerated or sufficient enough to halt disease progression. In these cases, surgery is necessary. Traditional surgical methods are relatively invasive, especially trabeculectomy. Microstents provide an opportunity to treat glaucoma in a less invasive manner and earlier in the disease process. They are procedurally efficient, minimally invasive, and tissue-sparing, and recovery is faster and with fewer complications than traditional procedures.
Although microstents do not lower IOP to the same degree as trabeculectomy, deep sclerectomy, or older tube shunts, they should be considered as surgical options, particularly as part of a combined procedure with cataract surgery (see Advantages of Combined Procedures: MIGS and Cataract Surgery). With an added 10 minutes in surgery, a cataract patient with moderate IOP elevation can have an additional microstent procedure that controls IOP in an acceptable fashion.
PERSONAL ALGORITHM
In my own treatment algorithm for management of primary open-angle glaucoma, topical therapy is still the first treatment option. After long periods of daily topical application, however, some patients develop chronic ocular surface problems or general side effects that make further topical therapy impossible. Some patients also have objective problems with topical application caused by systemic conditions such as arthritis, Parkinson disease, or tremor. Subjective problems, such as reduced quality of life due to dependence on self-administered therapy, can also weigh on patients. Further, sometimes the maximum-tolerated medical therapy is not sufficient to effectively reduce IOP, and worsening is seen on visual fields, optic nerve examination, or retinal nerve fiber layer (RNFL) imaging.
For patients with severe glaucoma with IOP greater than 30 mm Hg, trabeculectomy or deep sclerotomy may be necessary. However, postoperative bleb management is demanding, and these procedures require an experienced glaucoma surgeon. Microstent-based MIGS approaches, on the other hand, are minimally invasive and effective glaucoma procedures that are more convenient for use in outpatient surgery clinics.
PERSONAL EXPERIENCE
Of the devices listed above, the microstents with which I have experience are the first- and second-generation iStents and the CyPass.
iStent. The first-generation iStent is the only ultrasmall glaucoma drainage device that has obtained both FDA approval and the CE Mark. It is an L-shaped titanium tube that connects the anterior chamber with the canal of Schlemm and allows aqueous outflow to bypass the trabecular meshwork. For localization of Schlemm canal, without the pigmentation in the trabecular meshwork, implantation is nearly impossible. Implantation is easier after cataract extraction because the Schlemm canal is then usually filled with blood.
At a Glance
• IOP-lowering medications are still the first line treatment in glaucoma management; however, microstents provide an opportunity to treat glaucoma in a less invasive manner and earlier in the disease process.
• The reduction of IOP caused by cataract surgery alone is not great enough to reduce or eliminate a patient’s need for glaucoma medications. The combination of cataract surgery and MIGS with a microstent has opened a new avenue for effective surgical treatment of open-angle glaucoma.
• With an added 10 minutes in surgery, a cataract patient with moderate IOP elevation can have an additional microstent procedure that controls IOP in an acceptable fashion.
The device is implanted with an inserter that carries the tip of the stent through a corneal incision of 1.5 to 1.8 mm and then drives it through the trabecular meshwork. A gonioscopic lens is used to visualize the maneuver.
The second-generation iStent is cone-shaped and needs only a 1-mm corneal incision; it is smaller than the first-generation device. By pressing a button on the inserter, the stent is plunged into the trabecular meshwork.
Both versions of the iStent are indicated in patients with early to mild glaucoma. In my own practice, most patients considered for iStent implantation have cataract and use up to three glaucoma medications. In these instances, a combined procedure is performed. Other candidates have early glaucoma and no cataract with high IOP despite maximal meds. We offer the iStent as a first step to surgery in these patients.
CyPass. My current favorite microstent is the CyPass, a polyimide device that provides a permanent conduit for outflow of aqueous humor from the anterior chamber to the suprachorodial space, aided by the natural negative pressure in the eye. The microstent is inserted into the supraciliary space using an application device that allows a lateral, ab interno, and clear corneal approach. The miniature stent is 6 mm in length, with a 300 µm inner diameter. Fenestrations along the microstent allow egress of aqueous.
Placement of the device is minimally invasive, spares the conjunctiva, avoids formation of a filtering bleb, and can be performed either as a standalone intervention or in combination with phacoemulsification.1,2 I offer the operation to patients with a diagnosis of open-angle glaucoma; iridocorneal angle of Shaffer grade 3 or 4; unmedicated or medicated IOP between 21 and 31 mm Hg, or with maximum two to three medication therapy; visual field defect on the glaucoma hemified test that is outside normal limits, or a pattern standard deviation at more than 5% level; vertical cup-to-disc ratio of at least 0.7; and OCT-demonstrated thinness of the RNFL.
Other indications include intolerance of topical medication due to allergy, red eye or pain after application, or systemic side effects. The wish of the patient for independence from drops and self-administered therapy is also considered.
Because of the noninvasive, safe characteristics of the CyPass implantation procedure, I also offer it to patients with severe glaucoma progression with only minimal remaining visual field. If an invasive trabeculectomy were performed at this advanced stage of glaucomatous disease, probably the last of the patient’s remaining visual field would be lost.
In surgery, the CyPass is loaded on the specialized curved guidewire of the applicator. As the microstent is inserted, it conforms to the curvature of the guidewire to follow the scleral contour along the supraciliary space (see CyPass Surgical Technique). The stiffness of the stent material, along with a series of retention rings at the proximal end, helps to ensure the stability of the device in the angle and the supraciliary space. The microstent is designed to increase aqueous outflow via the suprachoroidal space.3
At a Glance
After phacoemulsification and IOL implantation through a 2.2- to 2-8 mm clear corneal incision, acetylcholine is injected into the anterior chamber to achieve miosis. OVD (Healon; Abbott Medical Optics) is then injected to deepen the anterior chamber, especially in the area of the angle where CyPass implantation is planned.
With a goniolens, the angle is carefully visualized to choose the spot for CyPass implantation. Then the delivery device with the loaded microstent is inserted through the clear corneal incision into the anterior chamber and directed toward the angle on the opposite side. The CyPass device is positioned on a small guidewire with an atraumatic tip that gently separates the iris from the scleral spur. The atraumatic tip creates a small cleft under the scleral spur, and the CyPass is slowly advanced without resistance into the supraciliary, suprachorioidal space and released at the desired depth, leaving only the proximal collar and the first retention ring of the device in the anterior chamber (Figure 1A through 1C).
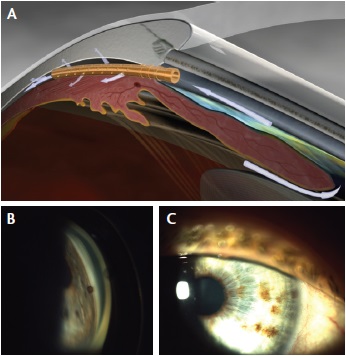
Figure 1. The CyPass is slowly advanced (A), leaving only the proximal collar and the first retention ring in the anterior chamber (B, C).
RESULTS
In a multicenter study and in an analysis of my own results, at 1-year follow-up, implantation of the CyPass resulted in reductions from mean preoperative baseline IOP and number of medications in patients undergoing standalone or combined procedures.1,2
For patients in my own study (n=33), the mean number of glaucoma medications was reduced, from 1.8 at baseline to 0.7 at 1 year for the combined surgery group, and from 2.2 to 0.8 for the standalone CyPass group. Mean IOP was also reduced, from 22.3 mm Hg at baseline to 15 mm Hg at 1 year for the combined surgery group, and from 26 to 13.2 mm Hg for the standalone CyPass group. In patients who have reached 3-year follow-up, reductions of IOP and number of medications have persisted.
CyPass migration can be addressed with a secondary repositioning procedure. This occurred in one case in my own series with no other clinical sequelae; this patient’s IOP is 13 mm Hg at 24 months, and he is on no medications.
In one case, we observed iris adherence to the CyPass device, and this was resolved by Nd:YAG laser coagulation of the adhered iris. In no case, even after 3 years, have we observed obstruction of the tube.
Thus far, I have implanted 98 CyPass microstents, with approximately 80% having been combined with cataract surgery. This procedure has almost replaced many other glaucoma procedures in our clinic.
CONCLUSION
The reduction of IOP caused by cataract surgery alone is not great enough to reduce or eliminate a patient’s need for glaucoma medications. The combination of cataract surgery and MIGS with a microstent has opened a new avenue for effective surgical treatment of open-angle glaucoma. As a standalone procedure, microstent implantion is also effective and safe, and it does not require intensive postoperative treatment as is the case with bleb-dependent surgeries. n
1. Grisanti S, Margolina E, Hoeh H, et al. Supraciliary microstent for open-angle glaucoma: clinical results of a prospective multicenter study. Ophthalmologe. 2014;111:548-552.
2. Hoeh H, Ahmed II, Grisanti S, et al. Early postoperative safety and surgical outcomes after implantation of a suprachoroidal micro-stent for the treatment of open-angle glaucoma concomitant with cataract surgery. J Cataract Refract Surg. 2013;39:431-437.
3. Saheb H, Ianchulev T, Ahmed II. Optical coherence tomography of the suprachoroid after CyPass Micro-Stent implantation for the treatment of open-angle glaucoma. Br J Ophthalmol. 2014;98:19-23.
Magda Rau, MD
• Head, Augenklinik Cham and Refractive Privatklinik-Dr.Rau, Cham, Germany
• Head, Eye Centre Prag, Czech Republic
• Member, CRST Europe Editorial Board
• info@augenklinik-cham.de
• Financial disclosure: Investigator, Consulting, Research Funding

