
Premium IOLs can provide improved quality of vision for our patients. Some ophthalmologists may not accept that premium IOLs can provide visual advantages over monofocal IOLs, but every ophthalmologist is obliged to tell patients about the potential benefits of aspheric, toric, and multifocal lenses.
Of course patients may perceive the advantages of these IOLs in different ways, and it is the patient’s choice whether to opt for a premium IOL or not. On the other hand, the pressure on ophthalmic surgeons to fulfill patient expectation continues to grow, and a premium IOL may be one way to achieve the visual goals of selected patients. In this article, I describe some pearls for premium IOL implantation to help ensure that we hit the targeted refractive outcome and that patients are happy postoperatively.
TARGET REFRACTION
The preoperative examination has to take place in a good atmosphere; the reception team and the doctor should be patient-oriented, so that the patient gains confidence in the clinic (Figure 1).

Figure 1. Clinics that focus on premium IOLs must offer a cultivated atmosphere.
The discussion with the patient regarding target refraction is important and independent of which IOL is to be implanted. A patient who has been able to read without glasses since his late 40s because of myopia will be not be happy if he suddenly needs reading glasses after surgery. In such a case, a target refraction of -2.50 D to enhance reading vision is a better option than a target of emmetropia. On the other hand, consider a 70-year-old woman who wears contact lenses for her myopia and is used to needing reading glasses but being free of glasses when driving. She will better accept a target refraction of plano, even though she was myopic preoperatively.
Another important consideration is this: What happens if the patient changes his or her mind after this consultation and calls the practice to change the target refraction? Standards must be put in place to ensure that the surgeon in the operating room has this information. Where must this requested change be recorded? If it is only in the patient’s chart, will the chart be present in the operating room? Creating a procedure to make sure the surgeon is aware of any such change is mandatory.
CHOOSING THE RIGHT LENS
Whatever IOL is chosen, be sure to have a good discussion with the patient before surgery about the particular advantages and drawbacks of the lens. Aspheric IOLs are appropriate for all patients, particularly for those who drive. Of the available aspheric IOLs, one should be chosen so that aberrations are slightly undercorrected.
If the patient has corneal astigmatism of greater than 1.00 D, a toric IOL will be advantageous. When discussing the advantages of this lens with the patient, be sure to emphasize that, in the event of an intraoperative complication such as posterior capsular rupture, a toric IOL cannot be implanted. Furthermore, the patient must be informed of the possibility that the toric IOL might rotate, and therefore a secondary surgical intervention may become necessary. Shrinkage of the capsular bag can also result in IOL misalignment. Toric IOLs should be implanted only in eyes with pupils that dilate sufficiently during surgery to allow control of the alignment; eyes with pseudoexfoliation often do not do so.
BIOMETRY
Biometry has become one of the most important parts of the preoperative exam. We use the IOLMaster (Carl Zeiss Meditec) for optical biometry and calculate powers for different IOL models. Corneal surface hydration is important when taking the reading; we use artificial tears to ensure good corneal wetting.
We also do corneal tomography Scheimpflug imaging with the Pentacam (Oculus Optikgeräte) to measure corneal astigmatism and higher-order aberrations (HOAs). A patient with total HOAs of about -0.20 µm will receive an IOL with moderate aspheric correction, such as the AcrySof IQ (Alcon). A patient with HOAs of more than 0.28 µm will receive an IOL with a higher aspheric correction, such as the Tecnis PCB00 (Abbott Medical Optics; Figure 2).
For patients who desire multifocal IOLs, we also examine the total HOAs, which should be less than 0.3 µm. If HOAs exceed this level, the patient will experience more side effects with a multifocal lens than he or she would with a monofocal (Figure 3).
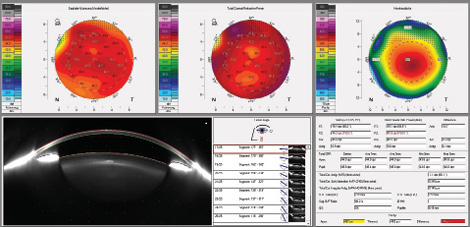
Figure 2. This patient, who has 0.345 μm of HOAs, will benefit from an aspheric IOL with a higher degree of aspheric correction, such as the Tecnis PCB00.
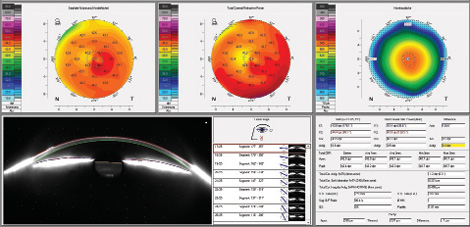
Figure 3. This patient with a multifocal IOL has HOAs greater than 0.4 μm. Despite postoperative near and distance UCVAs of 20/20, the patient had visual complaints.
We always compare the keratometry (K) readings of corneal topography with those of the IOLMaster. If there is a difference, both measurements are repeated. Additionally, we use different IOL power calculation formulas depending on the characteristics of the eye. For eyes with normal axial lengths, we use the SRK-T formula; for axial lengths less than 22.5 mm, we use the Hoffer Q formula, and for axial lengths greater than 26 mm we use Holladay or Haigis. Furthermore, we have the Okulix formula (Tomey) integrated into the Pentacam, which calculates IOL power with ray tracing. This may also be useful in eyes that have previously undergone corneal refractive surgery.
If different methods yield different IOL powers, I usually decide to use the mean value in the patient’s first eye and wait to see how the postoperative refraction comes out. The second-eye calculation can then be more precise, as I assume I have made a systematic mistake in the measurement of right and left eyes.
It is also wise to do spectral-domain OCT imaging before surgery. It is not unusual to detect vitreomacular traction syndrome, which can be associated with problems postoperatively such as macular edema. If the patient is informed of this condition preoperatively, he or she will be prepared and cooperative postoperatively (Figure 4).
STANDARD PEARLS FOR EXCELLENCE
Do not overpromise. Ultimately, we cannot guarantee the patient that he or she will achieve the desired postoperative target refraction, even if all measurements have been performed precisely. The final fit of the IOL in the capsular bag and the postoperative shrinkage of the bag will determine any myopic or hyperopic shift the patient might experience, even after having a plano refraction in the early weeks. Patients are disappointed when this happens. Therefore, we should never promise spectacle independence, but rather our intention to achieve spectacle independence. If biometry is difficult in a certain patient, we should tell the patient this and write it in the chart: “Difficult biometry; target refraction will be difficult to hit.”
At a Glance
• The pressure on ophthalmic surgeons to fulfill patient expectations continues to grow, and a premium IOL may be one way to achieve the visual goals of selected patients.
• When discussing the advantages of toric IOLs with patients, emphasize that, in the event of an intraoperative complication such as posterior capsular rupture, this type of lens cannot be implanted.
Create a quiet atmosphere on surgery day. It is important to establish a good environment in the surgery center on surgery days so that patients feel good and do not complain. Even if it sounds impolite, checking each patient’s data by asking for his or her birthdate and name is vital to be sure that you have the right patient in front of you. “Are you Mrs. Smith?” is the wrong way to phrase the question. Most cataract patients are old and often somewhat deaf, and many will automatically answer, “Yes.”
Not long ago, we had three different patients with the same name scheduled in the same week. Our chief nurse, who is always systematic in her work, revealed during preparation before surgery on Monday morning that we had the wrong patient’s chart. We were able to obtain the correct chart and continue with surgery.
Implement standards for all written communication between colleagues. If the patient decides on the day of surgery to have surgery done first in the eye originally planned as the second eye, the risk for implanting the wrong IOL power is high. The surgeon must be informed if the patient wants surgery on the other eye first, and it has to be marked everywhere so that the lens power will be checked. Standards must be implemented in the clinic regarding what is meant by everywhere—in the computer, on the handwritten patient chart, on a board for all to see in the operating room?
Practice purposeful postoperative follow-up and fielding of common complaints. When patients come in for follow-up, fortunately, halos, glare, and scotomas are relatively rare, occurring in less than 10% of eyes. If the ophthalmologist explains to the patient the reason for these positive or negative dysphotopsias, the patient will feel better. However, having paid out of pocket for a premium IOL, the patient wants to see well without any restrictions. Artificial tears can often help; many postoperative visual complaints are actually side effects of dry eye disease (DED). If the patient complains only about nighttime vision, application of brimonidine 30 minutes before driving may be helpful. For the most part, dysphotopsias go away during the first year.
Postoperative anisometropia after cataract surgery in the first eye can cause nausea. Most patients should have second-eye surgery scheduled before surgery is performed in the first eye, and the patient should be informed about the possible effects of anisometropia at that time. Use of a contact lens to treat ansiometropia is not a good choice in the typical cataract patient who is elderly and not used to contact lens care.
Postoperative DED will often be noted by the patient as, “something itching in my eye,” or “I can feel the sutures,” (when we know there were no sutures). It is ideal to prescribe artificial tears during the preoperative exam so that the patient can learn how to administer the drops. This also allows the patient to pretreat for DED, potentially avoiding the phenomenon postoperatively. It also prepares the patient for handling postoperative therapeutic drops such as topical steroids or NSAIDs.
Master timing of the toric lens follow-up visit. Residual astigmatism after toric IOL implantation often causes unhappiness. The first postoperative day visit does not tell you whether you hit the axis, as a toric lens can rotate even on day 2. Therefore, I consider the 1-week follow-up visit crucial for patients with toric IOLs.
If refraction reveals astigmatism greater than 0.75 D, check the IOL axis through a dilated pupil. If rotation of more than 10° is observed, the patient should undergo secondary surgery to have the toric IOL rotated back to the right axis. In my opinion, this second surgery should not be done until at least 14 days after the first; by that time, the capsular bag has begun to close, and further rotation will be less likely. If there is no rotation still but a remarkable amount of residual astigmatism, the IOL calculation should be rechecked. Topography should also be looked at again, to be sure that you have not ignored, for example, significant coma. Whether a lens exchange in this case is the proper solution should be discussed by surgeon and patient.
One possible mistake when there is over- or undercorrection of astigmatism is that the toric IOL power for the contralateral eye was implanted by mistake. Therefore, I always check K-readings and lens power on the toric calculator and compare it with topography and IOLMaster readings at the start of surgery.
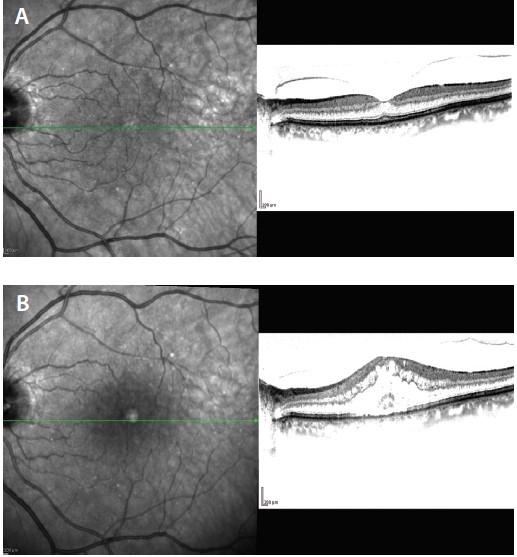
Figure 4. Before cataract surgery, this patient was informed of a heightened risk of macular edema due to vitreomacular traction (VMT) syndrome (A). Four weeks after surgery, macular edema related to VMT is seen (B).
SPECIAL CONSIDERATIONS FOR MULTIFOCALs
There are a number of particularly important considerations in postoperative care for patients with multifocal IOLs. First, these patients have to be treated specially because of their choice of a premium lens and managed by experienced staff. Poor treatment by team members may affect the patient’s expectations of the lens and make him or her unhappy.
Many multifocal lens patients may feel uncomfortable in the early postoperative period; they may question whether they made the right decision to choose this high-tech lens. To allay these fears, emphasize the patient’s good visual acuity result, and, if possible, do so in the presence of his or her partner or spouse. This helps the patient be comfortable that the right choice was made. The partner will also appreciate knowing about this good visual result.
Some aspects improve with time after multifocal implantation. The patient must be informed that reading speed may be slow at first and will become faster as time goes on. If there is residual ametropia, the patient may experience halos. Therefore, it is important to discuss the possible need for touch-up LASIK before first-eye surgery. Make sure the patient assents to this and signs the written informed consent before surgery, as he or she may not otherwise remember the discussion postoperatively.
Many problems in multifocal lens patients stem from DED and can be resolved by application of artificial tears every hour. Patients must be motivated to follow this routine, and the process can be facilitated by a highly motivated staff member.
Multifocal lens patients will be happy when they are treated in this special manner. When results are successful, this is the group of patients that will talk to others about their lens as no other lens group does. “I had cataract surgery, and I do not need any glasses anymore.”
CONCLUSION
Cataract surgery has gained a high level of sophistication, and so have cataract patients. Patients today are well informed about their choices and their surgery, and in many cases they only wish to have the best and the most modern solutions.
We should not withhold premium lenses. They can improve the quality of our patients’ vision and fulfill their desires to achieve postoperative visual goals; however, they can also present a challenge to the surgeon above and beyond one’s routine work.
In order to achieve the best results, the surgeon needs a well-trained staff that can communicate effectively with patients, precise measurements of patients’ eyes, and strong surgical skills. n
Stefanie Schmickler, MD
• Augen-Zentrum-Nordwest, Ahaus, Germany
• st.schmickler@augen-zentrum-nordwest.de
• Financial disclosure: None

