
Lens surgery with the aim to restore vision without spectacles is a multifactorial piece of art. As the surgery becomes ever more standardized—whether done by hand or by laser—the keys to success lie in the preoperative evaluation and the tools used to control the refractive result.
Premium IOLs are just like conventional IOLs, except that everything should be premium. This means that each step, from preoperative evaluation to surgery, should be carefully controlled in order to achieve the refractive goal. An error of 0.50 D in sphere or 0.75 D in cylinder may very well make the difference between success and failure with a multifocal implant; therefore, we should make every effort to be on target. The good news is that today’s diagnostic and surgical techniques together make it possible to achieve exactly that goal.
BIOMETRY
Thanks to laser biometry, introduced about 15 years ago, we can measure the length of the eye with unprecedented accuracy. The precision of optical biometers is typically within 20 µm between repeated measurements. Recalling that a 1-mm error in axial length is the equivalent of a 2.50 D error at the spectacle plane, you will find that the biometry error accounts for only 0.05 D error in the refraction.
Now I have a question for you: What is the average prediction error in your clinic? More than 0.05 D, I presume. Therefore, there are many other sources of errors in this process, including errors in IOL constants, keratometry (K) readings, and the prediction of the estimated lens position (ELP), which is a job performed by the IOL power calculation formula. For that last job, I prefer to use an optical biometer that measures anterior chamber depth and lens thickness, in addition to axial length, because this extra information can be used to improve the ELP prediction using the C-constant approach (described later in this article).
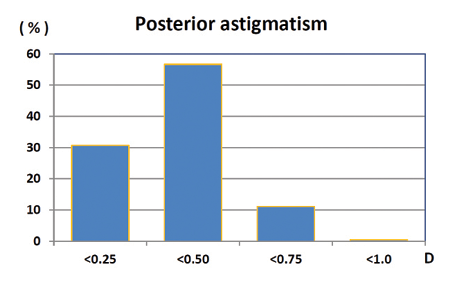
Figure 1. The posterior cylinder of the cornea as determined by Scheimpflug tomography in 880 normal eyes. The median was 0.30 D.
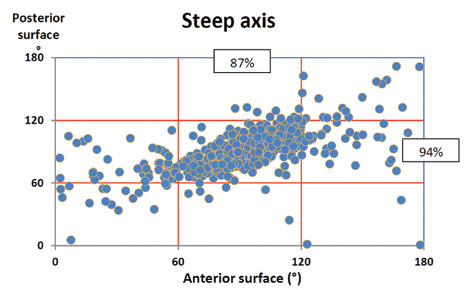
Figure 2. The steep axis of anterior and posterior corneal cylinder as determined by Scheimpflug tomography in 880 normal eyes. There was high correlation between withthe- rule axis of the front surface (steep axis of anterior surface around 90°) and the same axis of the posterior surface, resulting in a net cylinder lower than the cylinder measured on the front surface. Less consistent covariation was found for other axis orientations.
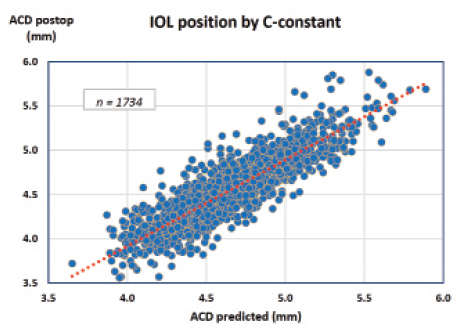
Figure 3. Observed versus predicted IOL position in 1,734 eyes using the C-constant concept of the Olsen formula (correlation coefficient r = 0.89, P<.00001). In 98% of eyes, results were within ±0.5 mm of predicted value.
KERATOMETRY
A good keratometer should provide accurate and reproducible readings of the sphere and cylinder equivalent of the corneal power. Many autokeratometers do a good job on reproducibility (ie, precision), but one must pay attention to the fact that they measure only a paracentral ring area of the cornea (about 3 mm) due to their use of Placido-disc–based measurement. Thus, in the case of an irregular cornea with a topographic variation from the center to the periphery, the more important central part may be missed.
Aside from being blind to the very center of the cornea, another limitation of keratometers (and topographers) is that they do not measure the true power of the cornea. Their measurements require an assumption about the refractive index of the cornea and the refraction on the posterior surface as well. In recent years, many papers have focused on the fact—which is actually old knowledge about the internal astigmatism of the eye—that the true total cylinder of the cornea is modified by the posterior cylinder of the cornea. Therefore, this may give rise to an error in the selection of toric IOL cylinder if not corrected for. Helpful nomograms, such as the Baylor nomogram proposed by Koch and coworkers,1 have been developed; however, it should be realized that exceptions to the rule occur, especially if the axis is in the oblique direction.
Figures 1 and 2 show our results from a population-based study using the Pentacam (Oculus Optikgeräte) to measure both the anterior and the posterior cylinder of the cornea in 880 normal eyes. As can be seen, the posterior cylinder contributed less than 1.00 D to the total cylinder of the cornea. Small as this seems, it may, however, be important when the decision is made whether to use a toric implant. The effect on the total net cylinder of the cornea depends on the axis and must be calculated using vector analysis combining the two cylinders. If the axis of the back axis coincides with the front axis, there will be a reduction of the front astigmatism, and vice versa.
Figure 2 shows the distribution of the steep axis of the anterior and posterior surfaces of the cornea in the same 880 eyes. There is a high correlation between with-the-rule axis of the front surface (steep axis of anterior surface around 90º) and the same axis of the posterior surface, giving a net cylinder lower than that measured on the front surface. Less consistent covariation is found for other axis orientations, and, therefore, it is more difficult to provide average corrections or nomograms in these cases.
At a Glance
• Sources of error with premium IOLs include errors in IOL constants, K-readings, and the prediction of the estimated lens position.
• Although keratometers and topographers provide precise measurements, they do not measure the center of the cornea or the true corneal power.
• Whatever IOL power calculation formula is preferred, it is important that the user know about its principles and, in particular, how the IOL constants should be optimized.
• In addition to all of the above, one should also remember that a good diagnostic evaluation begins with and depends upon a careful patient history.
IOL POWER CALCULATION
The field of IOL power calculation has evolved considerably over the years. Many of the old IOL power calculation formulas developed at the time of ultrasound biometry and less sophisticated diagnostic equipment are not able to give an accurate result within ±0.50 D in all cases, which is the tolerance we aim for with premium implants. Whatever formula is preferred, it is important that the user know about its principles and, in particular, how the IOL constants should be optimized. It is not recommended to rely entirely on the constants supplied by the IOL manufacturer, as these may be based on old studies or assumptive values. The best way is to keep track of your own results and monitor the actual IOL constants in your hands.
As the author of the Olsen formula, I am biased in the recommendation of which formula to use. The Olsen formula is available as an option on some biometers but also as PC software for complete IOL power calculation and data management, downloadable from the Internet (www.phacooptics.com). In my hands, this system gives me the accuracy I want with premium IOLs, in terms of both sphere and cylinder, in any type of eye.
One reason for the high accuracy of this formula is its improved prediction of IOL position (ie, ELP) with use of the C-constant concept.2 The C-constant uses the anatomy of the crystalline lens to predict the postoperative position of the IOL as a constant fraction of the axial thickness of the empty capsular bag. This simple concept has proven to be accurate and has the advantage of being independent of the K-reading and the axial length, which may be subject to variation, for example as a result of refractive surgery.
Figure 3 includes the results of 1,734 consecutive routine cases in which the final IOL position had been measured with the Lenstar LS 900 laser biometer (Haag-Streit) and the observed anterior chamber depth compared with the value predicted using the C-constant. Different types of IOLs were included, mostly from the AcrySof (Alcon) and Tecnis (Abbott Medical Optics) platforms. As can be seen, 98% of eyes were found to be within ±0.5 mm of the predicted value, and no bias was found with the K-reading nor the axial length, meaning that an unbiased, accurate IOL prediction was provided in all kinds of eyes.
FINALLY: THE IMPORTANCE OF HISTORY
In addition to all of the above, please also remember that a good diagnostic evaluation begins with and depends upon a careful patient history. The patient may forget to tell you about his or her laser surgery many years ago, leaving it to you to make the diagnosis by careful screening for anterior segment pathology. Watch out for patients who have previously undergone LASIK or keratoplasty and those who have keratoconus, megalocornea, astigmatism of any kind, a history of contact lens wear, unusual pupils, microphthalmos, short or long eyes, macular pathology, and so on. Any of these conditions may influence the choice of premium IOL and the IOL power calculation. n
1. Koch DD, Jenkins RB, Weikert MP, Yeu E, Wang L. Correcting astigmatism with toric intraocular lenses: effect of posterior corneal astigmatism. J Cataract Refract Surg. 2013;39(12):1803-1809.
2. Olsen T, Hoffmann P. C constant: new concept for ray tracing-assisted intraocular lens power calculation. J Cataract Refract Surg. 2014;40(5):764-773.
Thomas Olsen, MD, PhD
• Professor of Ophthalmology, University Eye Clinic, Aarhus Hospital, Denmark
• tkolsen@dadlnet.dk
• Financial disclosure: Paid consultant (Haag-Streit AG); Employee (CEO; IOL Innovations, manufacturer of the PhacoOptics PC software)

