Over the years, the evolution of ablation profiles has helped not only to improve visual outcomes after laser vision correction (LVC) but also to provide patients with safer and more customizable surgical treatments. Below, a group of surgeons helps to navigate the numerous advanced ablation patterns and treatments that are available from several manufacturers.
Topography-Guided LASIK With Contoura Vision
Outstanding results seen in corneas with normal topography.

By R. Doyle Stulting, MD, PhD
Over the past 20 years, refractive surgery lasers have evolved from devices that could correct only myopic spherical errors, to devices that could also correct hyperopia and astigmatism, to lasers with eye trackers, then to lasers that could deliver custom ablations based on wavefront measurements. Outcomes have also been improved by the recognition that increased energy is needed to obtain appropriate curvature change in the periphery.
One might ask why patients might benefit from a customized refractive procedure that is based on corneal curvature rather than wavefront measurement, which many consider standard of care. Theoretically, topographically customized treatment might be superior to wavefront-guided treatment for several reasons. For starters, topography allows measurement of many more points of the visual system than wavefront sensors measure. Also, wavefront measurements are restricted by the pupil size at the time of measurement, and corrections based on wavefront measurements are truly accurate for only one point in space.
Topographic measurements, by contrast, are more reproducible, and they can accurately capture abnormalities in the peripheral cornea that affect quality of vision in dim light. These factors are important for activities such as night driving. Topography-guided treatment is not influenced by lenticular abnormalities or vitreous opacities, as wavefront-customized treatment is. Finally, highly aberrated corneas are more accurately measured by topography than by wavefront sensors.
Corneal topography does not, however, give us complete information about the optical system of the eye. Contoura Vision (Alcon) is a topography-guided LASIK treatment that combines diagnostic information on lower-order aberrations from traditional manifest refraction with higher-order aberrations from the cornea to create an ablation pattern that is then applied to the eye in the usual fashion. Contoura Vision is performed with the WaveLight Allegretto Wave Eye-Q or WaveLight EX500 excimer laser system (both by Alcon) in conjunction with the WaveLight Topolyzer Vario diagnostic device (Alcon).
The key to successful use of Contoura Vision is obtaining accurate and reproducible corneal topography. This is an exacting and demanding process that requires the knowledge and attention of the refractive surgeon or someone on his or her staff who is meticulous and detail-oriented.
Topography-guided LASIK and PRK are generally considered internationally as a repair procedures, used in the treatment of patients with visual aberrations caused by previous corneal refractive surgery or naturally occurring irregular astigmatism. Alcon presented the US FDA with a proposal to perform a clinical trial on normal eyes, and, after analyzing early results from initial treatments, to treat eyes with abnormal topographies. The FDA did not accept this proposal, insisting that Alcon would have to complete a clinical trial in normal eyes before initiating a trial in eyes with abnormal topographies. Furthermore, because LASIK is contraindicated in many eyes with abnormal topographies, the FDA demanded that a clinical trial of PRK in normal eyes be completed before a trial could be initiated using topography-guided PRK for abnormal corneas. As a result of these requirements, clinical trials of topography-guided treatment of abnormal corneas have not been done.

Off-label treatment of highly aberrated eyes will certainly occur in the United States. It is unfortunate that good information from a well-conducted clinical trial will not be available to guide those treatments because of the FDA’s position.
Many of us involved in the FDA clinical trial of Contoura Vision did not believe we would have outcomes better than those seen with conventional or wavefront-optimized treatments. Early in the trial, however, when topography-guided LASIK could be performed on only one eye per patient, investigators found that patients preferred the quality of vision that they achieved in the eye with topography-guided treatment, compared with the contralateral eye that had undergone conventional or wavefront-optimized treatment.
The final results of the clinical trial were beyond the expectations of all involved. At final follow-up, 16% of eyes had 20/10 UCVA; 34% had 20/12.5 or better UCVA; 65% had 20/16 or better UCVA, and 93% had 20/20 or better UCVA. Additionally, 89% of eyes had a UCVA after surgery that was equal to or better than the BCVA before surgery, and 31% of eyes had better UCVA postoperatively than preoperatively.
At 12 months, topography-guided LASIK-treated eyes had statistically significant subjective improvement in light sensitivity and reductions in difficulty driving at night and reading, and in the incidence of glare, halos, and starbursts.
These were unexpectedly good outcomes for a population of patients with eyes that had normal topographies preoperatively and were treated with a technology that is known primarily for its ability to correct eyes with irregular astigmatism. The outcomes of this trial usher in a new era in which we can, based on data obtained in this clinical trial, offer patients a treatment that is likely to give them UCVA that is equal to or better than the best vision they currently experience with their glasses. We can also tell them they will probably have less severe visual aberrations, such as those that interfere with night driving, after they have topography-guided LASIK with Contoura Vision.
Many believe that LASIK, over a lifetime, is less expensive and more likely to provide exceptional vision than contact lenses. Additionally, it is probably less risky than contact lenses. Those of us who have been performing LASIK for the past 20 years remember the time when we had to discuss the likelihood that patients would have to endure an increase in visual aberrations and perhaps a decrease in visual acuity in exchange for the ability to see without glasses or contact lenses. Today, we can tell patients that we offer a method of correcting their vision that is probably better than glasses, probably safer than contact lenses, and probably less expensive than either over their lifetime.
Perhaps the era has arrived when surgery to correct vision without the aid of glasses will become accepted by patients and insurance companies, just as surgery to correct an abnormality of the foot or leg would be accepted to allow patients to walk without the need for crutches.
Supracor: Addressing Presbyopia on the Cornea
This treatment may be the optimal choice for patients not ready for refractive lens exchange.


By Federico Alonso Aliste, MD;
and Jonatan Amián Cordero, MD
Supracor is a technique for the correction of refractive error associated with presbyopia using excimer laser corneal ablation in a single procedure. The correction algorithm, developed by Technolas Perfect Vision, is available for the Technolas 217P and Teneo 317 excimer lasers (both by Bausch + Lomb).
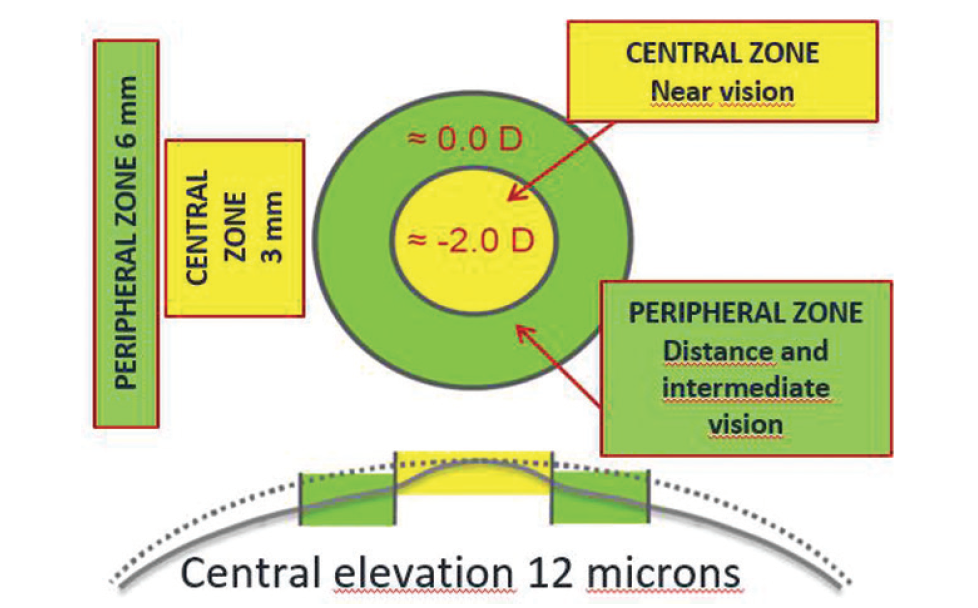
Figure 1. The corneal ablation pattern for Supracor.
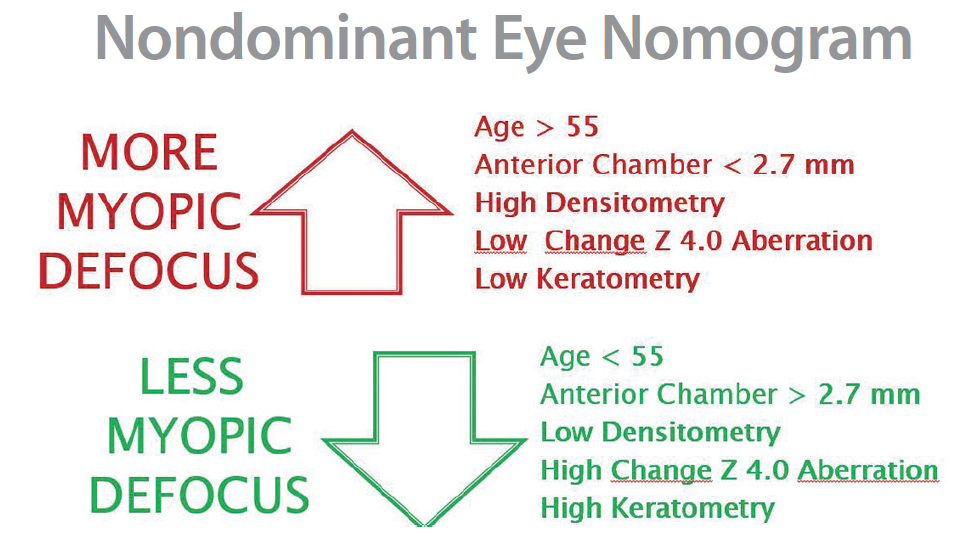
Figure 2. Nomogram for nondominant eye.
The basis of this surgical procedure is the creation of a varifocal cornea with two optical zones: a hyperprolate central 3-mm zone with 12 µm elevation, responsible for near vision, and a peripheral zone with an aspheric profile that extends up to 6 mm, responsible for distance vision. Between them there is a smooth transition zone for intermediate vision (Figure 1).
The mechanism of action of Supracor is pupil-dependent. For near vision, the pupil contracts, and the near image is focused on the retina due to the negative spherical aberration provided by the central optical zone. For distance vision, the pupil is slightly dilated, allowing 300% more light through the peripheral zone, which ensures good quality of distance vision.
Supracor can be performed on either myopic or hyperopic patients. Depending on the desired refractive addition to be provided by the central zone, two options of treatment are available: Supracor Mild (1.00 D add) and Supracor Regular (2.00 D add). In order to achieve better visual outcomes for near vision, the treatment is often used with a slight grade of defocus in the nondominant eye (between 0.00 and -0.50 D), with this micro-monovision providing a near addition of up to 2.50 D.
CLINICAL EXPERIENCE
After several years of experience using different surgical techniques for the correction of presbyopia, we have concluded that a key factor in successful presbyopia correction is not only obtaining a good visual outcome for near vision, but also achieving good distance vision for the patient.
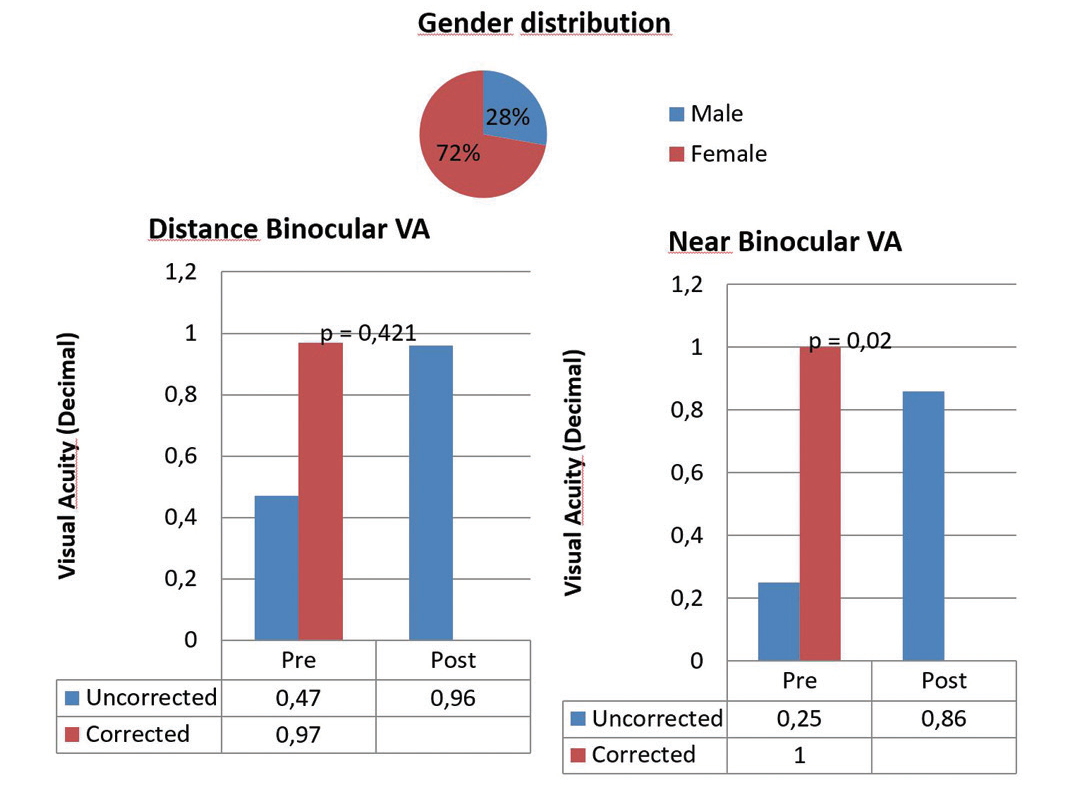
Figure 3. Near and distance binocular visual acuity before (pre) and after (post) Supracor.
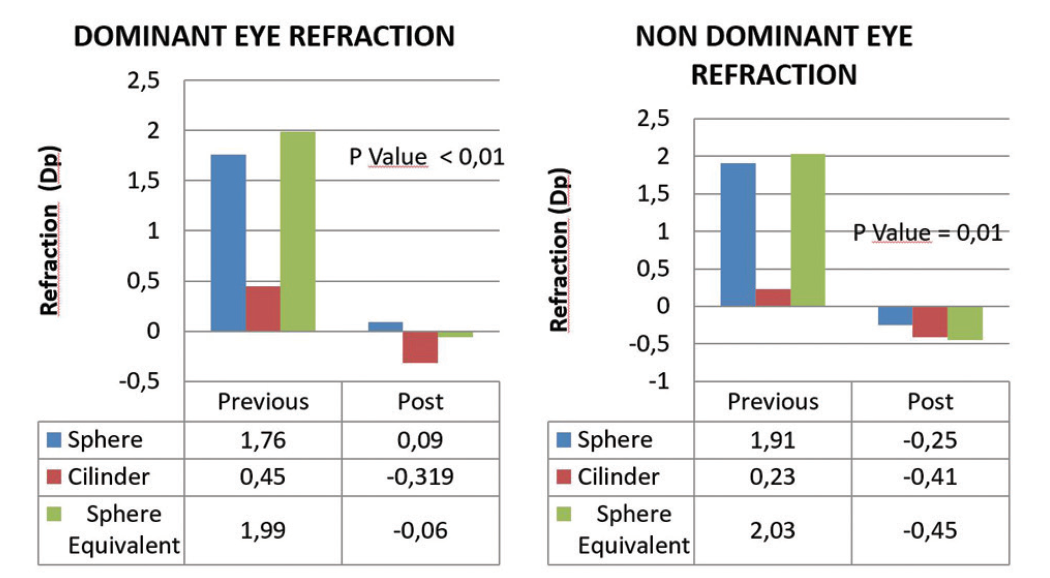
Figure 4. Refraction in dominant and nondominant eyes before (previous) and after (post) Supracor.
The two techniques that we use most frequently are (1) bilateral asymmetric Supracor Mild and (2) Proscan for distance vision in the dominant eye and Supracor Mild for near vision in the nondominant eye. Proscan is an algorithm on the Teneo 317 laser that corrects sphere and cylinder and reduces undesired surgically induced spherical aberration. With both approaches, outcomes have been good for near and distance vision.
As with any surgical procedure, a good preoperative eye examination is required. Among the essential elements are eye dominance testing, stereopsis testing, dry eye evaluation, topographic and tomographic corneal analysis, and aberrometry. In many cases, we check the patient’s tolerance for multifocality with the use of multifocal contact lenses for a few days. The final defocus target, chosen by the surgeon, will be higher or lower depending on the patient’s age, anterior chamber depth, lens density, amount of coma, amount of spherical aberration, and final keratometry (Figure 2). Generally, the dominant eye is targeted for distance vision with a slightly positive defocus (range, 0.25–0.50 D), whereas the nondominant eye is adjusted for near vision with a plano target or with a slightly negative defocus (range, 0.00 to -0.50 D).
Postoperatively, visual exercises such as use of Hart charts or a Brock string are recommended in order to obtain better outcomes. With these exercises, neural adaptation improves significantly and, as a result, so does patient satisfaction.
CONCLUSION
In our experience with this customized nomogram, clinical outcomes have been highly satisfactory. Both near and distance visual acuities have been excellent in our series. The average values obtained for spherical equivalent have been -0.06 D for distance vision in the dominant eye and -0.45 D for near vision in the nondominant eye (Figures 3 and 4).
We are aware that this is not a definitive treatment due to factors such as regression of refraction over time or the progression of the density and opacity of the crystalline lens; however, corneal treatments for presbyopia correction, and specifically the Supracor technique, are the preferred option of most patients because they are not as invasive as refractive lens exchange with multifocal IOLs.
Advanced CustomVue: Standard Deviation of LASIK Approaches Limits of Measurement
The technology has helped to make LASIK possibly the best optical alternative for vision-corrected patients.

By Julian D. Stevens, MRCP, FRCS, FRCOphth
The LASIK market in Europe and across most of the world has slowed dramatically from its heyday. In part that is because, 25 years after the introduction of LVC, a relatively large percentage of early adopters have already had the procedure. But there is still a big pool of candidates, including the millennial generation, which is just now reaching adulthood and refractive stability. Why is this generation not flocking to LVC?
Due to a natural cognitive bias known as probability neglect, people tend to overstate the risks of rare events, such as dying in a plane crash or having a serious LASIK complication. People then underestimate the risks involved in everyday activities such as driving a car or wearing contact lenses. A second cognitive glitch, called negativity bias, causes us to prioritize and assign greater credibility to bad news than to good news.
Probability neglect convinces people that LASIK is riskier than it is, and a single, anecdotal so-called “LASIK horror story” can unleash patients’ negativity biases. There are several antidotes to this situation.
ACCENTUATE THE POSITIVE
The first antidote is to increase the volume of good news so that it drowns out the bad news or puts the bad news in context. As surgeons, we know that LVC is extraordinarily safe. In studies published through 2008, more than 95% of people treated were satisfied with their results.1 The total number of people who have undergone LASIK is steadily growing and will eventually reach a critical mass at which it will be perceived to be commonplace.
Until then, we need to encourage our happy patients to make more noise. We can ask them to leave testimonials on the social media platforms that millennials rely on to guide their decision-making—platforms that were not even around when the early adopters had LASIK. There is no question that, if everyone who had LVC rated their experience publicly, the positive experiences would completely dominate online searches.
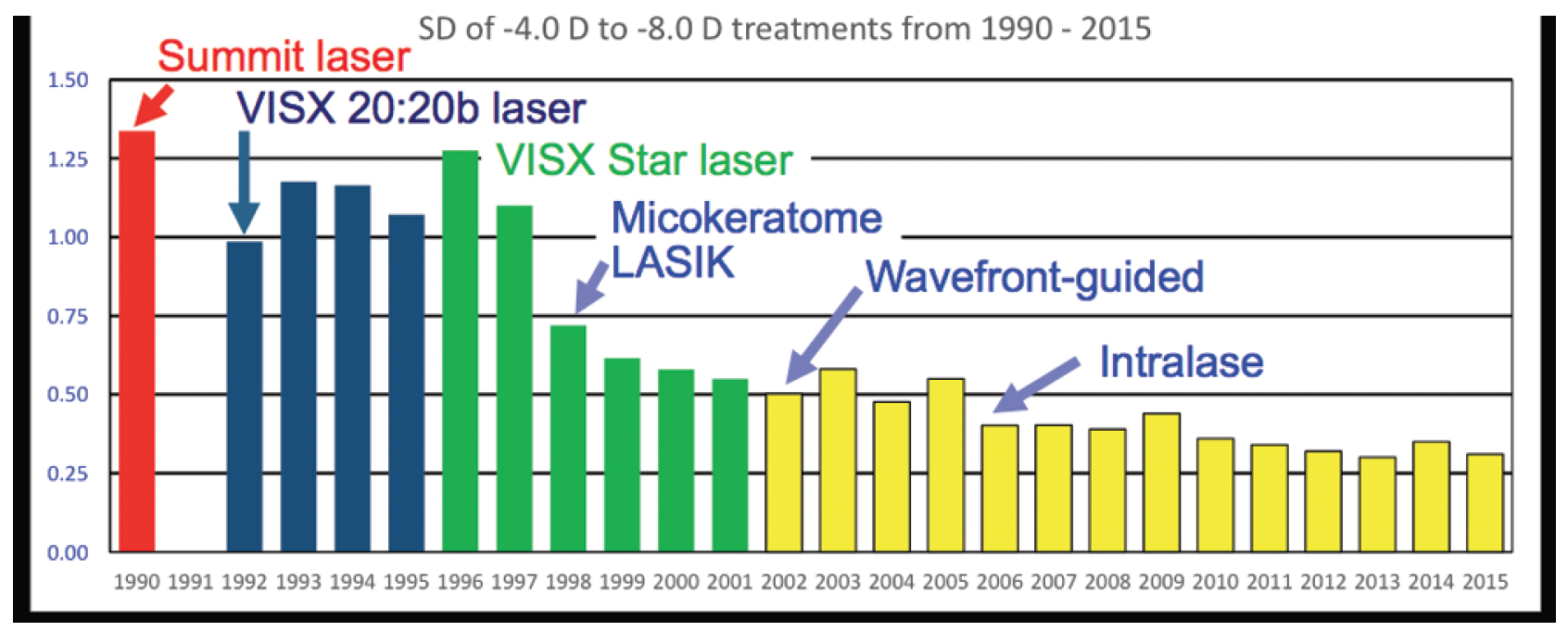
Figure 5. Over 25 years of technological advances, the standard deviation for LASIK has consistently gotten tighter, from 1.06 D in 1990 to 0.28 D in 2015.
We need to make it better known that the highest LVC penetration rates are found among eye surgeons, mission-critical military personnel such as combat aviators and special forces, and elite athletes who need to function at the limits of vision. That these groups have been willing to have LVC in higher numbers than the general population is a tribute to the technology and the surgical procedure itself.
IMPROVED TECHNOLOGY
Another antidote is that we should emphasize how the technology of LASIK has dramatically improved over the years. Today’s Advanced CustomVue procedure (Abbott Medical Optics) involves use of the iDesign aberrometer, which provides an increase in the fidelity of the wavefront data being measured compared with earlier aberrometers. With five times more data points measured and analyzed, it provides a very–high-resolution map, and, with its sophisticated data analysis, there is excellent reliability and constringency. Advanced Fourier mathematical reconstruction turns that data into a precision treatment that is delivered by the Star S4 IR excimer laser system (Abbott Medical Optics) under a flap reliably created by the IntraLase FS femtosecond laser (Abbott Medical Optics).
Thanks to this technology, the standard deviation for LASIK for correction of up to 4.00 D of myopia has tightened, from 1.06 D in 1990 to 0.28 D in 2015, a remarkable improvement in the precision of the procedure (Figure 5). We can now expect LVC to provide patients with visual acuity and optical quality that is often superior to other forms of vision correction such as glasses and contact lenses.
1. Solomon KD, Fernandez de Castro L, Sandoval HP, et al; Joint LASIK Study Task Force. LASIK world literature review: quality of life and patient satisfaction. Ophthalmology. 2009;116:691-701.
Triple A Nomogram
Incorporating this nomogram, the MEL 90 is faster and ablates less tissue than previous generations of the laser.



By Pedro Taña Rivero, MD, PhD; José Luis Rodríguez-Prats, MD, PhD, FEBO; and Yago Esperanza Quintana, OD
Refractive surgery is one of the mainstays of modern ophthalmology, allowing millions of patients to see better without optical correction. Since the middle of the past century, refractive surgery has evolved, with the goal of more accurate and safer correction of refractive errors.
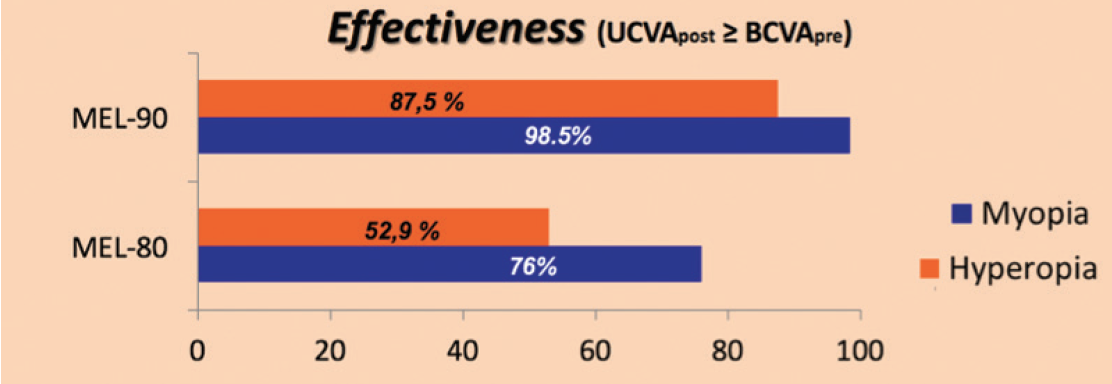
Figure 6. The MEL 90 platform had better effectiveness results overall, measured by the degree to which postoperative UCVA equaled or exceeded preoperative BCVA.
Currently, we use the MEL 90 (Carl Zeiss Meditec), one of the sixth generation of excimer lasers.1,2 Improvements with the MEL 90 include the ability to perform extensive treatment of optical zones; a reduction in treatment time, with more significant decrease in correction of high refractive error; and a new treatment profile called Triple A, a topography-guided nomogram adapted for myopic astigmatism. The MEL 90 also significantly reduces the thickness of ablation, with a greater reduction for corrections of high myopia.
COMPARISON STUDY
At our practice, Oftalvist, we performed a study comparing clinical outcomes in LASIK procedures with the fifth-generation MEL 80 excimer laser and the sixth-generation MEL 90. The results of the treatments were compared in terms of safety, efficiency, and accuracy. We found that the MEL 90 platform had better effectiveness results overall, as measured by the degree to which postoperative UCVA equaled or exceeded preoperative BCVA (Figure 6).
The accuracy of results was significantly better with the MEL 90 compared with the MEL 80 (Figure 7). Both platforms safely corrected myopic refractive errors (Figure 8). For correction of hyperopic errors, the MEL 90 platform was slightly safer than MEL 80. In terms of safety, efficiency, and accuracy, the MEL 90 was the better of the two platforms studied for correction of hyperopic refractive errors. At the same time, the MEL 90 significantly decreased the time of ablation in comparison with the MEL 80. It also significantly decreased the thickness of ablated tissue.
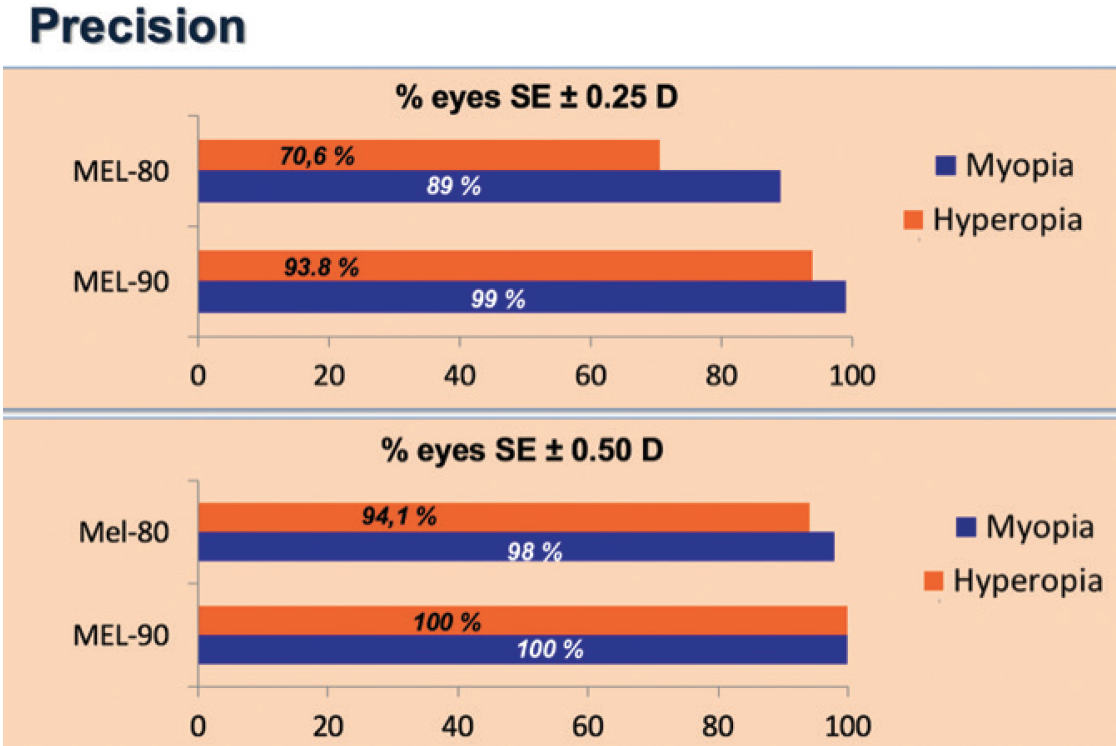
Figure 7. More eyes had spherical equivalent within ±0.25 D of target (top) and within ±0.50 D of target (bottom) with the MEL 90, in both hyperopic and myopic corrections.
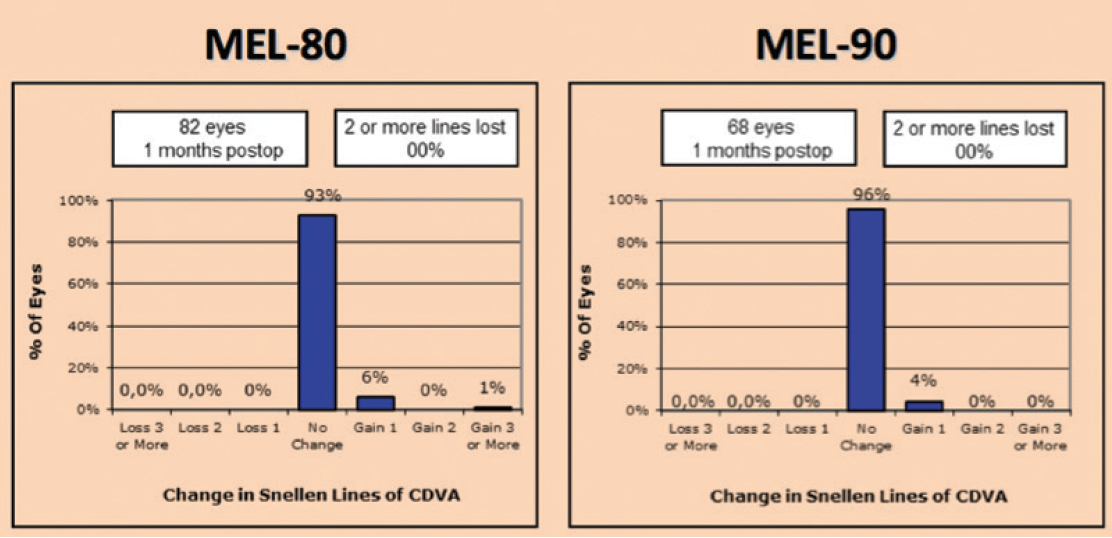
Figure 8. Both platforms safely corrected myopic refractive errors. No eyes lost 2 or more lines of CDVA.
CONCLUSION
Based on our clinical results, we believe that the MEL 90 is now the clear choice to replace the MEL 80 in our practice, as it provides the perfect platform for LASIK interventions in our hands.
1. Reinstein DZ, Carp GI, Lewis TA, Archer TJ, Gobbe M. Outcomes for myopic LASIK with the MEL 90 excimer laser. J Refract Surg. 2015;31(5):316-321.
2. Llovet F, Galal A, Benitez-del-Castillo JM, Ortega J, Martin C, Baviera J. One-year results of excimer laser in situ keratomileusis for hyperopia. J Cataract Refract Surg. 2009;35(7):1156-1165.
Amaris’ Aberration-Free Profile
The excimer laser offers a decision aid to help choose the best treatment.


By Samuel Arba Mosquera, MSc, PhD; and Arthur B. Cummings, MB ChB, FCS(SA), MMed(Ophth), FRCS(Edin)
The development of science and technology in LVC has been rewarding. The past few decades have seen improving standards for refractive surgery in the human eye and incremental growth in the effort to achieve perfect vision through surgical means. Achieving accurate clinical outcomes and reducing the likelihood of a retreatment procedure are major goals of refractive surgery. An ideal ablation profile should achieve these goals without the need for nomograms.
A Cartesian oval is the geometric shape that results from the condition of stigmatism; it represents a surface free of aberrations for the infinity point (distance point) in a human eye. If the anterior corneal surface were a Cartesian oval, it would have no aberrations; however, the anterior cornea is typically different from this shape and possesses its own individual aberration pattern. Therefore, a treatment whose application on a Cartesian-oval cornea would result in a new Cartesian-oval cornea with different dioptric power defines the concept of an aberration-free profile.1 For an aspheric anterior corneal surface, theoretically, an aberration-free ablation would balance the focus shift due to tissue removal, preserving the original location of the optical focus of the anterior corneal surface and avoiding the induction of postoperative hyperopic shift. Additionally, the aberration-free concept involves different aspheric compensation as a function of the size of the optical zone of treatment, further reducing the likelihood of inducing spherical aberration and higher-order astigmatism.1 This compensation is, however, implemented in discrete steps of 0.5 mm of optical zone size.
CENTRATION IS KEY
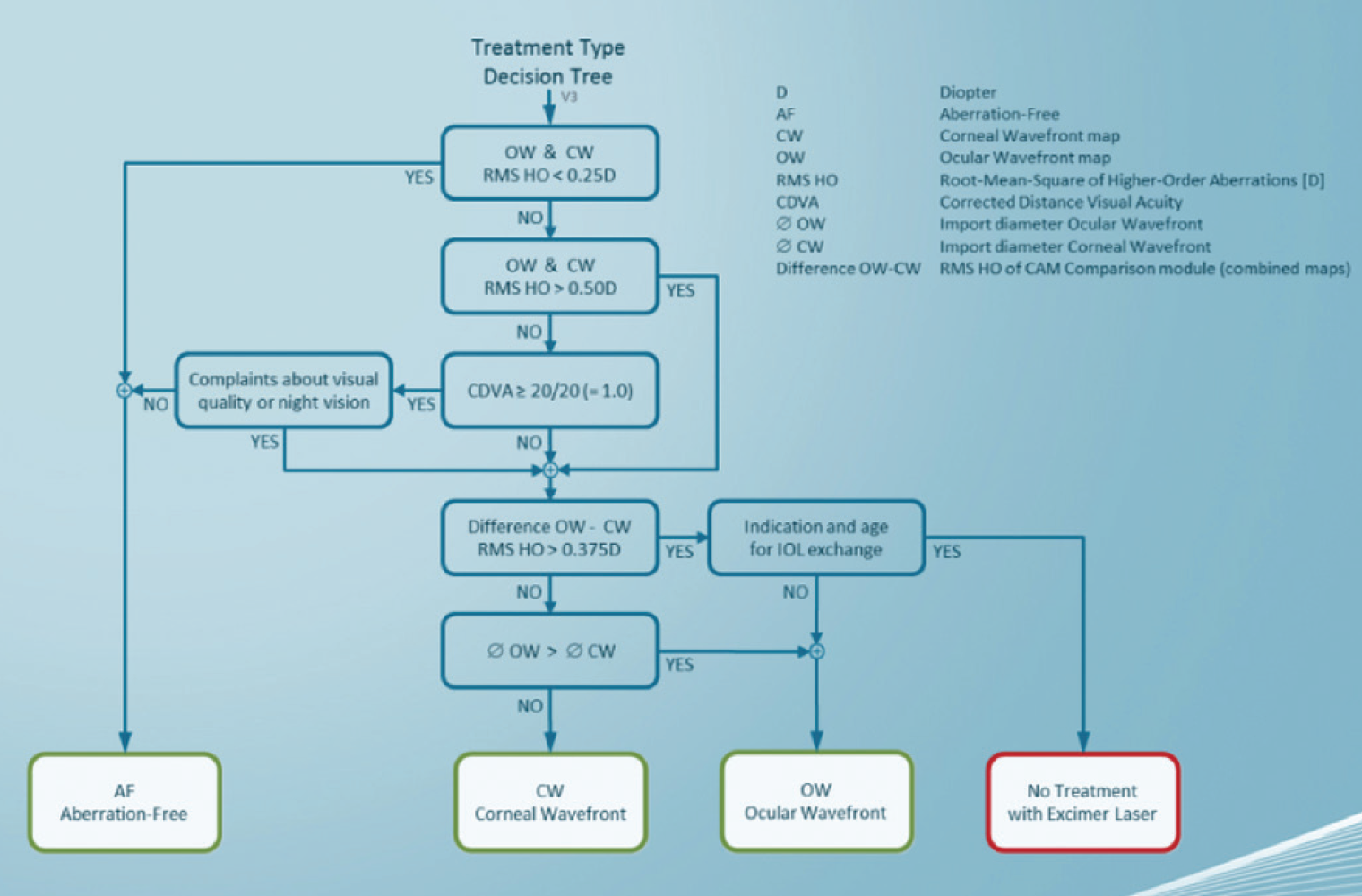
Figure 9. The Decision Assistant Wizard is a tool to select the treatment mode best suited to the needs and condition of each patient in a step-by-step fashion.
Centration of the ablation profile is crucial, and this is an often controversial topic in the realm of refractive surgery. The best method to determine the true visual axis is still an open question. The Schwind Amaris 1050RS, 750S, and 500E excimer laser systems (Schwind eye-tech-solutions) use a centration method for ablation profiles called asymmetric offset, which considers pupil center and corneal vertex information simultaneously.2 The ablation profiles cover the pupil aperture while respecting the corneal vertex as the optical axis of the ablation; in this offset profile, the eye’s higher-order aberrations are referred to the pupil center, while the manifest refraction values are referred to the corneal vertex. This centration methodology avoids induction of coma aberration from defocus correction (but also trefoil aberration from astigmatism correction), while saving tissue at the same time.
Dehydration of the cornea during excimer laser treatment can affect ablation efficiency and laser-tissue interaction. The cornea is a layered structure, with different cellular properties at different layers, rendering different refractive indices, hydration content, and ablation properties at each layer. Cutting a planar flap for stromal ablation in LASIK results in steepening the stroma by an amount equal to the flap thickness. This fine change in corneal curvature must be compensated within the ablation plan. Schwind Amaris lasers counterbalance these nuances through a multivariate dehydration model3 and cornea and flap thickness compensation algorithms. Furthermore, different treatment modalities associated with different ablation depths within the cornea (stromal ablation methods vs surface ablation methods) are implemented with use of different ablation spots within the aberration-free ablation profiles.
The Schwind Amaris family of excimer laser systems feature automatic dynamic transition zone calculation. On the one hand, this feature may limit the surgeon’s possibilities for exploring different transition zone sizes, but, on the other hand, it offers ease in treatment planning, always ensuring that identical treatments receive identical transition zones.
Patient age affects not only accommodative amplitude, but also the water content in the patient’s cornea.4 This change results in slightly different ablation rates for patients of different ages, and the aberration-free profiles compensate for this through a correlation-based algorithm that calculates a compensation factor for different age groups. It must be noted that patient age is not the only driving force for change in ablation rates among the patients; other confounding factors may also contribute. These factors are not individually analyzed in the Schwind Amaris system following a root-cause approach, but their combination is compensated through correlation-based compensation factors.
DECISION WIZARD
With myriad customization possibilities, each with its own benefits, limitations, and implications, it may seem complex to decide the optimum individualized treatment method for a given patient. The Schwind Amaris Decision Assistant Wizard acts as a tool to help select the treatment mode best suited to the needs and condition of each patient in a step-by-step fashion (Figure 9).
Whether treatments use corneal wavefront-driven, ocular wavefront-driven, or aberration-free profiles, it is imperative to achieve a baseline that does not induce any aberrations. In patients with low aberrations, the Decision Assistant Wizard favors aberration-free treatments because customized treatments offer no particular risks but also no foreseeable benefits in such cases. The larger the degree of aberrations, the higher the interest and inclination toward wavefront-guided treatments. The reasons for this are manifold: The larger the degree of aberrations, the higher the likelihood that these aberrations will affect vision, that they can be measured with sufficient accuracy, and that the system can reduce them efficiently. Furthermore, the larger the degree of aberrations, the higher the risk of refractive deviations, but also the higher the potential benefits in terms of distance BCVA and quality of vision.
Finally, among wavefront-guided treatments, corneal wavefront treatments are favored if corneal wavefront aberrations are similar to or smaller than ocular wavefront aberrations; otherwise, ocular wavefront-guided treatments are favored by the Decision Assistant Wizard.
The features described here highlight the state-of-the-art technological advances and useful tools offered by modern aberration-free profiles. Even with their associated physical limitations, these integrated features bring the aberration-free concept close to realizing its goal of delivering what it promises: a truly aberration-free treatment.
1. Arba Mosquera S, de Ortueta D. Analysis of optimized profiles for ‘aberration-free’ refractive surgery. Ophthalmic Physiol Opt. 2009;29(5):535-548.
2. Arba Mosquera S, Ewering T. New asymmetric centration strategy combining pupil and corneal vertex information for ablation procedures in refractive surgery: theoretical background. J Refract Surg. 2012;28(8):567-575.
3. de Ortueta D, von Rüden D, Magnago T, Arba Mosquera S. Influence of stromal refractive index and hydration on corneal laser refractive surgery. J Cataract Refract Surg. 2014;40(6):897-904.
4. Luger MH, Ewering T, Arba Mosquera S. Influence of patient age on high myopic correction in corneal laser refractive surgery. J Cataract Refract Surg. 2013;39(2):204-210.
Federico Alonso Aliste, MD
• Medical Director, Tecnolaser Clinic Vision, Sevilla, Spain
• Financial interest: None acknowledged
Jonatan Amián Cordero, MD
• Medical Subdirector, Tecnolaser Clinic Vision, Sevilla, Spain
• jamianc@gmail.com
• Financial interest: None acknowledged
Samuel Arba Mosquera, MSc, PhD
• Optical and visual researcher at Schwind eye-tech-solutions, Kleinostheim, Germany
• samuel.arba.mosquera@eye-tech.net
• Financial disclosure: Employed by company (Schwind eye-techsolutions)
Arthur B. Cummings, MB ChB, FCS(SA), MMed(Ophth), FRCS(Edin)
• Consultant Ophthalmologist, Wellington Eye Clinic and Beacon Hospital, Dublin, Ireland
• Associate Chief Medical Editor, CRST Europe
• abc@wellingtoneyeclinic.com
• Financial interest: None acknowledged
Yago Esperanza Quintana, OD
• Oftalvist, Spain
• Financial interest: None acknowledged
Julian D. Stevens, MRCP, FRCS, FRCOphth
• Consultant Ophthalmic Surgeon, Moorfields Eye Hospital, London
• Member, CRST Europe Editorial Board
• jds@uk.com
• Financial disclosure: Consultant (Abbott Medical Optics)
Pedro Taña Rivero, MD, PhD
• Co-Chairman, Ophthalmology Department, European University of Madrid, Spain
• Medical Director, Oftalvist, Spain
• pedrotana@oftalvist.es
• Financial interest: None acknowledged
José Luis Rodríguez-Prats, MD, PhD, FEBO
• Refractive Surgery Department, Oftalvist, Alicante, Spain
• jlrodriprats@gmail.com
• Financial interest: None acknowledged
R. Doyle Stulting, MD, PhD
• Director, Stulting Research Center, Woolfson Eye Institute, Georgia, North Carolina, and Tennessee
• Professor of Ophthalmology Emeritus, Emory University, Atlanta, Georgia
• dstulting@woolfsoneye.com
• Financial disclosure: Paid consultant (Alcon); Medical Monitor for FDA trial (Alcon; Wavelight Contoura Vision)

