
Cataract surgeons in the developing world must surmount countless obstacles beyond just getting trained—lack of equipment, lack of government funding for eye care, limited quantities of surgical supplies, and a shortage of competent ancillary help. However, the greatest challenge, which would undoubtedly overwhelm most of us in the developed world, is the huge backlog of advanced, mature cataracts in patients with multiple ocular comorbidities, including trauma, corneal scarring, glaucoma, zonulopathy, and diabetic retinopathy. From a personal standpoint, observing these hardships has given me a greater appreciation for my own work environment and tremendous admiration for our dedicated colleagues who toil in such resource-poor settings.
I have long felt that our single greatest challenge in ocular surgery is the rapidly increasing backlog of cataract blindness in the developing world, which accounts for half of all global blindness.1 This is hard for us to remember while preoccupied with that angry patient who still needs reading glasses despite his or her multifocal IOLs. Fortunately, some inspiring work is being done at places such as the Tilganga Eye Center in Kathmandu, Nepal, and the Aravind Eye Care System (AECS) in Southern India. I have had the great honor of learning from colleagues at both of these centers, where approximately 60% of all cataract surgeries are performed on charity patients. Both centers have perfected a standardized, assembly-line method of doing high-volume, low-cost cataract surgery at rates often exceeding 10 cases per hour per surgeon (Figures 1 and 2). To me, these are the most amazing eye surgical teams in the world, and it is a marvel to observe this surgical choreography in action.

Figure 1. One operating room at the Aravind hospital in Madurai. Two surgeons alternate between two adjacent operating beds by swinging the microscope from one table to the other.
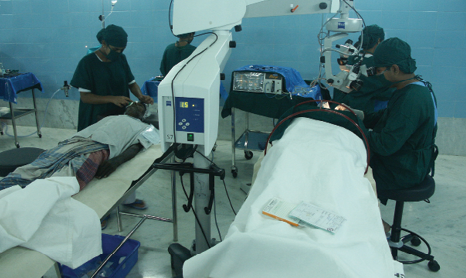
Figure 2. Rengaraj Venkatesh, MD, (Aravind Pondicherry) operating on one patient while his prior patient is patched. The next patient will be brought onto the adjacent bed and prepped so that he can immediately commence the next surgery without delay.
SIGNIFICANT LESSONS
I have also had the good fortune to collaborate with talented surgeons from Aravind and Tilganga on unique studies (Figure 3). Perhaps unexpectedly, these studies bear important and significant lessons for all cataract surgeons, which I highlight herein.
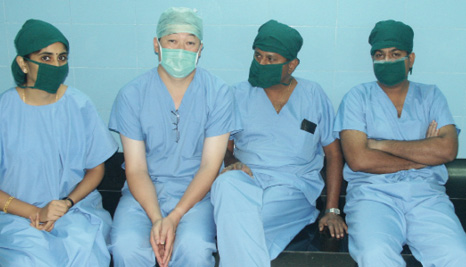
Figure 3. The author with three Aravind ophthalmologists who have coauthored numerous papers cited in this article (Pondicherry, 2003). From left to right: Aravind Haripriya, MS, co-chief of Aravind Chennai; the author; R.D. Ravindran, MS, DO, chief of Aravind Eye Care System; and Rengaraj Venkatesh, MD, chief of Aravind Pondicherry.

MSICS may be safer for mature cataracts. One of my early experiences doing phacoemulsification in a developing country was in 2005, when we conducted a randomized, prospective trial to compare phaco, as performed by me (Figure 4), with sutureless manual small-incision cataract surgery (MSICS), as performed by the leading Nepalese surgeon, Sanduk Ruit, MD (Figure 5).2 As pioneered by Dr. Ruit, surgeons at AECS, and others, MSICS uses a long, temporal, scleral-pocket incision that is tunneled wide enough to enable manual extraction of the undivided nucleus with an irrigating Simcoe cannula (Figure 6).3 After a PMMA IOL is implanted, the incision is self-sealing without sutures and is largely astigmatically neutral due to its sutureless construction and temporal location. Most important, these advantages are achieved with a low-tech method on exceptionally dense, mature cataracts. Aside from the microscope and the sterilizer, none of the manual instrumentation requires electricity.
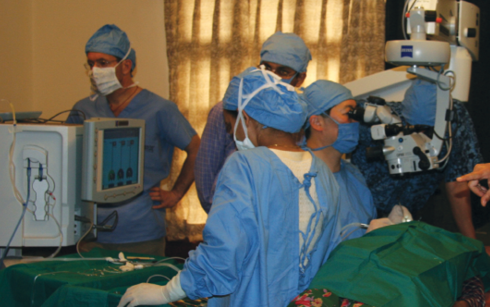
Figure 4. The author performing phacoemulsification within a makeshift operating room at the Pulihari Monastery in Nepal, 2005.
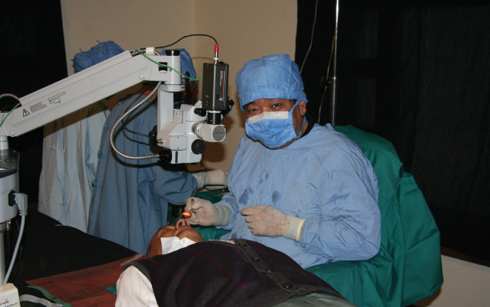
Figure 5. Dr. Ruit performing MSICS within a makeshift operating room at the Pulihari Monastery in Nepal, 2005.
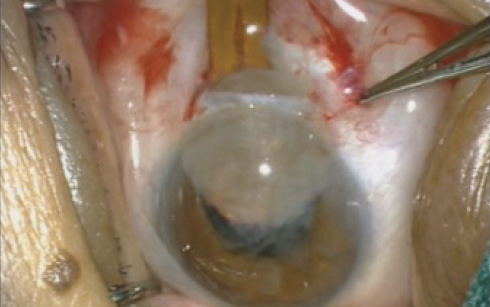
Figure 6. Dr. Ruit performing MSICS on a white cataract using an irrigating cannula.
The study included a population of charity patients near Kathmandu, and we used classrooms in a Buddhist monastery as our makeshift operating rooms (Figure 7). The patients were followed for 1 year, and we showed that UDVA and CDVA outcomes were equal between MSICS and phaco throughout the study period using the World Health Organization’s criterion of 20/60 for functional vision.2 Although outcomes and complication rates were comparable, MSICS was much faster and much less expensive to perform and resulted in faster visual recovery.

Figure 7. Postoperative cataract patients in Nepal.
I was struck by the tremendous difficulty of doing phaco when the majority of cataracts were white or ultrabrunescent, the corneas were often scarred from trachoma, and the poor tear film and ocular surface so often marred my surgical view. I concluded that, for the many mature cataracts in this population, I and most Western surgeons would have a much higher complication rate with phaco than with MSICS.
In a subsequent studies, including one of 79,777 consecutive cataract surgeries performed at the Aravind hospital in Madurai, India, we were able to show that, in experienced hands, both phaco and MSICS had comparably low surgical complication rates of approximately 1%.4,5 However, for inexperienced surgeons and trainees, MSICS had a much lower rate of complications, such as posterior capsular rupture.5 Avoiding retained nuclear fragments is crucial in the many developing countries where there may be no access to a vitreoretinal surgeon. We concluded that, to train new cataract surgeons in underserved societies, MSICS should initially be the procedure of choice.
In my own practice, I resort to MSICS for the densest cataracts when I feel that comorbidities, such as a crowded anterior segment, zonulopathy, corneal opacity, or severe miosis may make phaco the riskier procedure for the endothelium or posterior capsule. It is a wonderful option to have in my surgical armamentarium.

A squared posterior optic edge reduces PCO regardless of IOL material. To reduce cost, PMMA IOLs are typically used in conjunction with the large MSICS incision, but they have been associated with higher rates of posterior capsular opacification (PCO). PCO is an inconvenience for patients in developed countries, but in the developing world, where follow-up eye care or an Nd:YAG laser may not be accessible, it is a leading cause of visual impairment. Our prospective, bilateral eye study at the Aravind hospital in Madurai randomized implantation of a round-edged PMMA IOL in one eye and a square-edged PMMA IOL in the other.6 Using digital retroillumination image analysis over 9 years of follow-up, we showed that, with the round-edged optics, the PCO rate did not level off, but continued to climb. The square-edged PMMA IOLs we used significantly prevented PCO, and they were even statistically superior to square-edged AcrySof (Alcon) foldable IOLs used in a separate, parallel 9-year study.
This is the longest-term bilateral eye study prospectively comparing the impact of IOL edge design on PCO published to date. It demonstrated that this inexpensive edge modification, costing approximately US$1 per IOL, might markedly reduce a major cause of visual impairment in developing countries.

Intracameral moxifloxacin prophylaxis is safe and effective. Because of the expense and difficulty of performing a randomized controlled trial, some of our best evidence on endophthalmitis prophylaxis has come from large retrospective studies. Our recent study of charity patients at the Aravind hospital in Madurai compared 37,777 consecutive surgeries without intracameral (IC) antibiotic to 38,160 consecutive surgeries with IC moxifloxacin prophylaxis over a 14-month period.7 Nearly all surgeries (97%) were performed with MSICS technique.
AECS’s affiliated pharmaceutical company, Aurolab, manufactures a moxifloxacin solution commercially approved for intraocular use in India (Auromox), and one US$1 vial is used for five patients. The use of a single US$0.20 dose of IC moxifloxacin reduced the endophthalmitis rate from 0.08% in those who did not receive the IC drug to 0.02% in those who did.
We believe an IC antibiotic should be used, if available, when MSICS is employed in the developing world. We recently published additional data from 617,453 consecutive phaco and MSICS cases performed at all 10 regional AECS hospitals over a 29-month period.8 IC moxifloxacin reduced the endophthalmitis rate from 0.07% in the 302,815 cases without antibiotic to 0.02% in the 314,638 cases with antibiotic. In a follow-up correspondence, we will soon report an identical infection rate reduction with IC moxifloxacin in more than 1 million consecutive eyes over a 4-year period. This is the largest published efficacy data for IC moxifloxacin prophylaxis, and it is the basis for my use of compounded IC moxifloxacin in my own cataract patients.

Many OR regulations may unnecessarily increase costs without proven benefit. At one time, one of the main operating room (OR) regulatory agencies in the United States began mandating a full, wrapped terminal sterilization cycle for eye surgical instruments for every case. There was no published evidence to validate that commonly used shorter steam sterilization cycles were safe and effective. A subsequently published study of more than 42,000 consecutive cases at the Aravind hospital in Pondicherry was a key paper used to substantiate the safety of shorter cycle sterilization of ophthalmic instruments in the United States.9 This study also documented a low endophthalmitis rate of 0.09% despite the reuse of gowns, gloves, surgical tubing, irrigating solutions, and a number of instruments and drugs limited to single use in the United States. Another concern with reuse of instruments is the risk of toxic anterior segment syndrome (TASS). Our study at the Aravind hospital in Pondicherry found the rate of TASS to be 0.22% in 26,408 consecutive eyes, and included two identifiable clusters.10 This is the only large study to report the sporadic incidence of TASS, and we reported a very low incidence of serious long-term complications.
As noted previously, charity patients account for 60% of AECS’s cataract surgical volume. Therefore, maintaining safety and quality while reducing unnecessary costs and procedural steps is necessary to maximize surgical efficiency and access to those blinded by cataract. Surgeons at both AECS and Tilganga further improve efficiency by operating on multiple patients simultaneously in the same OR. Despite the use of innumerable OR practices that would never be permitted in the United States, the endophthalmitis rate in more than 528,000 consecutive cataract surgeries at the 10 AECS facilities following routine adoption of IC moxifloxacin is only 0.02%.8

Figure 8. The author and Dr. Haripriya in Madurai, India, 2015.

Figure 9. The author and Dr. Ruit in Kathmandu, Nepal, 2005.
CONCLUSION
Low-tech MSICS through larger incisions, use of extremely low-cost IOLs and supplies, reuse of as many supplies as possible, and 10 case per hour assembly-line efficiency would be unthinkable practices in developed countries. However, solving the challenge of eradicating cataract blindness in the developing world requires innovative and sometimes counterintuitive thinking. The noble surgeons at the Aravind Eye Care System and Tilganga Eye Hospitals should be celebrated not only for their compassion, but also for the lessons that they can teach the rest of us (Figures 8 and 9).
1. Chang DF. Tackling the greatest challenge in cataract surgery [editorial]. Br J Ophthalmol. 2005;89(9);1073-1074.
2. Ruit S, Tabin G, Chang DF, et al. A prospective randomized clinical trial of phacoemulsification vs manual sutureless small incision extracapsular cataract surgery in Nepal. Am J Ophthalmol. 2007;143:32-38.
3. Venkatesh R, Chang DF, Muralikrishnan R , Hemal K, Gogate P, Sengupta S. Manual small incision cataract surgery: a review. Asia Pac J Ophthalmol (Phila). 2012;1(2):113-119.
4. Venkatesh R, Tan CS, Sengupta S, Ravindran RD, Krishnan KT, Chang DF. Phacoemulsification versus manual small-incision cataract surgery for white cataract. J Cataract Refract Surg. 2010;36:1849-1854.
5. Haripriya A, Chang DF, Reena M, Shekhar M. Analysis of complication rates of phacoemulsification and manual small-incision cataract surgery at Aravind Eye Hospital. J Cataract Refract Surg. 2012;38:1360-1369.
6. Haripriya A, Chang DF, Vijayakumar B, et al. Long-term posterior capsule opacification reduction with square-edge polymethylmethacrylate intraocular lens. Ophthalmology. 2017;124:295-302.
7. Haripriya A, Chang DF, Namburar S, Smita A, Ravindran RD. Efficacy of intracameral moxifloxacin endophthalmitis prophylaxis at Aravind Eye Hospital. Ophthalmology. 2016;123:302-308.
8. Haripriya A, Chang DF, Ravindran RD. Endophthalmitis reduction with intracameral moxifloxacin prophylaxis: analysis of 600,000 surgeries [published online ahead of print February 14, 2017]. Ophthalmology.
9. Ravindran RD, Venkatesh R, Chang DF, et al. The incidence of post-cataract endophthalmitis at an Aravind Eye Hospital: Outcomes from more than 42,000 consecutive cases using standardized sterilization and prophylaxis protocols. J Cataract Refract Surg. 2009;35:629-636.
10. Sengupta S, Chang DF, Gandhi R, et al. Incidence and long-term outcomes of toxic anterior segment syndrome at Aravind Eye Hospital. J Cataract Refract Surg. 2011;37:1673-1678.



