

My wife Nikki reads and remembers everything. When I was walking in the door one day in 2012, she confronted me with a question: “How many Down syndrome patients have you seen in your keratoconus consultation so far?” She was holding a scientific journal in her hand, and she had just read statistics indicating that people with Down syndrome are more susceptible to keratoconus than the general population.
Her question was the impetus and the starting point for an international nonprofit initiative that is now called Light for Sight 21, under a Swiss umbrella organization called the Light for Sight Foundation.
AT A GLANCE
• The Down syndrome population has a higher risk for keratoconus than the general population due to genetic disposition.
• Light for Sight 21, an international nonprofit initiative started by the authors, was initially designed as a keratoconus screening program for Down syndrome patients, but the call to action has since expanded to include keratoconus screenings for all children and adolescents.
I was part of the original team of scientists and clinicians that introduced CXL technology to ophthalmology, and I have since built one of the largest keratoconus consultation centers in Switzerland. Although keratoconous is considered rare, we know that certain subpopulations have much higher risk levels for the disease because of hormonal influences, environmental causes, or behaviors such as eye rubbing. We also understand that the Down syndrome population has a higher risk for keratoconus than the general population due to genetic disposition.
However, in 10 years of practicing CXL, I have seen only three Down syndrome patients. Therefore, a question presented itself: Who is screening, detecting, and treating this patient group, if anyone at all?
To try to answer this question, we called upon our global network of cornea specialists to see if they had noticed a proportionally larger representation of Down syndrome patients presenting for consultation in their practices. Everyone who responded agreed that there is an elevated risk in this patient group, but no one had focused specifically on targeting or reaching this group of people. We decided to start an outreach effort locally to see the response before initiating an international call to action.
STARTING LOCALLY, THINKING GLOBALLY
From 2010 to 2014, I was the Medical Director of the Geneva University Eye Hospital and Chair of Ophthalmology at the University of Geneva. At the same time, Stylianos E. Antonarakis, MD, DSc, was the head of genetic medicine at the university. Professor Antonarakis is a world-renowned medical geneticist and a celebrity in the Down syndrome medical community. In 2012, Nikki and I met with him to understand how best to approach this community to initiate a screening program, and we determined that a simple referral network was the easiest and fastest way.
I dedicated one resident, David Tabibian, MD, and one clinical day per month for seeing only Down syndrome patients through internal referrals from the university’s medical genetics team. We indeed detected several patients with suspicious corneas and even with frank keratoconus over a period of 2 years (Figure 1).

Figure 1. Dr. Tabibian performs Pentacam (Oculus) measurements in a young patient with Down syndrome.
In parallel with our efforts at the university, we began to expand the Light for Sight network by adding international sites. Several of our friends and colleagues started their own outreach efforts. In Antwerp, Belgium, Marie-Jose Tassignon, MD, PhD, and Carina Koppen, MD, PhD, organized a symposium at their national ophthalmic congress dedicated to understanding eye diseases of the Down syndrome community (Figure 2). The purpose of their symposium was to educate general ophthalmologists about keratoconus. In Istanbul, Turkey, Efekan Coskukseven, MD, directly approached local Down syndrome patient organizations and opened his private consultation to free screenings for all Down syndrome children and their families (Figure 3A). In Thessaloniki, Greece, Miltos Balidis, MD, PhD, FEBO, organized a national media campaign in conjunction with World Down Syndrome Day to promote the importance of annual eye screenings (Figure 3B and 3C).

Figure 2. The authors (left) at the Down Syndrome Symposium at the 2012 Meeting of the Belgian Ophthalmological Society, organized by Professors Tassignon (third from left), and Koppen (right).

Figure 3. Keratoconus screening at the Dünyagöz Eye Hospital in Istanbul, Turkey, performed by Efekan Coskunseven, MD (A). Inauguration of the Thessaloniki Partner Site Ophthalmica.gr (B); with Miltos Balidis, MD, PhD, FEBO, (left) and Farhad Hafezi, MD, PhD (right; C).
EXPANDING THE FOCUS
Although our focus started on Down syndrome patients, Nikki and I decided to expand the call to action to include keratoconus screenings for all children and adolescents. Because keratoconus is one of the leading causes of preventable blindness in children, we believe that awareness and access to treatment are pillars in combating this debilitating disease.
While we are focused on reaching high-risk population groups, we also aim to support educational activities to teach vision care specialists about keratoconus and encourage them to generate public service announcements and contact local schools to implement annual eye screening programs.
Sheraz M. Daya, MD, FACP, FACS, FRCS(Ed), FRCOphth, in collaboration with the company formerly known as IROC Innocross (since acquired by Avedro), trained a group of ophthalmologists in Tanzania, using a donated UV-X 2000 lamp (Avedro), in how to detect keratoconus and how to treat the disease with CXL (Figure 4). A number of other companies in the CXL and keratoconus/contact lens industry have voiced their support to help supply equipment and products for these types of humanitarian efforts (Figure 5).
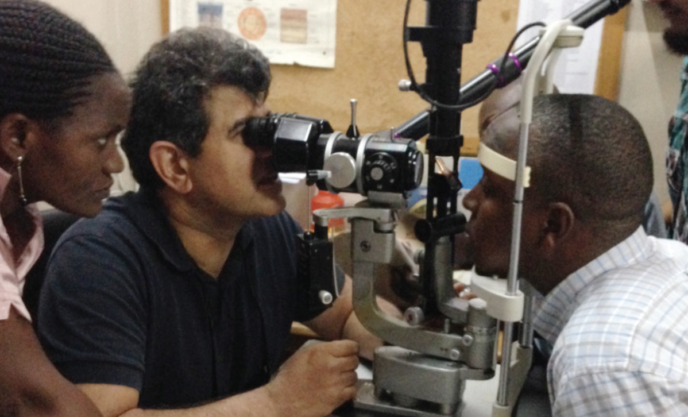
Figure 4. Sheraz M. Daya, MD, FACP, FACS, FRCS(Ed), FRCOphth, examines a patient with keratoconus in Tanzania.

Figure 5. Nikki Hafezi, MAS IP, ETHZ, and Mannie Ng, a keratoconus patient treated by Light for Sight, with a donation from Interlens, an association of contact lens practitioners in Switzerland.
Since 2005, I have treated more than 200 pediatric patients with keratoconus, three of whom were Down syndrome patients identified through the Light for Sight 21 screening program. Although a lot of work must still be done in outreach efforts to the Down syndrome community, I know that these three special patients retained their vision and quality of life specifically due to our efforts.
TREATMENT CONSIDERATIONS
I choose not to wait for documented progression before treating these patients because studies have shown that, in almost 90% of documented pediatric keratoconus cases, the disease will progress. Waiting may mean that our treatment will only stabilize the cornea in a worse state.
Compliance is always an issue when dealing with children or mentally challenged individuals. We have developed a Light for Sight protocol for poorly compliant and noncompliant patients (Figure 6). This protocol includes an individual assessment of the risks of the procedure (postoperative corneal infection due to low intra- and postoperative compliance) versus the efficacy of the method. Under certain circumstances, we advocate the use of epithelium-on (epi-on) CXL. Although epi-on CXL is less efficient than classic epithelium-off CXL, it carries a lower risk for postoperative infection, which might under certain circumstances be a determining factor in treatment decisions.
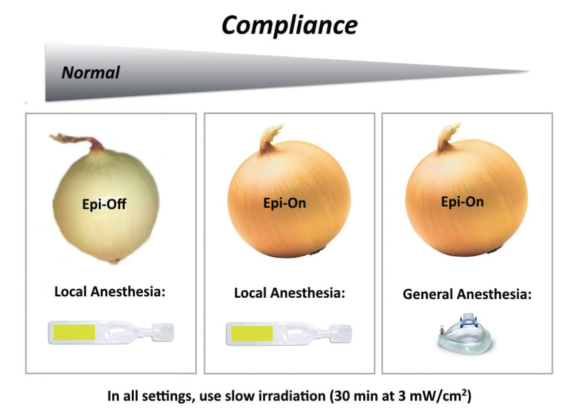
Figure 6. The Light for Sight protocol for patients with normal compliance, reduced compliance, and absence of compliance.
NOW AND THE FUTURE
The Light for Sight network already comprises 45 partner sites in 23 countries, and it continues to grow. To become a partner referral site, there are three requirements:
No. 1: The operating surgeon must have performed CXL for 3 years or for a total of 100 procedures;
No. 2: There must be access to general anesthesia in case of a noncompliant patient; and
No. 3: The surgeon must waive his or her honorarium for patients who are unable to afford treatment.
Partner referral sites will be included on the list of partners/associates on the Light for Sight website. Partners will also be invited to participate in collaborative research projects and will be used as a patient referral center for their city or region.
The Light for Sight Foundation’s mission is to eliminate preventable blindness caused by keratoconus in children and adolescents. The foundation focuses on increasing awareness of the disease among medical professionals with pediatric patients and on empowering parents with information about the importance of annual eye exams. We pay special attention to high-risk groups such as those in the Down syndrome community, and we depend on the support of our clinical and industrial partners to fulfill this mission.


