In most IOLs intended for fixation in the capsular bag, the haptics are formed of two loops with a C shape. One can call this familiar style a two-C-loop implant (Figure 1). This configuration has a drawback: The optic-haptic junctions, diametrically opposed, create an axis that allows tilting of the IOL.1 Tilt of the IOL optic results in astigmatism. With a 24.00 D lens, a 10° tilt induces 0.75 D of astigmatism.
Moreover, tilt leads frequently to partial optic capture in the capsulorrhexis. After pupil dilation, this can be seen at the slit lamp in approximately one-third of eyes with a two-C-loop implant (Figure 2). The loss of overlap of the IOL optic by the capsule margin changes the effective lens position (ELP).2 It can also facilitate PCO development (Figure 3).
An IOL with a four-C-loop haptic configuration, with the optic-haptic junctions at the cardinal points, could prevent optic tilt in the capsulorrhexis. These considerations led me to design two new implants, both described herein.
IMPLANT DESIGNS
The first implant has four open C-loops in the cardinal positions. The supplementary loops increase the volume that must pass through the delivery system. This increase is compensated by a reduction of volume in the optic portion using a lenticular configuration. In the past 2 years, I have implanted this one-piece, four-C-loop IOL, the FA7010 (Zabby’s; Figure 4), in more than 200 eyes. In my experience, the design prevents optic capture in the capsulorrhexis opening.
At a Glance
- Two-C-loop IOLs may tilt in the capsulorrhexis and may vary in effective lens position.
- Four-C-loop IOLs with haptics at the four cardinal points cannot tilt in the capsulorrhexis.
- The Oval Ring four-C-loop IOL has the additional advantage of limiting variation of the effective lens position.
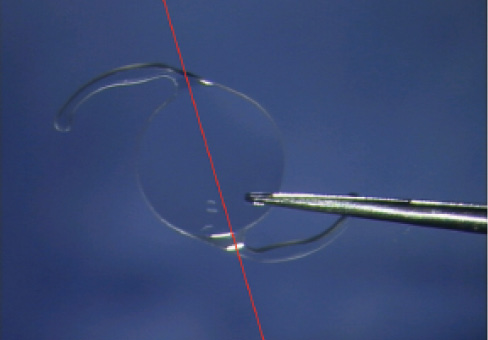
Figure 1. In two-C-loop IOLs, the optic-haptic junctions constitute an axis that allows tilt.
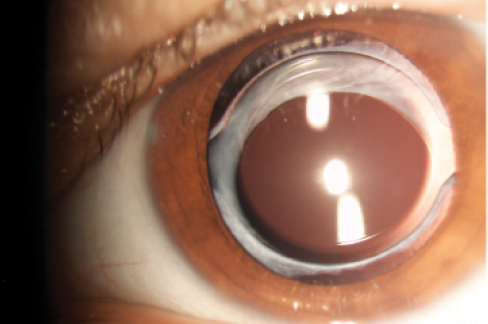
Figure 2. One-year postoperative, tilt of the optic in the capsulorrhexis opening is observed in about one-third of two-C-loop IOLs.
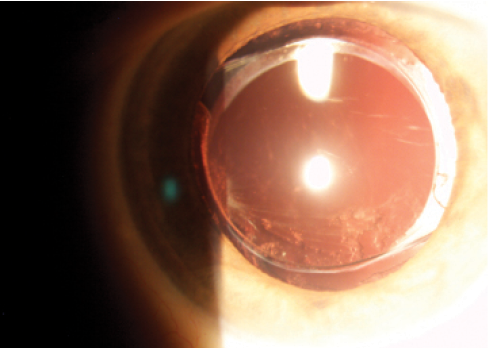
Figure 3. Optic tilt facilitates migration of lens epithelial cells and, thus, PCO development.

Figure 4. A four-C-loop haptic design (FA7010) prevents optic tilt.
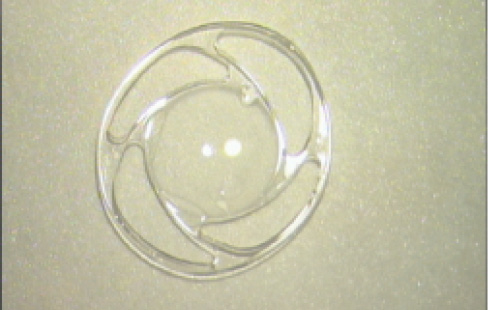
Figure 5. The Oval Ring four-C-loop IOL (42A) has the advantage of improving the consistency of the effective lens position and adapting to the size of the capsular bag.
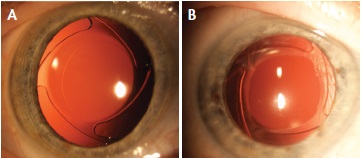
Figure 6. Oval Ring IOL in situ, early postoperative (A) and 1-year postoperative (B).
In the second implant design, the four cardinal C-loops are joined by a peripheral ring. This one-piece IOL was developed with Michel Delmelle, PhD, a physicist and former professor of physics at the Université de Liège, Belgium, and the founder of the Belgian IOL manufacturer PhysIOL. The lens model, now made by Morcher (Oval Ring 42A; Figure 5) is particularly sophisticated because it reduces the variations in ELP that are frequent with any foldable IOL.
We knew that a haptic system with 360° contact in the equator of the capsular bag might limit anteroposterior shifts of the optic.3 The inconvenience of a circular ring is its lack of adaptation to the diameter of the capsular bag. The Oval Ring 42A haptic system comprises an oval-shaped ring that allows a radial flexibility similar to open-loop haptics.
I have used the oval ring implant in more than 100 cases over a period of 1 year and found that it consistently prevented optic capture in the capsulorrhexis. Moreover, it resulted in a more consistent ELP than conventional implants in the capsular bag. Under coaxial illumination at the slit lamp, centration was excellent (Figure 6A) and stable (Figure 6B).
PREMIUM HAPTICS
These two new IOL models, but especially the Oval Ring 42A, deserve to be presented as premium-haptic IOLs. The four-C-loop configuration is a rational improvement, avoiding the tilt that is a common imperfection in the postoperative position of two-C-loop implants in the capsular bag. The one-piece Oval Ring IOL has the additional advantage of decreasing variation in ELP.
In my own series of two-C-loop implants, I have observed a dispersion of postoperative refractive errors of up to 2.40 D in spherical equivalent.4 With the Oval Ring four-C-loop IOL, the degree of dispersion is to date 1.65 D, and the prediction error is less than 0.50 D in 68% of cases. Theoretically, modifications in dimensions of the haptic and/or the optic in the Oval Ring IOL concept could lead to further improvement in the consistency of the ELP. n
1. de Castro A, Rosales P, Marcos S. Tilt and decentration of intraocular lenses in vivo from Purkinje and Scheimpflug imaging: Validation study. J Cataract Refract Surg. 2007;33:418-429.
2. Findl O, Struhal W, Dorffner G, Drexler W. Analysis of nonlinear systems to estimate intraocular lens position after cataract surgery. J Cataract Refract Surg. 2004;30:863-866.
3. Galand A. Possible improvements in IOL design. Electronic poster presented at: XXX Congress of the ESCRS; September 8-12, 2012; Milan, Italy.
4. Galand A. The Effective lens position is more constant with MultiLink haptic than with J-loop. Electronic poster presented at: XXXI Congress of the ESCRS; October 5-9, 2013; Amsterdam, Netherlands.
Albert Galand, MD, PhD
- Cataract Clinic, Neupré, Belgium
- Former Head of the Ophthalmology Department,
Hospital of the Université de Liège, Belgium
- professeur.galand@gmail.com
- Financial disclosure: Registered designs on haptic/optic
configurations

