SMILE: The Future is Minimally Invasive
The lenticule extraction technique is well established after only 3.5 years.
By Marcus Blum, MD
For several years, early in its development, the use of femtosecond laser systems in refractive surgery was restricted to flap creation, while the actual refractive ablation remained the domain of the excimer laser. Once flap creation with femtosecond lasers had demonstrated advantages over mechanical microkeratomes with regard to the quality of the surgical outcome, it was a logical step to investigate methods for refractive laser correction using this technology only.
A refractive procedure that required no excimer laser was brought to market by Carl Zeiss Meditec in September 2011. The procedure was introduced as ReLEx small incision lenticule extraction (SMILE), and the first series of 91 eyes was reported by Sekundo.1
SMILE is based on the removal of a disc of tissue—a lenticule—rather than tissue ablation. The refractive lenticule is created in the intact cornea using the VisuMax femtosecond laser (Carl Zeiss Meditec). The refractive correction is achieved by passing a dissector though a small 2- to 3-mm incision, separating the anterior and posterior lenticular interfaces, and then removing the lenticule though the incision (Figure 1). This eliminates the need to create a flap and makes SMILE a minimally invasive procedure. The cornea above the upper interface of the lenticule is referred to as the cap.
Practitioners of SMILE have reported that visual outcomes, predictability, and safety in low, moderate, and high myopes are comparable with or even better than with femtosecond LASIK (femto-LASIK).2,3 Additionally, SMILE offers a number of further potential advantages that have also been reported (see Potential Advantages of SMILE).
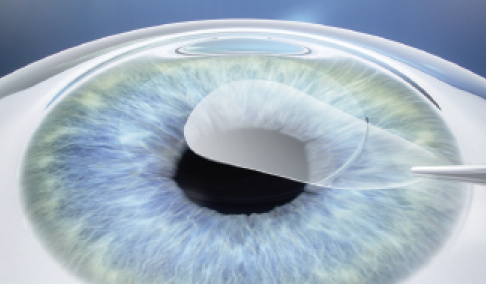
Figure 1. SMILE is a flapless and bladeless procedure based on the removal of a disc of tissue, called a lenticule, through a small incision.
ROUTINE CORRECTION
We now offer SMILE to our patients in Erfurt, Germany, as a routine refractive correction option. Except in conditions in which SMILE is not indicated, such as hyperopia, we treat all suitable patients with SMILE.
The SMILE procedure has become established in the market only 3.5 years after its international commercial introduction. However, there is great potential for further investigations and for broadening the range of indications, and studies using the SMILE technique to treat hyperopia have already begun. Still, there is a need for long-term follow-up studies to further validate the safety and efficacy of this concept and its assumed potential. I am sure that the future of laser vision correction will increasingly include minimally invasive procedures and that we will hear quite a lot about SMILE during the coming years.
1. Sekundo W, Kunert KS, Blum M. Small incision femtosecond lenticule extraction (SMILE) for the correction of myopia and myopic astigmatism: Results of a 6 months prospective study. Br J Ophthalmol. 2011;95:335-339.
2. Kamiya K, Igarashi A, Ishii R, Sato N, Nishimoto H, Shimizu K. Early clinical outcomes, including efficacy and endothelial cell loss, of refractive lenticule extraction using a 500 kHz femtosecond laser to correct myopia. J Cataract Refract Surg. 2012;38:1996-2002.
3. Ivarsen A, Asp S, Hjortdal J. Safety and complications of more than 1500 small-incision lenticule extraction procedures. Ophthalmology. 2014;121:822-828.
Potential Advantages of SMILE
Surface ablation and flap cutting cause loss of the subepithelial nerve plexus. SMILE generates less damage due to the small size of the pocket incision with a lower level of collateral damage and decellularization. Comparative outcome measurements have shown fewer dry eye symptoms in SMILE patients in comparison with other corneal refractive procedures.1-3
In SMILE, the Bowman layer is disrupted only along a small peripheral incision. This minimizes the biomechanical weakening of the cornea and leaves the anterior stroma more stable than in LASIK. No case of postoperative kerectasia has been reported after SMILE, and this procedure is likely to be useful to correct higher amounts of refractive error than other corneal procedures.4,5
At 1-year follow-up, better centration of the refractive zone, significantly less induction of higher-order aberrations, and better mesopic midterm contrast sensitivity were achieved with ReLEx than with wavefront-optimized femto-LASIK.6
1. Riau AK, Angunawela RI, Chaurasia SS, Lee WS, Tan DT, Mehta JS. Early corneal wound healing and inflammatory responses following refractive lenticule extraction (ReLEx). Invest Ophthalmol Vis Sci. 2011;52:6213-6221.
2. Wei S, Wang Y. Comparison of corneal sensitivity between FS-LASIK and femtosecond lenticule extraction (ReLEx flex) or small-incision lenticule extraction (ReLEx smile) for myopic eyes. Graefes Arch Clin Exp Ophthalmol. 2013;251:1645-1654.
3. Vestergaard AH, Gronbech KT, Grauslund J, Ivarsen AR, Hjortdal JO. Subbasal nerve morphology, corneal sensation, and tear film evaluation after refractive femtosecond laser lenticule extraction. Graefes Arch Clin Exp Ophthalmol. 2013;251:2591-2600.
4. Wu D, Wang Y, Zhang L, Wei S, Tang X. Corneal biomechanical effects: Small-incision lenticule extraction versus femtosecond laser-assisted laser in situ keratomileusis. J Cataract Refract Surg. 2014;40:954-962.
5. Roy AS, Dupps WJ, Roberts CJ. Comparison of biomechanical effects of small-incision lenticule extraction and laser in situ keratomileusis: Finite element analysis. J Cataract Refract Surg. 2014;40:971-980.
6. Gertnere J, Solomatin I, Sekundo W. Refractive lenticule extraction (ReLEx flex) and wavefront-optimized femto-LASIK: comparison of contrast sensitivity and high-order aberrations at 1 year. Graefes Arch Clin Exp Ophthalmol. 2013;251:1437-1442.
LASIK and PRK: Safety and Efficacy Continues to Improve
Sophisticated technologies, improved surgical skills, and optimal patient selection are key in boasting postoperative outcomes.
By Massimo Busin, MD
LASIK has been the dominant refractive surgery procedure in most countries in recent decades. Its main advantage over PRK consists in the preservation of the corneal epithelium over the flap created by a microkeratome or a femtosecond laser before excimer laser treatment of the exposed stromal surface.
At a Glance
- Practitioners of SMILE have reported that visual outcomes; predictability; and safety in low, moderate, and high myopes are comparable with or even better than with femto-LASIK.
- Although alternatives to LASIK exist, including the fairly recently introduced procedure ReLEx SMILE and implantation of phakic IOLs, neither will replace LASIK in the near future.
- Phakic IOLs offer a lenticular rather than corneal approach to refractive surgical correction.
Preservation of the epithelium leads to increased comfort during the early postoperative period, faster visual recovery, and reduced wound healing response in comparison with PRK. Reduced wound healing correlates with less regression for high corrections and a lower rate of complications such as significant stromal opacity or haze.
PRK, however, remains an excellent option for correction of mild to moderate degrees of myopia, particularly in eyes with thin corneas or recurrent erosions, and for patients with a predisposition to trauma (eg, those who practice martial arts or are in the military).
LASIK has been considered a riskier procedure than PRK because of the possibility of complications occurring during the creation and subsequent manipulation of the flap. Additionally, if the flap is not properly cut, it may fail to adhere to the stromal surface and/or present microscopic wrinkles or striae, leading to poor visual outcomes. Studies suggest that flap complications occur in 0.3% to 5.7% of LASIK procedures.1
Dry eye syndrome is another drawback experienced by many post-LASIK patients. Some patients after LASIK experience a marked decrease in tear production that can cause eye discomfort and blurred vision. In one study, almost half of all LASIK patients reported some degree of temporary dry eye syndrome;1 however, post-LASIK dry eye syndrome is usually transient and can be effectively treated with lubricating eye drops. It usually disappears when healing of the cornea is completed, which may require up to 6 months.
The safety and effectiveness of LASIK continues to improve, thanks to increasingly sophisticated technology, improved surgical skills, and optimal patient selection. In a study of 3,000 consecutive LASIK procedures employing a femtosecond laser for flap creation, the incidence of flap-related complications was 0.37%, and all complications were managed successfully during the same surgical session.1 For these reasons and more, I predict that LASIK will continue to be one of the most dominant keratorefractive surgery techniques employed in the coming years.
COMPARED WITH ALTERNATIVE PROCEDURES
Although alternatives to LASIK exist, including the fairly recently introduced procedure ReLEx SMILE and implantation of phakic IOLs, neither will replace LASIK in the near future.
ReLEx SMILE. This procedure induces fewer higher-order aberrations (HOAs) and spherical aberration than a femtosecond LASIK procedure,2-4 and it has a less pronounced impact on the ocular surface and corneal innervation5 and in return a reduced incidence of dry eye;6-9 but, there are concerns related to the postoperative risk of keratectasia after SMILE. In my opinion, these concerns seem unjustified in view of several publications showing that the postoperative tensile strength of the cornea is considerably higher after SMILE than after either PRK or LASIK, but, for this and other reasons, the widespread adoption of SMILE is still years away. Considering that the Bowman layer remains untouched with SMILE, the procedure may allow the correction of higher levels of myopia in the future.10,11
Phakic IOLs. One year postoperatively, phakic IOL implantation appears to be safer than excimer laser surgical correction for treatment of moderate to high myopia,12 and it results in significantly less reduction of BCVA, better contrast sensitivity than excimer laser correction for moderate to high myopia, and better acceptance by patients.12 However, analysis of long-term results of phakic IOL implantation will be needed to rule out an increase in more severe complications affecting the safety and efficacy of this procedure, namely early cataract onset, corneal decompensation, and rhegmatogenous retinal detachment.
CONCLUSION
Results after LASIK surgery are good now, and the trend has been continued improvement. With the addition of other surgical techniques for the correction of refractive errors surfacing, LASIK volume may go down, but it will not disappear.
1. Schallhorn SC, Amesbury EC, Tanzer DJ. Avoidance, recognition, and management of LASIK complications. Am J Ophthalmol. 2006;141(4):733-739. Review.
2. Reinstein DZ, Carp GI, Archer TJ, Gobbe M. Outcomes of small incision lenticule extraction (SMILE) in low myopia. J Refract Surg. 2014;30(12):812-818.
3. Lin F, Xu Y, Yang Y. Comparison of the visual results after SMILE and femtosecond laser-assisted LASIK for myopia. J Refract Surg. 2014;30(4):248-254.
4. Ganesh S, Gupta R. Comparison of visual and refractive outcomes following femtosecond laser-assisted LASIK with SMILE in patients with myopia or myopic astigmatism. J Refract Surg. 2014;30(9):590-596.
5. Mohamed-Noriega K, Riau AK, Lwin NC, Chaurasia SS, Tan DT, Mehta JS. Early corneal nerve damage and recovery following small incision lenticule extraction (SMILE) and laser in situ keratomileusis (LASIK). Invest Ophthalmol Vis Sci. 2014;55(3):1823-1834.
6. Denoyer A, Landman E, Trinh L, Faure JF, Auclin F, Baudouin C. Dry eye disease after refractive surgery: comparative outcomes of small incision lenticule extraction versus LASIK [published online ahead of print November 22, 2014]. Ophthalmology.
7. Gao S, Li S, Liu L, et al. Early changes in ocular surface and tear inflammatory mediators after small-incision lenticule extraction and femtosecond laser-assisted laser in situ keratomileusis. PLoS One. 2014;9(9):e107370.
8. Xu Y, Yang Y. Dry eye after small incision lenticule extraction and LASIK for myopia. J Refract Surg. 2014;30(3):186-190.
9. Li M, Zhao J, Shen Y, et al. Comparison of dry eye and corneal sensitivity between small incision lenticule extraction and femtosecond LASIK for myopia. PLoS One. 2013;8(10):e77797.
10. Reinstein DZ, Archer TJ, Randleman JB. Mathematical model to compare the relative tensile strength of the cornea after PRK, LASIK, and small incision lenticule extraction. J Refract Surg. 2013;29(7):454-460.
11. Wang D, Liu M, Chen Y, et al. Differences in the corneal biomechanical changes after SMILE and LASIK. J Refract Surg. 2014;30(10):702-707.
12. Barsam A, Allan BD. Excimer laser refractive surgery versus phakic intraocular lenses for the correction of moderate to high myopia. Cochrane Database Syst Rev. 2014 Jun 17.
Phakic IOLs Will Continue to Complement Cornea-Based Procedures
The future holds great promise for phakic IOLs.
By Alaa El-Danasoury, MD, FRCS(Ed)
Since the introduction of the excimer laser to refractive surgery, keratorefractive procedures have become the standard of care for a wide range of refractive errors. Excimer laser ablative techniques including PRK and LASIK have been shown to be safe, effective, and accurate for the correction of myopia, hyperopia, and astigmatism within certain limits.
As versatile as the excimer laser is, however, not every refractive error can be corrected on the cornea—particularly very high myopia. Additionally, although the vast majority of LASIK and PRK cases are performed without complications or sequelae, iatrogenic ectasia remains a concern after photoablative procedures, especially in eyes with high refractive errors or suspicious corneas. In recent years, better understanding of the effects of flap creation and laser ablation on corneal biomechanics has ensued. At the same time, the effects of HOAs on quality of vision have been increasingly appreciated, and investigators have recognized that corneal ablation patterns can induce increases in an eye’s HOAs.
Phakic IOLs have continued to be commercially available in a number of styles, offering a lenticular rather than corneal approach to refractive surgical correction. Phakic IOLs have passed through many stages of development since Strampelli and Barraquer first proposed the technology in the 1950s. In the modern era, there have been three basic phakic IOL designs: angle-supported and iris-supported lenses in the anterior chamber and sulcus-supported lenses in the posterior chamber. Currently no angle-supported phakic IOL model is available. Iris-supported phakic IOLs include the rigid Artisan (Ophtec; marketed in the United States as the Verisyse by Abbott Medical Optics) and the foldable Artiflex (Ophtec; marketed in the United States as the Veriflex by Abbott Medical Optics).

Figure 2. The KS-Aquaport is designed to restore more natural aqueous flow and eliminate the need for an iridotomy.
One commonly used posterior chamber phakic IOL is the Visian Implantable Collamer Lens (ICL; STAAR Surgical), also available in a toric version (Toric ICL; STAAR Surgical). The most recent iteration of the ICL, the Visian ICL V4c, includes a central hole that enables more physiologic flow of aqueous through the pupil, eliminating the need for a peripheral iridectomy.
Candidates for phakic IOL implantation must meet certain anatomic criteria, including an adequately deep anterior chamber (at least 3 mm), an open angle, a flat or concave iris configuration, and a healthy endothelium.
There are numerous potential advantages of phakic IOLs over excimer laser vision correction. Most important may be the preservation of corneal biomechanics. This eliminates concerns over iatrogenic ectasia and the induction of HOAs through laser ablation. In moderate and high corrections, phakic IOLs have minimal effect on quality of vision—a major concern with keratorefractive procedures. Other advantages are listed in Benefits Associated With Phakic IOL Implantation.
The primary disadvantage of anterior chamber phakic IOLs is their proximity to the corneal endothelium, making continued monitoring of corneal health a priority. Iris-fixated lenses are closer to the endothelium than posterior chamber lenses, and the rigid versions of these lenses require large incisions. The primary disadvantage of posterior chamber phakic IOLs is their proximity to the crystalline lens, with the risk of inducing lenticular opacities. The incidence of induced cataract after ICL implantation is very low; moreover, the central hole introduced with the latest version of the ICL may help to reduce the already low incidence of cataract (Figure 2); long-term results will reveal whether this is the case.
Benefits Associated With Phakic IOL Implantation
- Reversible procedure: Unlike corneal tissue ablation, phakic IOLs can be removed or exchanged
- High predictability: Refractive outcome is not modulated by individual corneal tissue response, especially in high refractive errors
- Preservation: Phakic IOL implantation preserves the tear film and corneal nerves as well as natural accommodation (at least in prepresbyopic patients)
Sizing may be the greatest challenge for certain phakic IOL styles, the exception being the one-size-fits-all iris-fixated lenses. Angle-supported lenses must be selected based on the angle-to-angle distance, which is best measured by anterior chamber OCT. An undersized angle-supported lens can rotate in the angle and damage the angle or endothelium. Sulcus-supported lenses are sized based on the sulcus-to-sulcus diameter, which is best determined with high-frequency ultrasound. Undersizing of these lenses can leave a low vault, with increased risk of lens touch. Rotation of an undersized Toric ICL can lead to a poor refractive result.
THE FUTURE
The future holds great promise for phakic IOLs, as all manufacturers continue to refine their designs. Additionally, development of better diagnostic instruments will assist with lens sizing. Hopefully an angle-supported lens design will be reintroduced to the market. A preloaded version of the ICL, introduced last year, improved the implantation procedure for that lens.
Finally, it must be recognized that phakic IOLs and laser refractive surgery are complementary modalities, not competitors. Both surgical options are important components of the comprehensive refractive surgeon’s armamentarium. While keratorefractive surgery may address patients with low to moderate refractive errors, phakic IOLs, in my experience, are a better option for patients who have higher myopia, thin corneas, or suspicious topographies, or who are at a higher risk of ectasia, so that safe refractive surgery with a good quality of vision can be offered to all appropriate candidates. n
Marcus Blum, MD
– Augenklinik, Helios-Klinikum Erfurt, Erfurt, Germany
– marcus.blum@helios-kliniken.de
– Financial disclosure: None
Massimo Busin, MD
– Head of the Department of Ophthalmology, Villa Serena Hospital, Forli, Italy
– mbusin@yahoo.com
– Financial disclosure: None
Alaa El-Danasoury, MD, FRCS(Ed)
– Chief, Cornea and Refractive Surgery Service, Magrabi Eye Hospitals and Centers, Jeddah, Saudi Arabia
– malaa@magrabi.com.sa
– Financial disclosure: Consultant (STAAR Surgical)