In the past 4 years, femtosecond lasers have been used in cataract surgery for the creation of corneal incisions and lens capsulotomy and for lens fragmentation. This appears to be a rapidly evolving technology that is attracting worldwide attention, mainly because cataract surgery is the most frequently performed surgical procedure in ophthalmology.1
Use of the femtosecond laser guarantees stability and precision in the length, shape, and width of corneal incisions.2 Another unique advantage is the ability to image the cutting process, which can be done before, during, and after surgery. This is possible because the laser uses a 3-D scanning process, and the essential 3-D beam delivery for acquiring a 3-D image is already part of the system. Thus, the target tissue can be scanned without making room for additional scanning mirrors and lenses for OCT or other imaging technologies.3
Cataract surgeons continue to adopt femtosecond technology to perform laser capsulotomy, lens fragmentation, clear corneal incisions, and limbal relaxing incisions.4 A precise central continuous curvilinear capsulorrhexis (CCC) is a prerequisite for good postoperative effective lens position. Irregular capsulotomies may cause refractive surprises such as myopic or hyperopic shifts, unwanted surgically induced astigmatism (SIA), posterior chamber IOL tilt, increase in higher-order aberrations, and glare and halo phenomena.1 Additionally, femtosecond lasers are being used to address other issues associated with manual cataract surgery by improving precision and reducing risks.5-7
This article presents the outcomes of a study my colleagues and I conducted to evaluate laser lens fragmentation patterns in laser-assisted cataract surgery (LACS) in terms of effective phaco time (EPT) and phaco power in different grades of cataract.
PATIENTS AND METHODS
Seventy-one eyes of 71 patients (27 male, 44 female) were included in the study. All patients were provided written informed consent in accordance with the Declaration of Helsinki, and institutional review board approval was obtained. The mean patient age was 60 ±6.66 years (range, 45–75 years), and all enrolled patients underwent standard preoperative examinations. Two groups were created based on the femtosecond laser-assisted lens fragmentation pattern used: In group 1, a quadrant fragmentation pattern was used (Figure 1A), and in group 2, a complete fragmentation pattern was used (Figure 1B).
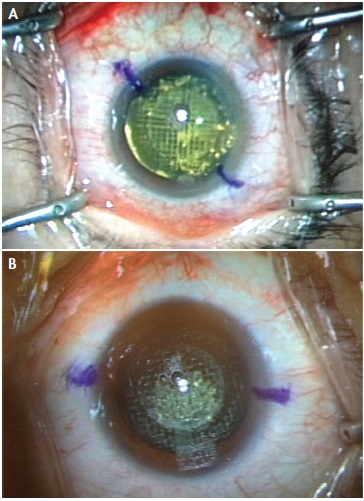
Figure 1. Fragmentation patterns used in LACS: the quadrant (A) and the complete pattern (B).
Each group was divided into subgroups according to the Emery-Little Classification System of nuclear opacity grade. Because all of the patients’ cataracts were either grade 1 or grade 2, each group included only those two subgroups. All LACS procedures were performed using the Catalys Precision Laser System (Abbott Medical Optics).5
SURGICAL TECHNIQUE
The surgery had a double setup procedure: the first part included the femtosecond laser portion of the case, and the second part included phacoemulsification. Laser capsulotomy and lens fragmentation were carried out using the Catalys. LACS was performed in one eye of each patient. After pupil dilation (phenylephrine 2.5% and tropicamide 0.5%) and corneal anesthesia (oxybuprocaine HCl, Conjucain EDO 0.4%; Bausch + Lomb), the patient was placed in a supine position in the operating chair. No eye speculum was used. After the patient was docked to the system, OCT was used to image the anterior chamber, and the system created a 3-D treatment plan. In all cases, the laser created a 5-mm diameter CCC (4 mJ pulse energy). The lens was segmented into quadrants and softened with a grid spacing of 350 mm (9 mJ pulse energy). After the laser procedure was completed, the patient was transferred to the operating table, where phacoemulsification was performed.
The manual cataract procedure was performed directly after laser treatment and measurements. A modified divide-and-conquer technique was employed using the Infiniti Ozil (Alcon). The softened nucleus was aspirated. Residual cortex removal and posterior capsular polishing were performed through the nasal and temporal incisions using bimanual I/A. After cortical removal, an Acriva Reviol (VSY Biotechnology) or AcrySof Restor (Alcon) multifocal IOL or a monofocal IOL (AcrySof; Alcon), depending on patient preference, was placed in the posterior chamber.
POSTOPERATIVE PROTOCOL
In all cases, the following postoperative medications were given: oral levofloxacin 500 mg once daily for 3 days, and topical diclofenac sodium 0.1%, dexamethasone sodium m-sulfobenzoate 0.1%, and moxifloxacin HCl 0.5% five times a day for 1 week.
After 1 week, diclofenac was reduced to four times a day for 1 month, and the last two medications were shifted to topical fluorometholone 0.1% and ofloxacin 0.3%, four times a day up to 1 month, before discontinuation.
Postoperative examinations were performed at 1 day, 1 week, and 1 and 3 months and included visual acuity and biomicroscopy.
RESULTS
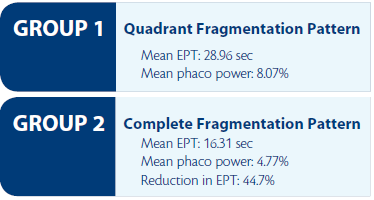
The mean EPT was 28.96 sec in group 1 and 16.31 sec in group 2. The mean phaco power was 8.07% in group 1 and 4.77% in group 2. Using the complete fragmentation pattern, there was a 44.7% reduction in EPT and a 40.9% reduction in phaco power compared with the quadrant pattern. The mean values of EPT and phaco power for each subgroup are shown in Figure 2. There was no significant difference in EPT between groups for the grade 1 cataract subgroup (P=.16), but there was a significant difference in phaco power (P=.03). Comparing the grade 2 subgroups between fragmentation patterns showed significant differences in EPT (P=.01) and phaco power (P=.003). Additionally, we performed a statistical analysis of the differences in change in distance UCVA, distance BCVA, spherical equivalent, and endothelial cell density between groups 1 and 2; no significant differences were found.
DISCUSSION
The excess energy created during phacoemulsification may cause capsular complications and damage to the corneal epithelium at the probe insertion site; thus, it is beneficial to try to limit ultrasound energy exposure.5,8
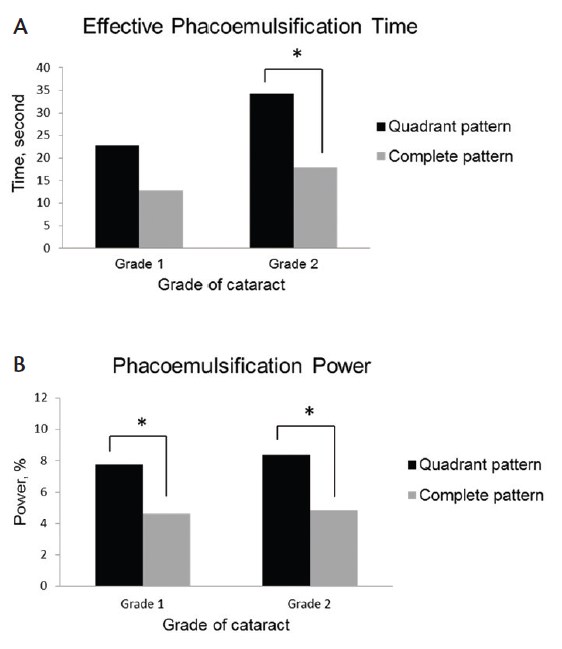
Figure 2. Mean values for EPT (A) and phaco power (B). * = statistically significant difference between patterns
Use of the femtosecond laser in LACS has been found to significantly reduce EPT and mean phaco energy.9-11 Initial results by Palanker et al12 indicated an approximate 40% reduction in ultrasound energy used during phacoemulsification (cumulative dissipated energy) with LACS compared with manual cataract surgery. Abell et al9 reported a 70% reduction in mean EPT with LACS compared with the manual technique. Conrad-Hengerer et al10 found a significant reduction (>96%) in EPT using the femtosecond laser compared with the manual technique. With growing user experience and the optimal use of various fragmentation patterns for different cataract grades, we anticipate further reduction in EPT with use of the femtosecond laser.
LACS is a rapidly developing procedure, but it is still in its infancy. Investigators are working on creating more efficient capsulotomy and lens fragmentation processes, including the use of optimal geometric fragmentation patterns and liquefaction to reduce the energy needed for phacoemulsification and to reduce SIA. Researchers are also working to develop better nomograms to increase the predictability of arcuate keratotomy. None of these elements of the procedure have been perfected.
The primary endpoint of our study was EPT, as reduced phaco time decreases the total number of joules of energy delivered to the eye and may lead to better preservation of ocular structures. Less energy delivered to the eye may also be associated with earlier improvement of postoperative visual acuity due to reduction of endothelial cell loss and corneal edema, with less anterior chamber cells and flare caused by alteration of the blood-aqueous barrier.
Phaco time has been shown to directly correlate with endothelial cell loss.14 Several studies have used phaco time or energy (power X time) to compare aspects of phaco procedures.14-19 Surgical trauma during phacoemulsification has been reported to injure corneal endothelial cells,13,20-22 and both ultrasound energy and irrigation directly injure the endothelium.13,20,21
In this study, we paid particular attention to the role of nucleus firmness, laser lens fragmentation patterns, and ultrasound energy in endothelial cell loss. The firmness of the nucleus was the most significant risk factor for endothelial injury. The size of the nucleus was also significantly associated with this risk. That is to say, the harder and larger the nucleus, the greater the degree of endothelial injury. Although the total ultrasound energy was proportional to the grade of the nucleus, ultrasound energy was not an independent predictor. Therefore, the characteristics of the nucleus itself were considered to predict degree of injury to the endothelial cells.
Our results showed no significant difference in endothelial cell loss (P=.77) between the two groups with different laser lens fragmentation patterns. Additionally, phaco time and average phaco power are known to have a positive correlation with nuclear opalescence.23 It has been shown that reductions in power and time may be seen when laser phacofragmentation is performed in conjunction with phacoemulsification.6
Further, we created subgroups based on the hardness of the cataract. There was a significant difference in phaco power and EPT between the two patterns in grade 2 cataracts. Based on the mean value, we may consider that the quadrant fragmentation pattern for grade 2 cataracts expends more phaco power and with a higher EPT. For grade 1 cataracts, the difference between the two patterns lies only in phaco power, not in EPT.
It is worth noting the laser interface suction used in our surgical technique. To prevent the eye from moving and to ensure precise results, the laser interface physically touches the eye and maintains its grasp by creating gentle suction. With the patient supine, the eye is stable in this position for about 40 seconds. However, a strong movement of the head or eye can break the suction. The procedure is then interrupted while the suction is replaced, or, in some situations, the case must be postponed or converted to standard cataract surgery.
The suction is mild, but it can cause some redness of the eye for the first week after cataract surgery. If the eye does not open widely, or if the white part of the eye is puffy, it may be difficult to get the laser into the correct position for surgery. In this case, the procedure must be postponed or done in a standard fashion. Corneal scars or folds may likewise block the path of the laser light, rendering the laser incapable of treating what lies beneath the obstacle. This could create a scenario in which that part of the surgery would have to be completed by hand.
At a Glance
- Excess energy generated in phacoemulsification may cause capsular complications and damage to the corneal epithelium at the probe insertion site.
- In several studies, use of the femtosecond laser has been found to significantly reduce effective phaco time and mean phaco energy.
- A smaller laser lens fragmentation pattern (complete) yielded significantly reduced phaco time and power compared with a larger fragmentation pattern
(quadrant) in a clinical study.
CONCLUSION
We have found the smaller of the two fragmentation patterns of the femtosecond laser that we studied (complete) to be more beneficial, with significantly reduced EPT and phaco power compared with the larger fragmentation pattern (quadrant). Further studies are needed to compare the advantages and disadvantages of each fragmentation pattern in relation to the size of the phaco tip, lumen, and laser system used. n
1. Nagy ZZ. Femtolaser cataract surgery: how to evaluate this technology, read the literature, and avoid possible complications. J Refract Surg. 2012;28:855-857.
2. Sugar A. Ultrafast (femtosecond) laser refractive surgery. Curr Opin Ophthalmol. 2002;13:246-249.
3. Gibson EA, Masihzadeh O, Lei TC, Ammar DA, Kahook MY. Multiphoton microscopy for ophthalmic imaging. J Ophthalmol. 2011;2011:870-879.
4. Uy HS, Edwards K, Curtis N. Femtosecond phacoemulsification: the business and the medicine. Curr Opin Ophthalmol. 2012;23:33-39.
5. He L, Sheehy K, Culbertson W. Femtosecond laser-assisted cataract surgery. Curr Opin Ophthalmol. 2011;22:43-52.
6. Nagy Z, Takacs A, Filkorn T, Sarayba M. Initial clinical evaluation of an intraocular femtosecond laser in cataract surgery. J Refract Surg. 2009;25:1053-1060.
7. Naranjo-Tackman R. How a femtosecond laser increases safety and precision in cataract surgery? Curr Opin Ophthalmol. 2011;22:53-57.
8. Ernest P, Rhem M, McDermott M, Lavery K, Sensoli A. Phacoemulsification conditions resulting in thermal wound injury. J Cataract Refract Surg. 2001;27:1829-1839.
9. Abell RG, Kerr NM, Vote BJ. Femtosecond laser-assisted cataract surgery compared with conventional cataract surgery. Clin Experiment Ophthalmol. 2013;41:455-462.
10. Conrad-Hengerer I, Hengerer FH, Schultz T, Dick HB. Effect of femtosecond laser fragmentation on effective phacoemulsification time in cataract surgery. J Refract Surg. 2012; 28:879-883.
11. Conrad-Hengerer I, Hengerer FH, Schultz T, Dick HB. Effect of femtosecond laser fragmentation of the nucleus with different softening grid sizes on effective phaco time in cataract surgery. J Cataract Refract Surg. 2012;38:1888-1894.
12. Palanker DV, Blumenkranz MS, Andersen D, et al. Femtosecond laser-assisted cataract surgery with integrated optical coherence tomography. Sci Transl Med. 2010;2:58ra85
13. Polack FM, Sugar A. The phacoemulsification procedure. II. Corneal endothelial changes. Invest Ophthalmol. 1976;15:458-469.
14. Zetterstrom C, Laurell CG. Comparison of endothelial cell loss and phacoemulsification energy during endocapsular phacoemulsification surgery. J Cataract Refract Surg. 1995;21:55-58.
15. Hayashi K, Nakao F, Hayashi F. Corneal endothelial cell loss after phacoemulsification using nuclear cracking procedures. J Cataract Refract Surg. 1994;20:44-47.
16. Brint SF, Blaydes JE, Bloomberg L, et al. Initial experience with the HydroSonics instrument to soften cataracts before phacoemulsification. J Cataract Refract Surg. 1992;18:130-135.
17. Lavery KT, McDermott ML, Ernest PH, et al. Endothelial cell loss after 4 mm cataract surgery. J Cataract Refract Surg. 1995;21:305-308.
18. Klemen UM. V-style phacoemulsification. J Cataract Refract Surg. 1993;19:548-550.
19. Pirazzoli G, D’Eliseo D, Ziosi M, Acciarri R. Effects of phacoemulsification time on the corneal endothelium using phacofracture and phaco chop techniques. J Cataract Refract Surg. 1996;22:967-969.
20. Dick HB, Kohnen T, Jacobi FK, Jacobi KW. Long-term endothelial cell loss following phacoemulsification through a temporal clear corneal incision. J Cataract Refract Surg. 1996;22:63-71.
21. Olson LE, Marshall J, Rice NS, Andrews R. Effects of ultrasound on the corneal endothelium: I. The acute lesion. Br J Ophthalmol. 1978;62:134-144.
22. Edelhauser HF, Van Horn DL, Hyndiuk RA, Schultz RO. Intraocular irrigating solutions; their effect on the corneal endothelium. Arch Ophthalmol. 1975;93:648-657.
23. Bencic G, Zoric-Geber M, Saric D, Corak M, Mandic Z. Clinical importance of the Lens Opacities Classification System III (LOCS III) in phacoemulsification. Coll Antropol. 2005;29(suppl 1):91-94.
Minoru Tomita, MD, PhD
- Medical Director, Tomita Minoru Eye Clinic Ginza, Chuoku Tokyo, Japan
- tomita@eyecanmedical.com
- Financial disclosure: Consultant (AcuFocus, Schwind eye-tech solutions)

