
The Galilei G6 system (Ziemer) fully meets the expectations of refractive surgery specialists. What makes this system attractive is its ability to combine all the technologies needed to screen patients for LASIK and to obtain optimal outcomes in cataract surgery. In refractive surgery, we use it to determine who is at risk for ectasia and to monitor keratoconus patients; in cataract surgery, we use it for optical biometry and total corneal power measurements as a means to optimize IOL selection. Additionally, the ability to perform highly reliable and repeatable measurements on the posterior surface, and for the system to achieve accurate pachymetry, corneal aberrations, and total corneal power data, helps us to further strengthen the safety of the decision-making process.
My clinical practice is mostly focused on LASIK procedures and refractive cataract surgery with premium IOLs. In that regard, two of the greatest advantages of the Galilei G6 are its robust refractive screening program and artificial intelligence tools, which help me rule out patients who are at risk for ectasia after surgery. The plethora and complexity of data provided by current imaging systems presents a challenge of interpretation for the ophthalmologist. Therefore, decisions are often based on personal experience and subjective recognition of patterns or empiric cutoff values that are not necessarily the same between imaging systems.
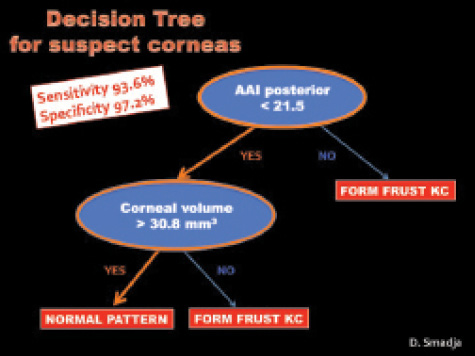
Figure 1. An AAI score of 21.5 is the most discriminant
parameter to differentiate between normal corneas and
subclinical keratoconus.
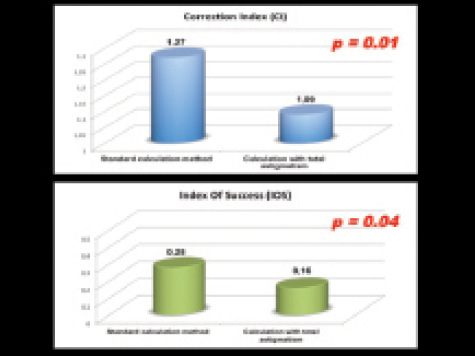
Figure 2. The author has reported a significant improvement
in astigmatism correction when the total corneal
astigmatism is used in place of keratometric astigmatism.
The Santhiago percentage of tissue altered (PTA) report1,2 on the Galilei G6 helps me to predict the level of risk a patient has of developing post-LASIK ectasia by taking into account the expected biomechanical alteration due to one's surgical plan. The PTA considers the relationship between corneal thickness, tissue alteration through ablation and flap creation, and residual stromal bed thickness. A PTA level of 40% can be considered a robust risk factor for ectasia, and, in our clinical practice, patients with a PTA this high are treated with PRK instead of LASIK, as long as the cornea does not show any other contraindications for refractive surgery.
The Galilei G6 also offers a more morphologic approach to identifying subclinical keratoconus at its earliest stages. Using an automated decision tree, the system is able to identify topographically normal contralateral eyes of patients with frank keratoconus with 93.6% sensitivity and 97.2% specificity (Figure 1).3,4 While waiting for the upcoming release of this feature, our group has pointed out that an Asphericity Asymmetry Index (AAI) of 21.5 is the most discriminant parameter to differentiate between normal corneas and subclinical keratoconus. This index measures the asymmetry of the posterior corneal surface; we have found that patients with an AAI greater than 21.5 or a corneal volume lower than 30.8 mm3 are not recommended for LASIK.
The combination of a morphologic approach (decision tree) and a biomechanical approach (PTA report) is a unique feature in ocular biometry that can increase the sensitivity of detection of corneas at risk for ectasia.
The other key feature that we routinely use is the ability to measure posterior astigmatism and total corneal astigmatism in the calculation of toric IOL power. We recently reported a significant improvement in astigmatism correction when the total corneal astigmatism is used in place of keratometric astigmatism (Figure 2).5 Like us, others have also noted the reliability and repeatability of posterior astigmatism and total corneal power measurements in toric IOL calculation.6,7
For a refractive, cataract, and corneal surgery specialist, the Galilei G6 diagnostic system allows the most extensive analysis of corneal features that exists today in a single device. It combines Placido-disc–based corneal topography, posterior corneal surface analysis, pachymetry report including corneal volume, corneal aberrations analysis, total corneal power measurements, densitometry, and optical biometry for optimal IOL selection. Additionally, the development and introduction of artificial-intelligence–based programs for screening ectasia-susceptible corneas, and especially the synergistic combination of morphologic and biomechanical approaches, can assist refractive surgeons in their decision-making process without relying only on subjective interpretation and personal expertise. Furthermore, technically speaking, the level of accuracy and repeatability of the parameters measured with the Galilei G6, which have been demonstrated in the literature, helps surgeons to comfortably rely on these measurements.
David Smadja, MD
• Anterior Segment Unit, Ophthalmology Department, Tel Aviv
Sourasky Medical Center, Israel
• Institute of Nanotechnology and Advanced Materials, Bar Ilan
University, Tel Aviv, Israel
• National Reference Center for Keratoconus, Bordeaux Hospital
University, France
• davidsmadj@hotmail.fr
• Financial disclosure: Consultant (Ziemer, Alcon)




