

Epidemiologic studies estimate that the prevalence of dry eye disease (DED) is as much as 25% in the general population.1,2 Significantly associated with aging, DED occurs most frequently in those over the age of 45 years.3 It is more prevalent in women compared with men.4-6 Contributing factors to DED may be classified as ocular, medical, pharmaceutical, iatrogenic, environmental, and contact–lens-related.7
A CHALLENGING DIAGNOSIS
DED is a common clinical problem for eye care providers worldwide. The importance of proper and timely distinction between healthy and affected eyes is beyond doubt.8-10
Current options in clinical investigation include slit-lamp observation, tear film stability assessment (invasive and noninvasive tear breakup time [TBUT] measurement and tear film interferometry),11 tear secretion assessment tests (Schirmer with or without anesthesia and thread methods), tear clearance assessment (fluorescein clearance test, tear function index, and fluorophotometry),12 ocular surface damage assessment (corneal and conjunctival rose bengal and lissamine green staining and cytology),13 lipid layer assessment (precorneal and meibomian gland grading),14,15 tear osmolarity,16,17 and subjective symptom questionnaires.4,18
Among these investigative techniques, some, such as Schirmer and TBUT tests, can show bias due to examiner subjectivity,19 influence of external stimuli,20 and difficulty of accurate result documentation.21 The same is true for investigative techniques based on patient-reported symptoms or questionnaires.22-24 Additionally, evidence suggests that clinical dry eye symptoms alone may be insufficient for proper diagnosis of the disease.25,26
EPITHELIAL THICKNESS MAPPING
One novel objective investigative technique for dry eye screening is corneal epithelial thickness mapping with anterior segment OCT (AS-OCT).27 We have found that epithelial thickening may be an alarming indication for corneal abnormality—specifically, overall epithelial thickness may reflect conditions such as moderate or subclinical dry eye.
In a recent analysis, we found that screening with AS-OCT was a highly repeatable, quantitative, accurate, and easy-to-document procedure.27 This comparative study of 70 women with normal or dry eyes was designed to assess the functionality of DED diagnosis with AS-OCT. We chose to enroll women only because, in our practice, they compose most of the dry eye population, with a ratio of 10:1 to men (unpublished data). The findings reported herein may also be applicable to the screening of refractive surgery candidates and to the assessment of postoperative iatrogenically induced DED.
Patients in the control group (n=35; group A) had normal eyes that were previously unoperated, with no ocular pathologies other than refractive error and no dry eye conditions as confirmed by a complete ocular clinical evaluation. Patients in the dry eye group (n=35; group B) had clinically confirmed dry eye but were unoperated and had no other ocular pathologies other than refractive error. Exclusion criteria were anterior basement membrane dystrophy, other corneal dystrophies, and rheumatic diseases. No patient who reported previous use of contact lenses or recent use of artificial tears was enrolled in either group.
At a Glance
• Epithelial thickening may be an indication of corneal abnormality—specifically, overall epithelial thickness may reflect conditions such as moderate or subclinical dry eye.
• AS-OCT may provide a repeatable, quantitative, accurate, and easy-to-document procedure for dry eye screening.
• Although average epithelial thickness as assessed by AS-OCT can be used as an indicator of DED, one must be aware that the false-positive and missed diagnosis rates could be as high as 15%.
Dry eye was diagnosed with TBUT measurement (DED considered if less than 5 seconds) and Schirmer test (DED considered if less than 5 mm). Additionally, AS-OCT with the RtVue-100 (Optovue) was acquired with the device’s L-Cam lens; eight meridional B-scans were taken per acquisition, each consisting of 1,024 A-scans with axial resolution of 5 μm. Following correct fixation and centering, acquisition time was on the order of a few seconds per scan. Four individual acquisitions were performed in each case on the same day, and all measurements were obtained by the same investigator prior to TBUT and Schirmer testing.
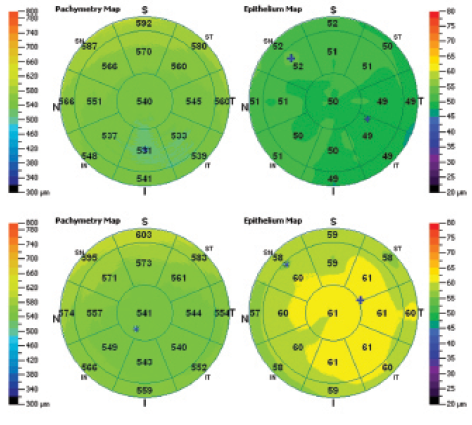
Figure 1. Representative corneal total thickness maps (left) and corneal epithelium thickness maps (right) of a normal patient from group A (top) and a dry eye patient from group B (bottom), as provided by the AS-OCT system report.
For each eye, we measured and analyzed the average, superior, and inferior epithelial thickness within the central 5-mm zone as well as the topographic thickness variability as calculated from the standard deviation of 17 local thickness measurements (Figure 1). The average epithelial thickness was computed for each eye within the 5-mm zone as the average of the 17 local thickness measurements.
We found an overall greater epithelial thickness in group B as compared with group A, with a statistically significant difference in epithelial thickness between the groups. For central thickness, the mean difference between dry and normal eyes was 6.5 μm; for average thickness, the difference was 6.2 μm. All tests of respective epithelial thickness metrics between groups A and B showed statistically significant differences (P<.05).
From these findings, we concluded that average epithelial thickness can be used as an indicator of DED. However, one must be cautious with use of this medium, as the false-positive rate and missed diagnosis rate were approximately 15%. The findings reported above may also be applicable to the screening of refractive surgery candidates and to the assessment of postoperative iatrogenically induced DED.
PREVIOUS INVESTIGATION
In a previous investigation of 3-D epithelial thickness in keratoconic eyes, we identified an overall thicker epithelium that might be a result of a reactive process. In short, the epithelium appeared to thicken in less rigid corneas, as it was more susceptible to mechanical variations produced by one or multiple factors including eye rubbing and increased blinking.28 The difference between DED and keratoconic eyes is epithelial thickness: In DED patients, there is a near-normal topographic distribution;29 however, the distribution is highly disturbed in keratoconic patients. On AS-OCT, this is reflected by the standard deviation of epithelial thickness, where normal is anything less than 3 µm.
Although AS-OCT has the advantages of in vivo, noncontact application and speed of optical imaging,30 until recently its application in epithelial thickness imaging involved either investigator-modified software or hardware31-33 or caliper software measurement techniques.29,34 The RtVue-100 is a Fourier-domain AS-OCT system that incorporates epithelial thickness map analysis, currently extending up to a 6-mm diameter. The system’s software automatically identifies the air-tear film and epithelium-Bowman layer interfaces and produces total corneal and corneal epithelial thickness pachymetry maps, making it a potential tool for dry eye assessment.
CONCLUSION
One may wonder how a time-intensive procedure such as epithelial thickness assessment with AS-OCT can be clinically viable as a detector of DED when other methods, such as TBUT and Schirmer testing, take only a few seconds. However, we believe that the clinical insights offered by epithelial thickness mapping will make it worthwhile for use in routine screening and treatment assessment. It may even supersede other specific dry eye measurements that may or may not be part of one’s screening protocol.
The anticipated clinical ramifications of epithelial thickness mapping by means of AS-OCT are positive. Because this proposed screening indicator is based on a commercially available instrument that can be integrated into daily clinical practice, we believe it has potential value in the diagnosis of DED.
The clinical relevance of these findings, for us, is the fact that we should no longer assess refractive surgery and/or refractive cataract surgery patients without correlating the refraction to topographic maps along with epithelial maps, both pre- and
postoperatively. n
1. McCarty CA, Bansal AK, Livingston PM, et al. The epidemiology of dry eye in Melbourne, Australia. Ophthalmology. 1998;105(6):1114-1149.
2. Schaumberg DA, Sullivan DA, Dana MR. Epidemiology of dry eye syndrome. Adv Exp Biol Med. 2002;506(Pt B):989-998.
3. Viso E, Rodriguez-Ares MT, Gude F. Prevalence of and associated factors for dry eye in a Spanish adult population (the Salnes Eye Study). Ophthalmic Epidemiol. 2009;16(1):15-21.
4. Gonen T, Celik C, Oznur M, et al. Tear osmolarity and ocular surface changes in patient with polycystic ovary syndrome. Curr Eye Res. 2013;38(6):621-625.
5. Chen SP, Massaro-Giordano G, Pistilli M, et al. Tear osmolarity and dry eye symptoms in women using oral contraception and contact lenses. Cornea. 2013;32(4):423-428.
6. Schaumberg DA, Sullivan DA, Buring JE, Dana MR. Prevalence of dry eye syndrome among US women. Am J Ophthalmol. 2003;136(2):318-326.
7. Doughty MJ, Fonn D, Richter D, et.al. A patient questionnaire approach to estimating the prevalence of dry eye symptoms in patients presenting to optometric practices across Canada. Optom Vis Sci. 1997;74(8):624-631.
8. Williamson JF, Huynh K, Weaver MA, Davis RM. Perceptions of dry eye disease management in current clinical practice. Eye Contact Lens. 2014;40:111-115.
9. Nichols KK, Nichols JJ, Zadnik K. Frequency of dry eye diagnostic test procedures used in various modes of ophthalmic practice. Cornea. 2000;19:477-482.
10. van Tilborg MM, Murphy PJ, Evans KS. Agreement in dry eye management between optometrists and general practitioners in primary health care in the Netherlands [published online ahead of print April 18, 2015]. Cont Lens Anterior Eye.doi: 10.1016/j.clae.2015.03.005.
11. Goto T, Zheng X, Klyce SD, et al. A new method for tear film stability analysis using videokeratography. Am J Ophthalmol. 2003;135(5):607-612.
12. Afonso AA, Monroy D, Stern ME, et al. Correlation of tear fluorescein clearance and Schirmer test scores with ocular irritation symptoms. Ophthalmology. 1999;106:803-810.
13. Brignole F, Ott AC, Warnet JM, et al. Flow cytometry in conjunctival impression cytology: a new tool for exploring ocular surface pathologies. Exp Eye Res. 2004;78:473-481.
14. Yanga Z, San CTL, Tong L. Intra-observer and inter-observer repeatability of ocular surface interferometer in measuring lipid layer thickness. BMC Ophthalmology [in press].
15. Robin JB, Jester JV, Nobe J, et al. In vivo transillumination biomicroscopy and photography of meibomian gland dysfunction. Ophthalmology. 1985;92:1423-1426.
16. Tomlinson A, McCann LC, Pearce EI. Comparison of human tear film osmolarity measured by electrical impedance and freezing point depression techniques. Cornea. 2010;29(9):1036-1041.
17. Bunya VY, Fuerst NM, Pistilli M, et al. Variability of tear osmolarity in patients with dry eye [published online ahead of print March 26, 2015]. JAMA Ophthalmol. doi: 10.1001/jamaophthalmol.2015.0429.
18. Alves M, Reinach PS, Paula JS, Vellasco e Cruz AA. Comparison of diagnostic tests in distinct well-defined conditions related to dry eye disease. PLoS One. 2014;9(5):e97921.
19. Cho P, Yap M. Schirmer test: a review. Optom Vis Sci. 1993;70(2):152-156.
20. Yokoi N, Komuro A. Non-invasive methods of assessing the tear film. Experiment Eye Res. 2004;78:399-407.
21. Foulks GN. Challenges and pitfalls in clinical trials of treatments for dry eye. Ocul Surf. 2003;1:20-30.
22. Simpson TL, Situ P, Jones LW, Fonn D. Dry eye symptoms assessed by four questionnaires. Optom Vis Sci. 2008;85(8):692-629.
23. Dogru M, Nakamura M, Shimazaki J, Tsubota K. Changing trends in the treatment of dry-eye disease. Expert Opin Investig Drugs. 2013;22(12):1581-1601.
24. Schiffman RM, Christianson MD, Jacobsen G, et al. Reliability and validity of the Ocular Surface Disease Index. Arch Ophthalmol. 2000;118(5):615-621.
25. Begley CG, Chalmers RL, Abetz L, et al. The relationship between habitual patient-reported symptoms and clinical signs among patients with dry eye of varying severity. Invest Ophthalmol Vis Sci. 2003;44(11):4753-4761.
26. Sullivan BD, Crews LA, Messmer EM, et al. Correlations between commonly used objective signs and symptoms for the diagnosis of dry eye disease: clinical implications. Acta Ophthalmol. 2014;92(2):161-166.
27. Kanellopoulos AJ, Asimellis G. In vivo 3-dimensional corneal epithelial thickness mapping as an indicator of dry eye: Preliminary clinical assessment. Am J Ophthalmol. 2014;157(1):63-68.e2
28. Rodriguez JD, Ousler GW, Johnston PR, et al. Investigation of extended blinks and interblink intervals in subjects with and without dry eye. Clin Ophthalmol. 2013;7:337-342.
29. Tao A, Shao Y, Jiang H, et al. Entire thickness profiles of the epithelium and contact lens in vivo imaged with high-speed and high-resolution optical coherence tomography. Eye Contact Lens. 2013;39(5):329-334.
30. Li Y, Tan O, Brass R, et al. Corneal epithelial thickness mapping by Fourier-domain optical coherence tomography in normal and keratoconic eyes. Ophthalmology. 2012;119(12):2425-2433.
31. Wirbelauer C, Pharo DT. Monitoring corneal structures with slitlamp-adapted optical coherence tomography in laser in situ keratomileusis. J Cataract Refract Surg. 2004;30(9):1851-1860.
32. Haque S, Simpson T, Jones L. Corneal and epithelial thickness in keratoconus: a comparison of ultrasonic pachymetry, Orbscan II, and optical coherence tomography. J Refract Surg. 2006;22(5):486-493.
33. Francoz M, Karamoko I, Baudouin C, Labbé A. Ocular surface epithelial thickness evaluation with spectral-domain optical coherence tomography. Invest Ophthalmol Vis Sci. 2011;52(12):9116-9123.
34. Rocha KM, Perez-Straziota E, Stulting RD, Randleman JB. SD-OCT analysis of regional epithelial thickness profiles in keratoconus, postoperative corneal ectasia, and normal eyes. J Refract Surg. 2013;29(3):173-179.
George Asimellis, PhD
• Laservision.gr Clinical and Research Eye Institute, Athens, Greece
• asimellis@laservision.gr
• Financial disclosure: None
A. John Kanellopoulos, MD
• Clinical Professor of Ophthalmology, NYU Medical School, New York
• Laservision.gr Clinical and Research Eye Institute, Athens, Greece
• ajk@brilliantvision.com
• Financial disclosure: Consultant (Alcon/WaveLight, Allergan, Avedro, i-Optics, Keramed)

