
This noncontact meibography device allows infrared visualization of meibomian glands without a fiber optic probe. Instead, it uses a background illumination device and an infrared filter to illuminate the meibomiam glands and allows the clinician to assess their integrity. According to company literature,1 images can be displayed on a computer screen, digitally captured, and archived, and the device can be mounted on top of Topcon slit lamp models SL-D7 and SL-D8. Key features include:
• One-touch changeover switch allowing visualization of the meibomian glands;
• Capture of full upper and lower meibomian glands in one shot; and
• Easy attachment to the slit lamp.
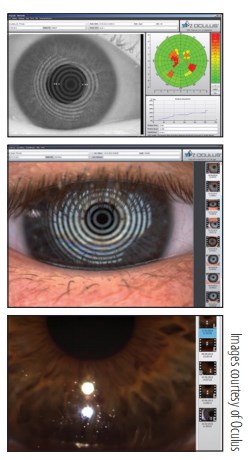
This corneal topographer has a built-in keratometer and color camera that can be used to examine the meibomian glands, lipid layer, tear breakup time (TBUT), and tear meniscus height. According to company literature,1 the system’s infrared illumination ring—using white diodes for the tear film dynamics, blue diodes for fluorescein images, and infrared diodes for meibography—provides adequate analysis of the tear film. Key software components include:
• Tear film scan to noninvasively and automatically assess the lipid layer and tear film dynamics, including TBUT;
• Meniscus tear height assessment of tear film quantity with an integrated ruler and magnification options;
• Lipid layer assessment of the interference phenomenon, whereby the thickness of the lipid layer is assessed based on its structure and color; and
• Tear film dynamics assessment of the particle flow and viscosity of the tear film with video recording (up to 32 frames per second).

This point-of-care test detects all known serotypes of adenoviral conjunctivitis. According to company literature,1 the test, designed for rapid, accurate diagnosis, can help physicians isolate and manage contagious patients and prevent the spread of infection. It can also help reduce unnecessary antibiotic prescriptions and the potential for ocular allergies and toxicities associated with antibiotic use.

This in-office test is designed to identify elevated levels of the inflammatory marker matrix metallopeptidase 9, which is typically elevated in the tears of patients with dry eye disease (DED). After a tear fluid sample is taken from the inside lining of the palpebral conjunctiva, results are achieved in about 10 minutes, according to company literature.1
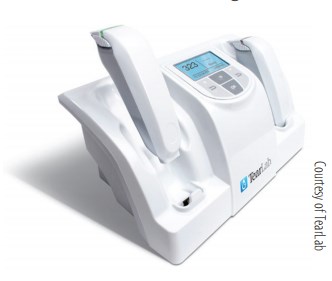
Intended to indirectly assess the osmolarity of human tears as a means to diagnose DED, this objective test provides results in seconds using a 50-nL tear film sample collected from the eyelid margin. According to company literature,1 the system uses a temperature-corrected impedance measurement, applies a lot-specific calibration curve, and calculates and displays the osmolarity measurement as a quantitative numerical value. Company literature also states that incorporating osmolarity testing into one’s practice can give the best indication of early stage DED.

This noncontact ocular surface interferometer is designed to assess the tear film by measuring the absolute thickness of the tear film lipid layer. According to company literature,1 the device captures, archives, manipulates, and stores digital images of specular observations of the tear film in about 5 minutes. The company’s Meibomian Gland Evaluator is a complementary technology that allows examination of the quantity and quality of lipids released from the meibomian glands.

Both units provide quantitative results of the company’s lactoferrin and IgE tear diagnostic tests, which can help physicians to diagnose DED and ocular allergies. According to company literature,1 after each test, a report is automatically generated for lab records or patient charts. These can also be directly linked to electronic health record systems.
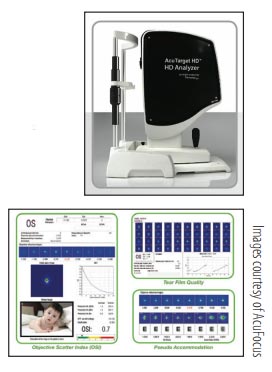
This diagnostics tool is designed to optimize clinical outcomes by identifying factors that may affect visual outcomes. According to company literature,1 the device can evaluate the quality of vision, including objective scatter index; assess tear film quality over time; and measure pseudoaccommodation to visually demonstrate depth of focus. Postoperatively, AcuTarget HD can be used to determine if patients are compliant with their dry eye drop
regimens.









