Dry eye symptoms and related epitheliopathy occur commonly after traditional refractive surgery techniques. The manifestations most often reported are ocular dryness, which is seen in almost 40% of eyes treated with LASIK and PRK, followed by nonspecific ocular surface discomfort and sharp pain on waking.1
LASIK remains the gold standard refractive surgical procedure, with good visual outcomes and short postoperative recovery time demonstrated in numerous investigations. However, changes in corneal shape, tear film dynamics, and subepithelial innervation after surgery may play a role in the onset of ocular discomfort syndrome. The impact of these factors may be detrimental to visual results, with fluctuation of vision quality and decrease of BCVA.2
Key points to consider in this phenomenon are that different patients may respond with a variable spectrum of symptoms to identical surgical stimuli and that preexisting dry eye before surgery can be a risk factor for developing severe ocular discomfort after refractive surgery. All types of keratorefractive surgery damage the integrity of corneal nerve fibers, therefore influencing the reflex arc that regulates tear film dynamics.
EXCIMER ABLATION AND CORNEAL NERVES
It is well known that excimer–laser-based procedures result in sudden central corneal nerve fiber damage; this is related to the flap cut in LASIK and to the laser photoablation of the stroma containing nerve fibers in LASIK and PRK.
Central corneal subepithelial nerve wound healing has been reported to be only slightly different between LASIK and similar flap-based procedures regarding the time to recovery and morphologic appearance of the regenerated nerve plexus. At 1 month after surgery, the central cornea appears devoid of nerve fibers in conventional LASIK, sub-Bowman keratomileusis (SBK), and femtosecond LASIK (femto-LASIK).3 The process of reinnervation begins in the peripheral region of the flap with the formation of thin nonbranching fibers crossing the sidecut. Nerve morphology changes rapidly throughout the following months, reaching the central 3-mm zone by 6 months.3 Despite this rapid growth, the density and morphology of the nerves remain altered for years after surgery, probably explaining some clinical conditions such as dryness and recurrent superficial epithelial punctate erosions.
Flapless surface excimer techniques have faster nerve recovery times, with a mean time to restoration of 2 years compared with 5 years for flap-based procedures.4 Corneal sensitivity follows a pattern similar to that for flap-based procedures, decreasing in the first 3 months and returning to normal values by 6 months after surgery.5 Some authors have reported persistent incomplete normalization out to
1 year and beyond.6
The LASIK procedure has been associated with worse results in terms of corneal sensitivity, tear breakup time, Schirmer test values, and corneal reinnervation compared with LASEK and PRK procedures at 6 months. Corneal reinnervation was highly correlated with recovery of corneal sensitivity; the nerve-healing pattern in LASEK was similar to that of PRK.7
At a Glance
• All types of keratorefractive surgery damage the integrity of corneal nerve fibers, therefore influencing the reflex arc that regulates tear film dynamics.
• Despite rapid reinnervation after LASIK and surface ablation procedures, the density and morphology of the nerves remain altered for years after surgery, probably explaining some clinical conditions such as dryness and recurrent superficial epithelial punctate erosions.
• Sparing the superficial subbasal nerve plexus, as the SMILE procedure does, may play a role in maintaining partial corneal innervation and may also be responsible for the rapid nerve regrowth observed in the early months after surgery.
The risk of developing dry eye disease (DED) postoperatively appears to be correlated with the degree of preoperative myopia and the depth of laser ablation and stromal dissection; moreover, in patients affected by DED, the risk of myopic regression is increased.8,9
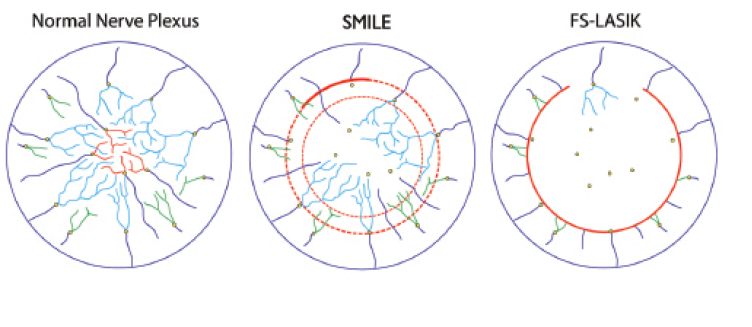
Figure 1. Stromal nerve fiber bundles run centripetally and toward the surface, perforating the Bowman layer (spots indicated by yellow circles). Once the fibers penetrate the Bowman layer, the subbasal nerve plexus is originated. Different colors illustrate the subbasal fibers originating from central (red), paracentral (light blue), and peripheral (green) perforating stromal fibers, respectively (A). With the SMILE procedure, in the absence of a full flap sidecut, peripheral nerve fibers are resected only where the 50° arc of the incision is placed (thick red line). Moreover, fibers are resected if they rise superficially to perforate the Bowman layer within the area of the created and extracted refractive lenticule. The other fibers that had penetrated the Bowman layer outside the lenticule area may run undisturbed as subbasal nerve plexus. (For simplicity, central subbasal surviving fibers are not depicted in the central zone; B). In the femto-LASIK (FS-LASIK) procedure, all fibers are cut throughout the extension of the 300º to 310° degree of arc flap sidecut; all deeper fibers are disrupted within the photoablation area (C).
FEMTOSECOND FLAP CREATION
The introduction of the femtosecond laser in corneal refractive surgery improved the reliability of LASIK procedures by increasing the predictability of flap parameters, facilitating planar configuration of flap-cut geometry, providing more predictable depth of dissection, and reducing the incidence of intraoperative complications.10
Despite these improvements, the corneal nerve plexus is still affected by severe damage for a long time after femto-LASIK. Flap creation, by whatever means, implies the transection of all nerve fibers at the lamellar border. Despite the better alignment of flap margins achieved with femto-LASIK, the reinnervation process is similar between LASIK with a mechanical microkeratome and with a femtosecond laser. Central nerve fiber density is comparable in the two techniques at every follow-up time.10 Similarly, the incidence of DED symptoms after surgery was found to be similar in both procedures. Therefore, the planar configuration of thin femto-LASIK flaps does not seem to be associated with clinically significant advantages in terms of induced neurotrophic epitheliopathy.
SMILE ANd CORNEAL NERVE FIBERS
In ReLEx small incision lenticule extraction (SMILE), two intrastromal dissection planes are produced by a femtosecond laser: the first forms the posterior face of the lenticule, and the second creates the anterior face and enlarges to form a side pocket dissection plane for later extraction (the edge of the so-called cap). The procedure takes place without affecting the superficial tissue, fashioning an intrastromal disc that can be extracted through the 40° to 50° single superficial incision.
To understand the nerve fiber resection induced by SMILE and other refractive surgical procedures, it is necessary to appreciate the intrastromal distribution of the nerve fibers that form the subbasal nerve plexus and the topography of the emerging nerve fibers that penetrate the Bowman membrane and, thereafter, run superficially. The anatomy of corneal nerves has been recently revisited by Al-Aqaba and colleagues,11 who described the presence of about 40 stromal nerves—coming from the suprachoroidal space—that penetrate the peripheral cornea, coursing toward the center in a radial manner. These nerves penetrate the Bowman layer at various locations, mostly in the midperipheral zone (outside the central 8 mm), creating the subbasal plexus. The mean number of piercing fibers ranges from 30 in the central zone to 160 in the midperiphery. A few fibers enter the subbasal plexus in the paralimbal region.11
Figure 1A presents a schematic illustration of the nerve fiber pathway, which runs centripetally. It is reasonable to hypothesize that, when a flap-based technique is performed, all nerve fibers that run within the circumference of the flap sidecut are resected (ie, the vast majority of the fibers). Only the few fibers running in the region of the flap hinge may survive untouched within the lifted flap as subbasal fibers (Figure 1C).
This hypothesis correlates well with clinical and confocal microscopy results reported in the literature,2-4 showing a dramatic reduction of the central subbasal corneal nerve plexus after LASIK. By contrast, when the SMILE procedure is performed, it is presumed that a certain number of fibers are resected by the sidecut incision and because of the interruption of fibers perforating the Bowman zone inside the refractive lenticule and cap area. Conversely, fibers that rose to the superficial subepithelial location after perforating the Bowman layer in areas located outside the lenticule and cap area may run undisturbed over the refractive zone without interruption, with the exception of those interrupted by the sidecut incision (Figure 1B).
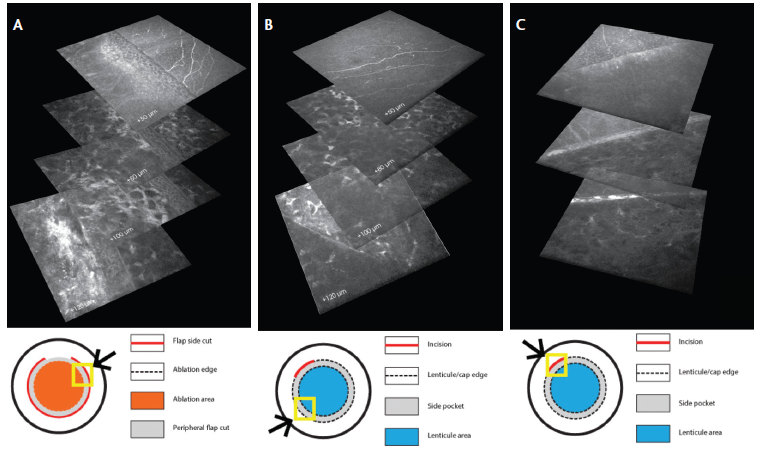
Figure 2. Sidecut IVCM stack reconstruction: In femto-LASIK, all fibers are resected throughout the extension (approximately 300°) of the flap sidecut (top panel shows resected fibers at the flap border 1 week after surgery; underlying panels show deeper stromal layers; A). In SMILE, untouched superficial fibers are observed (top panel) running over the lenticule stromal edge (bottom panel; B) The small incision represents a zone of superficial nerve fiber resection in the SMILE technique (top panel; C).
EVIDENCE OF CORNEAL NERVE FIBER SPARING
Few investigations have, to the best of our knowledge, evaluated the corneal innervation and corneal sensitivity after SMILE. Wei et al12 reported that corneal sensitivity was significantly better after SMILE than after LASIK. Moreover, they observed similar corneal sensitivity scores preoperatively and at 3 months after SMILE, suggesting that the remarkable changes of corneal sensitivity typical after femto-LASIK are not common after SMILE.
Vestergaard et al investigated corneal sensitivity and used confocal microscopy to compare the subbasal nerve plexus morphology in two groups of patients that underwent femtosecond laser refractive surgery: One group underwent the SMILE procedure, and the other underwent ReLEx FLEX, which included creation and lifting of a complete flap.13 These authors reported better corneal sensitivity and a significantly higher density of central corneal nerve fibers, as observed by confocal microscopy, in the SMILE group compared with the FLEX group at the single 6-month follow-up time point. The study authors noted the importance of a flapless procedure to support rapid nerve restoration; however, they did not include follow-up data from the early postoperative period, and no comparison to the LASIK technique was included. Comparing SMILE- and femto-LASIK–treated eyes, Li et al reported better preservation of nerve morphology and density with SMILE.14
We have used laser scanning in vivo confocal microscopy (IVCM) to examine the induced alterations and corneal nerve wound healing patterns in eyes after femto-LASIK and SMILE for similar degrees of myopic correction. We observed remarkable differences between the two techniques using this method of analysis.15
In studying the integrity of the peripheral nerve fibers (in the areas corresponding to the flap sidecut in LASIK and the lenticule-cap area in SMILE), it was observed that all fibers running centripetally are resected by the presence of a sidecut (Figure 2). In femto-LASIK, the fibers are circumferentially interrupted throughout the extension of the flap cut at the time of surgery. The excimer ablation of the central stroma may also damage deeper fibers (Figure 2A). In the SMILE procedure, nonresected peripheral fibers may be observed running centripetally in the area overlying the edge of the lenticular lamellar cut (Figure 2B). Similar to LASIK, the fibers are also resected where the sidecut incision is placed in SMILE (Figure 2C).
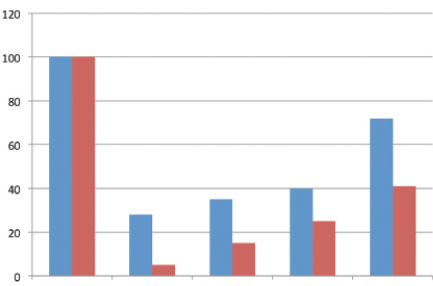
Figure 3. Percentage changes over time in nerve fiber density in SMILE (blue) and femto-LASIK (red).
The combination of peripheral transection of the nerve fibers and central stromal ablation explains the fact that, starting 1 week after surgery, the density of central nerve fibers is markedly reduced (by up to 95% of preoperative values) in LASIK (Figure 3). A period of several months is generally necessary to observe a partial recovery of fiber density. By contrast, in the SMILE technique, the reduction of nerve fiber density is significantly less that in the LASIK group at each follow-up time point. Central reinnervation was found to be faster with SMILE than with LASIK. Approximately 30% of central fibers appeared to be spared by SMILE. Central nerve fibers are detectable at 1 week after SMILE (Figure 4), and a rapid gain of central nerve fiber density was observed.
The main reason for nerve sparing in SMILE can be assumed to be the absence of the broad LASIK sidecut that transects all edge-crossing fibers. The sparing of the superficial subbasal nerve plexus may play a role in maintaining partial corneal innervation in the early postoperative period after SMILE and may be also responsible for the rapid nerve regrowth (Figure 3).
CONCLUSION
In vivo findings suggest that there is significantly less surgical denervation in SMILE than in femto-LASIK. Significantly faster nerve regeneration also seems to occur after SMILE. The mechanisms of these findings are related to the neuroanatomy of the cornea described above and to the flapless nature of the SMILE technique. The paths of some nerve fibers intersect with the lenticule-cap plane and become transected by the femtosecond laser cut. These fibers appear to degenerate during the first weeks after surgery, reducing the overall central corneal nerve fiber density in SMILE patients. However, this process is significantly reduced in SMILE in comparison with femto-LASIK, in which nerve fiber resection is almost total. After SMILE, some of the spared stromal fibers run over the lenticule plane in the midperiphery, piercing the Bowman layer just inside the area of treatment, remaining intact and continuing to participate in the subbasal central plexus.
These findings suggest that SMILE favors better preservation of the corneal neural architecture and greater postoperative corneal sensitivity compared with a flap-based technique such as LASIK. This favorable condition is likely to positively affect the incidence and course of postoperative ocular surface symptoms and dry-eye related epitheliopathy in treated patients. n
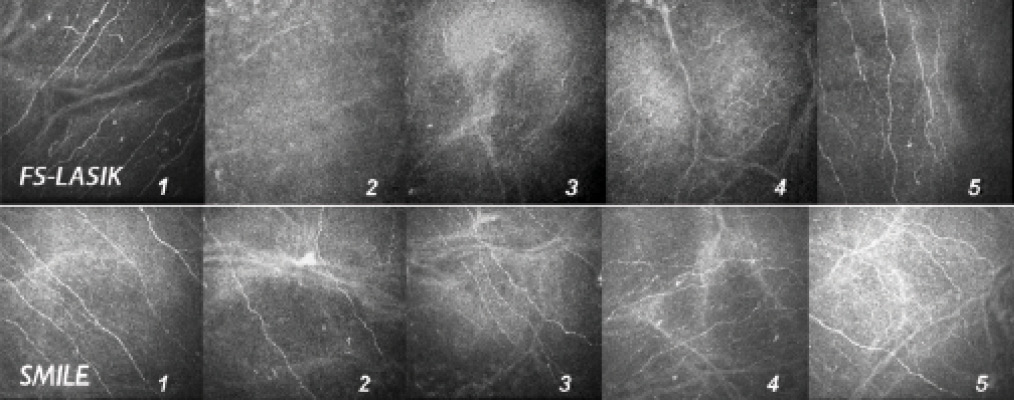
Figure 4. In vivo confocal microscopy images of the central subbasal nerve plexus before and after femto-LASIK (FS-LASIK; top row) and SMILE (bottom row). 1 = preoperative; 2 = 1 week; 3 = 1 month; 4 = 3 months; 5 = 6 months. Note that viable fibers are visible at 1 week and 1 month after surgery in the SMILE group; partial recovery of nerve fiber density starts 3 months after LASIK.
1. Hovanesian JA, Shah SS, Maloney RK. Symptoms of dry eye and recurrent erosion syndrome after refractive surgery. J Cataract Refract Surg. 2001;27(4):577-584.
2. Ambrósio R Jr, Tervo T, Wilson SE. LASIK-associated dry eye and neurotrophic epitheliopathy: pathophysiology and strategies for prevention and treatment. J Refract Surg. 2008;24(4):396-407.
3. Zhang F, Deng S, Guo N, Wang M, Sun X. Confocal comparison of corneal nerve regeneration and keratocyte reaction between FS-LASIK, OUP-SBK, and conventional LASIK. Invest Ophthalmol Vis Sci. 2012;53(9):5536-5544.
4. Erie JC, McLaren JW, Hodge DO, Bourne WM. Recovery of corneal subbasal nerve density after PRK and LASIK. Am J Ophthalmol. 2005;140(6):1059-1064.
5. Pérez-Santonja JJ, Sakla HF, Cardona C, Chipont E, Alió JL. Corneal sensitivity after photorefractive keratectomy and laser in situ keratomileusis for low myopia. Am J Ophthalmol. 1999;127(5):497-504.
6. Murphy PJ, Corbett MC, O’Brart DP, Verma S, Patel S, Marshall J. Loss and recovery of corneal sensitivity following photorefractive keratectomy for myopia. J Refract Surg. 1999;15(1):38-45.
7. Lee SJ, Kim JK, Seo KY, Kim EK, Lee HK. Comparison of corneal nerve regeneration and sensitivity between LASIK and laser epithelial keratomileusis (LASEK). Am J Ophthalmol. 2006;141(6):1009-1015.
8. De Paiva CS, Chen Z, Koch DD, et al. The incidence and risk factors for developing dry eye after myopic LASIK.
Am J Ophthalmol. 2006;141(3):438-445.
9. Albietz JM, Lenton LM, McLennan SG. Chronic dry eye and regression after laser in situ keratomileusis for myopia. J Cataract Refract Surg. 2004;30(3):675-684.
10. Patel SV, McLaren JW, Kittleson KM, Bourne WM. Subbasal nerve density and corneal sensitivity after laser in situ keratomileusis: femtosecond laser vs mechanical microkeratome. Arch Ophthalmol. 2010;128(11):1413-1419.
11. Al-Aqaba MA, Fares U, Suleman H, Lowe J, Dua HS. Architecture and distribution of human corneal nerves.
Br J Ophthalmol. 2010;94(6):784-789.
12. Wei S, Wang Y. Comparison of corneal sensitivity between FS-LASIK and femtosecond lenticule extraction (ReLEx flex) or small-incision lenticule extraction (ReLEx smile) for myopic eyes. Graefes Arch Clin Exp Ophthalmol. 2013;251(6):1645-1654.
13. Vestergaard AH, Grønbech KT, Grauslund J, Ivarsen AR, Hjortdal JØ. Subbasal nerve morphology, corneal sensation, and tear film evaluation after refractive femtosecond laser lenticule extraction. Graefes Arch Clin Exp Ophthalmol. 2013;251(11):2591-2600.
14. Li M, Niu L, Qin B, et al. Confocal comparison of corneal reinnervation after small incision lenticule extraction (SMILE) and femtosecond laser in situ keratomileusis (FS-LASIK). PLoS One. 2013;9;8(12):e81435.
15. Mastropasqua L, Nubile M, Calienno R, Mastropasqua A, Salgari N, Lanzni M. Femtosecond refractive lenticule extraction for myopia-interface morphology and nerve fiber recovery. Paper presented at: the 4th Eucornea Congress; Amsterdam, Netherlands; October 4-5, 2013.
Roberta Calienno, MD
• National High Technology Eye Center, University of Chieti and Pescara, Italy
• roberta.calienno@gmail.com
• Financial disclosure: None
Leonardo Mastropasqua, MD
• Director of the Ophthalmology Clinic, Center of Excellence in Ophthalmology, National High Technology Eye Center, University G. D’Annunzio of Chieti and Pescara, Italy
• mastropa@unich.it
• Financial disclosure: None
Mario Nubile, MD
• Supervisor, Cornea and Ocular Surface Unit of the Ophthalmology Clinic, Center of Excellence in Ophthalmology, University G. d’Annunzio of Chieti and Pescara, Italy
• m.nubile@unich.it
• Financial disclosure: None
Niccolò Salgari, MD
• National High Technology Eye Center, University of Chieti and Pescara, Italy
• n.salgari@gmail.com
• Financial disclosure: None
