‘I Can See You, Doctor’
By Ashvin Agarwal, MS
A 68-year-old woman from Oman walked into my clinic with her son, who was holding her hand. Upon asking for the mother’s history, I was informed that other medical professionals had previously advised them that nothing could be done to treat her condition. As any surgeon/clinician/ophthalmologist would, I wondered, “Oh boy, is this a retina case for which there is no cure?” and “Will I be able to help?”
When I asked the patient how she was feeling, she said only, “I have been unable to see my loved ones for some time now.” Upon examination, I observed that she had a grade 4 cataract and a capsular bag that was dangling and on the verge of falling on the retina. The retina was completely invisible on indirect ophthalmoscopy; the only visual we had was a B-scan ultrasound image showing that it was attached.
Chief line of treatment
I decided to perform a planned manual cataract extraction and glued IOL procedure, even though we had no clear picture of the retina. No one in the operating room—myself included—was clear on whether the patient would regain sight.
The cataract extraction involved a lot of struggle, as I was facing a loose capsular bag with weak zonules and a grade 4 cataract. After the nucleus was removed, the bag gave way at many points. Retaining this weak and unhealthy bag was not ideal, so I performed a three-port pars plana vitrectomy to knock off the remnants of the bag. I then made partial thickness scleral flaps 180° apart to perform the glued IOL procedure, which has become my chief line of treatment in eyes with deficient capsular bags.
After the vitrectomy, I implanted a three-piece IOL behind the iris and externalized the haptics on both sides under the scleral flaps. I then tucked the haptics into Scharioth tunnels at the base of the flaps. I closed the case with a whisper to the patient saying, “Khallas,” which means “It is done” in Arabic. She replied with a simple “Shukran,” meaning “Thank you.”
THE waiting game
To me, the rest of that surgery day felt like waiting for my GMAT score and not knowing whether it was green or red, and, if red, then what next?
On postoperative day 1, the son brought his mother in while holding her hand again, as the patch was still on. At this point, my prayers to and belief in God increased to a maximum.
We removed the patch, and, the moment she opened her eye, she pointed at my face and said, “I can see you, doctor,” in Arabic. However, not even that was my proudest surgical moment. The patient then turned her head to her son and with watery eyes told him, “I see you.” You may think this was my proudest surgical moment, but that still was not it.
At that point, everyone in the room was happy. All of a sudden, the patient stood up and started walking toward the door. I said, “Where are you off to? I haven’t finished my examination.” To my amazement, she replied, “I want to see if I can open the door myself.”
At this point, I felt that the result of my surgery had brought a kind of joy to the patient that cannot be measured by any scale and had probably changed her life permanently, making this my proudest surgical moment.

The ‘Aha’ Moment
By Suven Bhattacharjee, MS, DO, DNB, FRF
The year 2007 was a turning point in the development of pupil expansion devices. A strand of polypropylene used as a pupil expansion ring, developed by Boris Malyugin, MD, PhD, was a truly disruptive innovation. Around the same time, I was thinking of a pupil expansion device on similar lines, but I had set my target at a 20-gauge or 1-mm incision. My goal was to develop a device that could be inserted and removed through an incision of that size.
Figure 1. A flange of an open ring is held with 23-gauge forceps at the incision.
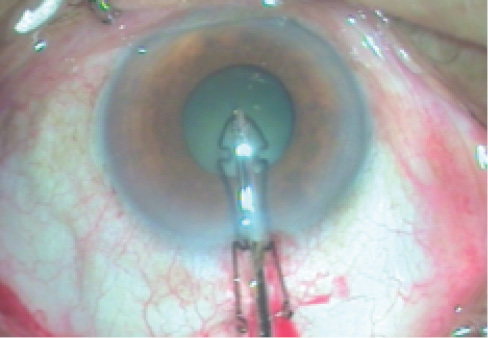
Figure 2. The flange of an open-ring device bends and the notches straighten as the device is carried through the incision.
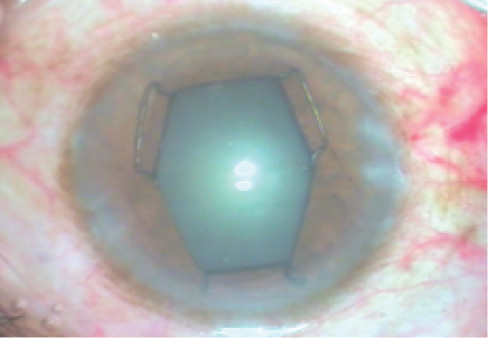
Figure 3. A pupil expanded with an open-ring device.
I was interested in creating a platform on which future pupil expansion devices could be developed for very small incisions. The device had to have a design that was user-friendly, easy to manufacture, and able to withstand the test of time over multiple iterations—something like what the J-loop or C-loop haptic has been to IOL design. It had to be simple, upscalable, and machinable. Machine manufacturing would produce a truly continuous ring without a joint and also reduce production times and costs. I also wanted the same platform to work for an expandable pupil expansion ring that I had been pondering. An expandable device would engage the nondilating pupil in a small contracted state and then manually or automatically undergo controlled expansion to attain an enlarged state, resulting in controlled, atraumatic expansion of the pupil. This would reduce the number of anterior chamber manipulations needed and the risk of collateral damage.
THE CONCEPT OF A SINGLE-PLANE DEVICE
The pupil margin-engaging part (scrolls or pockets) of available devices had upper and lower parts, making them actually biplanar structures. The space or gap that received the pupil margin was oriented either perpendicular or oblique to the principal plane of the device.
I thought of a radically different concept to engage the pupil margin. I brought all of the bends at the corners to a single plane, effectively bringing the entire device to a single plane and reducing its vertical profile. This led to the single-plane design of the Bhattacharjee Pupil Expansion Ring, which has notches at the corners and flanges at the sides. Alternate flanges are placed behind the pupil margin, so that the pupil margin is straddled across the plane of the device, whereas in all previous devices a part of the device straddled across the plane of the pupil margin. This works like a paper clip: The clip remains in a single plane while the paper bends.
THE MANUFACTURING PROCESS
When I had a reasonable drawing, I looked for a manufacturer. Designing the jig was difficult, and no one wanted to invest time and money in something that might not see the light of day. The writing on the wall clearly read, “Do it yourself.” I began drawing designs, researching plastic materials, and working on different manufacturing technologies. Eventually, I made the jig and the device myself in a home-based laboratory.
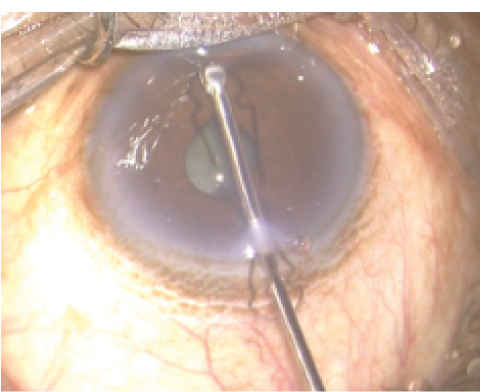
Figure 1. A flange of an open ring is held with 23-gauge forceps at the incision.
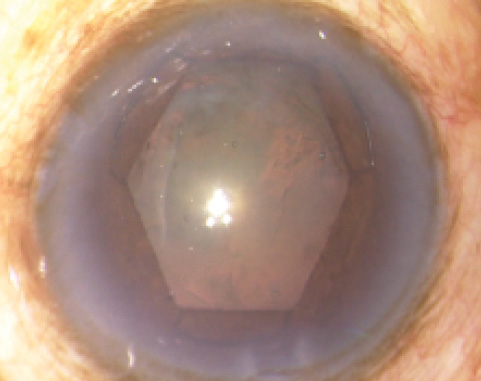
Figure 2. The flange of an open-ring device bends and the notches straighten as the device is carried through the incision.
Nylon is stiffer than polypropylene, and I knew that a thin strand of nylon would work well to expand the pupil. This augured well for insertion through a 20-gauge incision. Additionally, the thermoplastic properties of nylon allowed the fine notches to be shaped. Nylon also has better shape memory, so the notches regain shape even after being fully straightened.
My first few designs were open-ring devices because I believed that the only way I could pass a pupil expansion device through a 20-gauge incision was by inserting the free end. The design progressed from a house-shaped version to a tower-shaped version. I would insert one free end through a sideport and pass 23-gauge forceps through an opposite sideport to hold the end and draw the device into the anterior chamber. This worked but called for a lot of instrumentation and manipulation.
I racked my brain to find a better way of inserting the device through a 20-gauge incision. I tried inserting the notch first by holding it with 23-gauge forceps; this was an ordeal because nylon is flexible and resilient. As I inserted one side of the notch, the other side would slip out. I was able to coax the device in after many attempts, but I was not convinced that this was the best method; I had run out of ideas.
Success
During one such exercise, the device slipped and positioned a flange at the 20-gauge incision. Out of sheer desperation, I held the flange with 23-gauge forceps and thrust it through the incision (Figure 1). Lo and behold, the flange bent completely over itself, the notches straightened out, and the device sailed through the 20-gauge incision (Figure 2).
This was my proudest surgical moment, my “aha” moment. I had successfully inserted a pupil expansion device through a 20-gauge incision smoothly, convincingly, and reproducibly. I had realized my dream, and the pupil expanded well too (Figure 3).
This paved the way for further improvements. I quickly moved to a closed-ring version, as I no longer needed the free end. The ends of 5-0 (0.1 mm) nylon are joined with glue to form the closed ring. The current square- and hexagon-shaped devices are easily inserted and removed through a 1-mm incision and provide reproducible and convincing expansion of the pupil (Figures 4 and 5).
I firmly believe this simple design will be the platform for all future pupil expansion devices. These manually manufactured devices are undergoing clinical trials, and I have pending patent applications. The machine-manufactured ring without a joint will be available soon. And research is underway for the Expandable Pupil Expansion Ring, for which I have a published US patent application.

Helping to Evolve MICS and Keratorefractive Surgery
By Detlev R.H. Breyer, MD
My proudest surgical moments involve my modest participation in the evolution of microincision cataract surgery (MICS) and keratorefractive surgery.
It all started many years ago, when I was involved with Rupert Menapace, MD; Christian Prünte, MD; and Oertli in the development of coaxial MICS with a 1.5-mm incision. This was a time when most surgeons were using an incision size of greater than 3 mm; further, some were performing fluidics-unfriendly bimanual MICS, often either inducing astigmatism or fighting leakage and wound burn. In contrast, we preferred coaxial over bimanual MICS to avoid surgically induced astigmatism (SIA), as SIA-free surgery seemed to us the perfect way to improve the results of toric IOL procedures.

Figure 6. Screen transparency for toric IOLs (STACY), developed by Dr. Breyer.
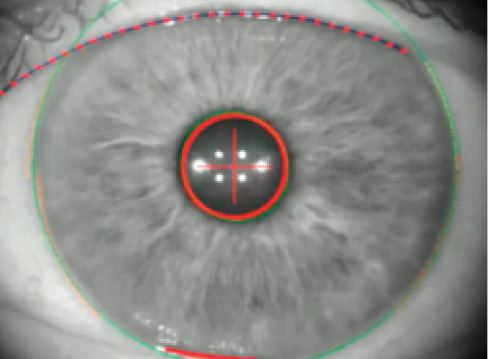
Figure 7. Simultaneous keratoscopy and iris registration with the Cassini.
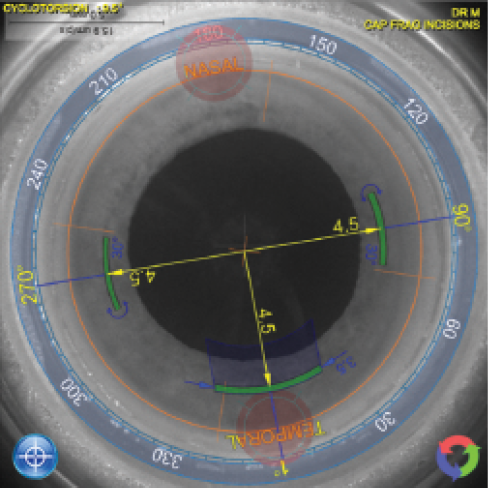
Figure 8. Import of Cassini data into the Lensar femtosecond laser and correction for cyclotorsion after comparison of Cassini and Lensar iris registration.
To further improve toric IOL implantation, in 2006, with the help of optometrist Ekehard Emmel, we developed the surgical principle to orient toric IOLs using a screen transparency, or STACY (Carl Zeiss Meditec; Figure 6), rather than the widely used and relatively imprecise Mendez ring. STACY was more accurate and has subsequently been adopted by many companies producing toric IOLs over the years. We recently published the long-term results of this surgical approach together with Gerd U. Auffarth, MD, PhD, FEBO; and Florian D. Kretz, MD.1
In 2009, I implanted the first bitoric bifocal multifocal IOL, made by AcriTec (now the AT LISA by Carl Zeiss Meditec). My first two patients to receive this lens were a journalist and a lawyer, both of whom had astigmatism greater than 4.00 D. The results were stunning. Although it was somewhat progressive at the time, nowadays the combination of multifocal and toric components is a part of every cataract refractive surgeon’s portfolio.
Overall, my proudest surgical moment related to astigmatism correction was reached 2 months ago, when I was able to produce individual customized toric multifocal IOLs (Oculentis) based on aberrometry (KR-W1; Topcon) and perform simultaneous iris registration and LED-guided keratoscopy using the Cassini (i-Optics; Figure 7). Those data were then transferred via cloud to our Lensar femtosecond laser. After checking for cyclorotation by comparing these data with the Cassini, we used the laser to mark the cornea intrastromally with 5° LRIs on the target axis (Figure 8). On postoperative day 1, the IOL was precisely on axis, and the patient’s visual acuity was 20/20 (Figure 9). This is one of the few moments in a surgical career when the surgeon realizes that surgical art has turned into science and the goal of optimal patient care has been achieved.
MOMENTS OF PERSONAL SUCCESS
My proudest personal moment as an ophthalmic surgeon, however, was being selected to receive several best presentation of session papers for my scientific work on small incision lenticule extraction (SMILE). Initially, it was difficult to gain acceptance as one of the early pioneers of a new method in keratorefractive surgery when most surgeons were doing femtosecond LASIK. I remember giving my first course on SMILE at a scientific congress: It was last day of the meeting, in the furthest room, with a total of four participants. At this year’s European Society of Cataract and Refractive Surgeons (ESCRS) meeting in Barcelona, it seemed more like 200 participants who judged SMILE to be the keratorefractive procedure of the future following femtosecond LASIK.
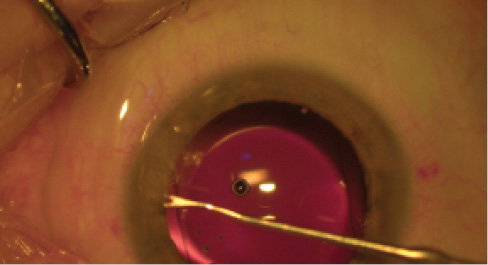
Figure 9. Perfect alignment of a toric multifocal IOL with corneal marks.
My most recent pioneering work was the invention of the Düsseldorf formula. Last year at the International Society of Presbyopia (ISOP) and ESCRS meeting, I introduced a multifocal concept without photopic phenomena: blended vision with the segmental, nonsymmetric Lentis Comfort (Oculentis) multifocal IOL. With this approach, the far dominant eye was targeted for emmetropia and the near dominant for -1.50 D, resulting in a distance visual acuity of 20/20 or better and a near visual acuity of at least Nieden 0.7, without halo and glare. Glasses were required only for reading small print.
A SOLID SUPPORT SYSTEM
I want to thank all of my industry and scientific partners and friends for believing in my work and trusting me. A very special thank you goes to my wife Anitra, who always supported my attempts to reach out for my proudest surgical moment while being away from home. And one last remark: I am more thankful than proud that luck was with me at the right time.
1. Kretz FA, Koss MJ, Auffarth GU. Intermediate and near visual acuity of an aspheric, bifocal, diffractive multifocal intraocular lens with +3.25 D near addition. J Refract Surg. 2015;31(5):295-299.

The Birth of Lamellar Techniques With the Laser
By Lucio Buratto, MD
In July 1985, I was in Houston, Texas, visiting Ralph Berkeley, MD, a refractive surgeon who was performing a large number of corneal surgery procedures (RK) to correct myopia. Dr. Berkeley was using the incisional techniques developed by Svyatoslav Fyodorov, MD, and Leo Bores, MD.

Figure 10. Refractive resection using the blade of the Barraquer cryolathe.

Figure 11. Lamellar disk on the bench following the refractive cut.
On a break between surgeries, Dr. Berkeley mentioned to me that Lee T. Nordan, MD, a surgeon from California, was operating in the same building that day. Dr. Nordan traveled to Houston regularly to operate on patients with high myopia using the freeze cryolathe technique developed by José Ignacio Barraquer, MD (Figure 10). Driven by curiosity, I went to watch Dr. Nordan perform a few of his operations, and that was when my interest in lamellar surgery for myopia commenced.
In September 1985, a couple of months after my visit to Houston, I traveled to the Centre Hospitalier Regional in Brest, France, to take part in live sessions of cataract surgery with phacoemulsification and posterior chamber IOL implantation. There, I met Jörg H. Krumeich, MD, who was also taking part in the live surgery and demonstrating a no-freeze (and consequently no-cryolathe) technique for myopic keratomileusis (Figure 11).
In the space of 2 months, I had observed two different techniques for keratomileusis, one more interesting than the next. I was enthralled by what I had seen, and I asked Dr. Krumeich if I could visit him in his clinic—a journey I made a few weeks later. On my return, I began using Dr. Krumeich’s technique, albeit with some difficulty, as the technique was by no means easy. In the meantime, Dr. Nordan had started traveling to Italy to operate in the clinic of Fabio Dossi, MD, and he taught me the keratomileusis technique using the cryolathe that I had also learned from Dr. Krumeich.
At this point, my interest in keratomileusis was high. When Antonio Ruiz, MD, developed the in situ keratomileusis technique involving two cuts with the microkeratome—one 360° cut to produce a flap that was 250 μm deep and 7 to 8 mm in diameter, and the other a refractive cut in situ on the stroma that was much thinner and smaller in size—I began using this technique as well (Figure 12).
At that same time, another surgeon, Stephen L. Trokel, MD, was testing the excimer laser for use on corneal tissue. Soon after, some of my international colleagues began using the laser to correct myopia. In April 1989, I went to Berlin, Germany, to visit Theo Seiler, MD, PhD, the first surgeon in Europe to use the excimer laser, and there I observed the first cases of PRK.
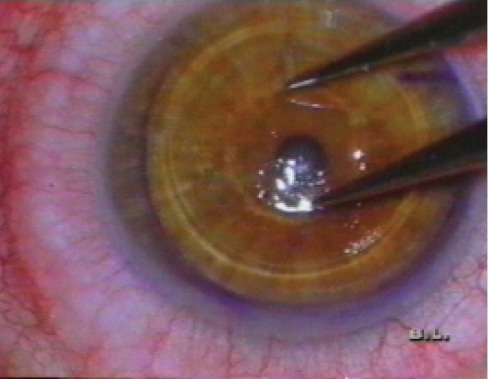
Figure 12. In situ keratomileusis. Note the wide lamellar cut and smaller refractive cut.
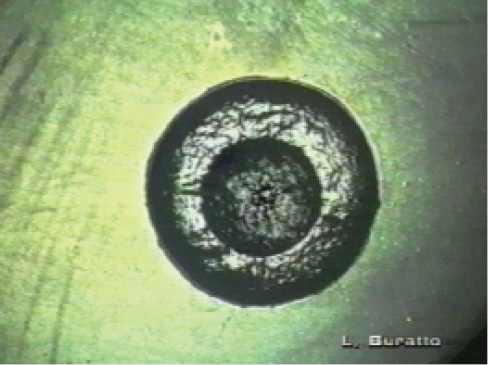
Figure 13. Mono-zone ablation with the Summit excimer laser (4.5 mm).
AN EXCIMER LASER OF MY OWN
I was fascinated by the future prospects of the excimer laser, and, immediately after this experience in Germany, I ordered a device from Summit Technologies. I transported the laser to the hospital in Mestre-Venice, where the chief consultant, Giovanni Rama, MD, was the best Italian corneal surgeon at the time.
Assisted by Claudio Genisi, MD, I began correcting a variety of corneal pathologies using these new laser techniques. In parallel, I continued treating numerous cases of severe myopia with the keratomileusis techniques, mostly using the no-freeze technique at the Krumeich bench and occasionally the Ruiz technique with the double cut at the microkeratome.
Despite being fairly satisfied with some of the results I achieved, I was aware that the refractive cuts in these two keratomileusis methods were created with metal blades that lacked precision in terms of the diameter of the optical zone and the centration of the cut. More important, repeatability in removing the exact quantity of tissue necessary to achieve the target refractive correction was poor. Further, in addition to the far-from-easy execution, these techniques were associated with a considerable number of complications.
At that point, I asked myself, “Why not use the laser?” In reality, the question was, “How can the laser be used?”
Dr. Krumeich’s technique involved cutting a flap of corneal tissue that was 8.5 to 9 mm in diameter and 300 to 350 μm thick. The flap was positioned on the work bench, and the microkeratome was used to perform a refractive cut on the stromal face of the flap, removing a piece of tissue 20 to 30 μm thick, with an optical zone diameter of between 4 and 5 mm. I decided to do exactly the same thing using the laser (Figure 13).
I began treating some cases of severe myopia using the laser on the back of the flap. Then, just a few weeks later, I had an idea to modify the technique developed by Dr. Ruiz, namely performing the refractive cut in situ, using the laser instead of the microkeratome. That is how lamellar techniques with the laser were born!
Countless improvements have been made to the technique over the years, and today the procedures are completely different from what they were back then. However, it gives me great joy and satisfaction to know that I played an instrumental role in the evolution of refractive surgery.

Operating on a 5-Day-Old Girl in Conjunction With a Multidisciplinary Team
By Sunil Shah, MBBS, FRCOphth, FRCS(Ed)
As surgeons, we come up against surgical challenges all the time. More often than not, these are challenges that arise in the middle of what should otherwise have been routine surgery.
Recently, I encountered a notable challenge. This was an urgent but planned intervention not only to save a patient’s eyes but also with the additional pressure that systemic intervention could not occur until the eyes were treated. A multidisciplinary team including pediatric ophthalmologists (Manoj Parulekar, FRCS(Ed); Joe Abbott, FRCOphth; and John Ainsworth, FRCOphth), a corneal surgeon (myself), and a liver transplant team (led by Girish Gupte, MRCPI) rose to the challenge.
securing integrity of the eyes
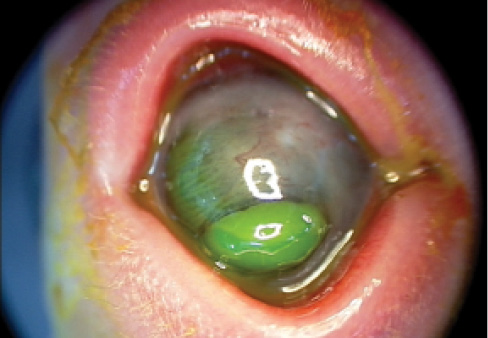
Figure 14. A 2-day-old baby was noted to have bilateral anterior segment dysgenesis with bilateral corneal perforations, with the lens partially sealing the larger defect (anterior capsule stained green). This eye, which had the larger corneal perforation, was operated on first.
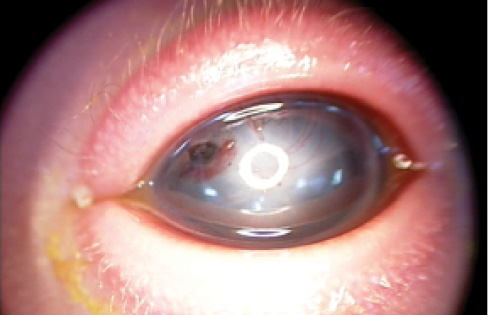
Figure 15. The patient’s other eye had a smaller corneal perforation than the first, but the cornea was paper-thin.
A 2-day-old girl with DiGeorge syndrome was referred to Birmingham Children’s Hospital. DiGeorge syndrome is the most common microdeletion syndrome (22q11 deletion). It is autosomal-dominant in inheritance and has an estimated prevalence of 1:4000. DiGeorge syndrome is associated with cardiac anomalies, including primarily the tetralogy of Fallot and truncus arteriosis.
The baby was admitted to intensive care at Birmingham Children’s Hospital. She was noted to have bilateral anterior segment dysgenesis with bilateral corneal perforations (Figures 14 and 15), with the lens partially sealing the larger defect. Both posterior segments were grossly normal on ultrasound, and good and equal light perception in each eye had been previously documented.
That day, the patient underwent corneal gluing in both eyes as a temporizing measure (Figure 16), and corneal material was arranged for urgent tectonic corneal grafts. The cardiac team anticipated surgical intervention once the integrity of the eyes had been secured.
bilateral corneal transplantation
With the surgical team having had just a few days to consider the prospects of performing bilateral corneal transplantation on a 5-day-old girl, the surgery commenced.
The eye with the largest perforation was operated on first. Although it is normal to use a device such as a Flieringa ring to support pediatric eyes, all of the rings were too large for this case; we ended up securing scleral sutures to the drape with tape. The cornea was dissected off the lens with great care, as the lens was plugging the perforation, and, hence, there was no anterior chamber.
After this dissection was successfully achieved, it was observed that the lens was proud and bulging where it had sealed the original perforation. Therefore, lensectomy and anterior vitrectomy were performed. As part of the anterior dysgenesis, there was no appreciable angle. A donor corneal button, oversized by 0.75 mm, was secured with interrupted 10-0 nylon.
Normally, one would have finished at this stage. However, the other eye (Figure 15) still needed examination. The hope was that the corneal glue would be enough to hold this eye. On examination, however, the cornea was paper-thin. There was no pupillary aperture. The corneal glue was not firmly adherent, and it was removed to enable a better examination.
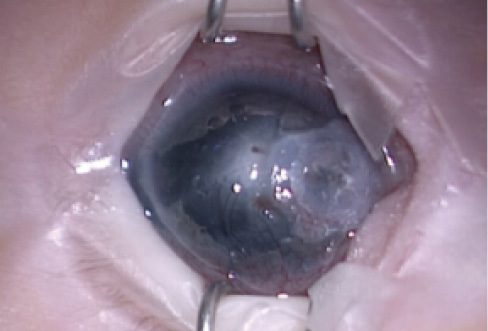
Figure 16. The patient’s left eye after corneal gluing but before PKP.
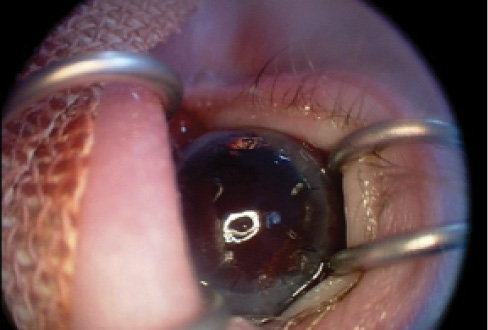
Figure 17. The patient’s eye postoperatively, after alternate suture removal.
Given the state of the cornea, we felt it was necessary to proceed with corneal grafting in that eye as well. Findings were similar to those in the first eye, with the iris and lens attached together to the cornea. The cornea and iris were dissected off the lens, but the lens was abnormal, with a lobulated appearance. Lensectomy and anterior vitrectomy were once again performed, and a new corneal button was secured in the same manner as in the other eye.
The baby was transferred, still on the ventilator, back to the intensive care unit. In consultation with the liver transplant team, she was started on tacrolimus suspension. The cardiology multidisciplinary team decided to perform a transcatheter procedure and to consider a Blalock-Taussig shunt (a temporary surgical procedure) if that failed. To date, they have decided to hold off on other cardiac surgery.
Although the corneas remain clear and the globe secure (Figure 17), after 1 month, the baby girl has so far had multiple failed extubation attempts.
CONCLUSION
For a primarily adult surgeon, this sort of case raises a number of concerns, including the age of the baby, the bilateral nature of the surgery, and the systemic considerations. One does not come across many cases in which the equipment does not fit because it is too big or when one is trying to secure the eyes to allow potentially life-saving cardiac surgery.
Additionally, although pediatric ophthalmologists may be used to removing neonatal cataractous lenses, it requires some mental adjustment for an adult surgeon to remove a 5-day-old clear lens. However, after 6 hours in the operating room, because of both the complex nature of the anesthetic and the complex nature of the surgery, I felt we had given this child every possible chance for the future.

‘The Doctor Who Saved My Eyes and My Life’
By Gustavo E. Tamayo, MD
Identifying my proudest surgical moment is a difficult task. During my years in ophthalmology, many advances in technology have given me opportunities to offer new treatments to my patients and, thus, provided me with many moments of happiness and pride. Through my practice, I have been able to improve the quality of life of thousands of patients.
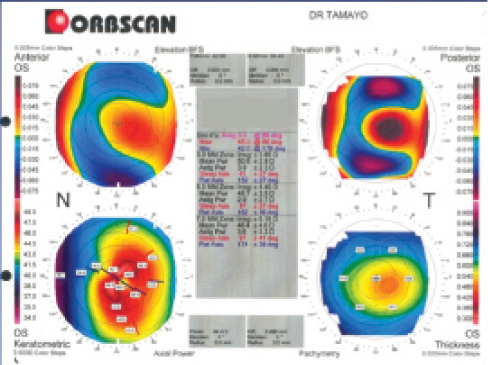
Figure 18. The patient’s preoperative elevation topography.
However, there was one case that turned out to be a life-changing experience for both the patient and me. The result of that surgery encouraged me to persevere in the fight for another surgical option for keratoconus, a disorder that can be a devastating problem for young people who become intolerant to contact lenses and are unable to achieve decent vision with glasses.
In 1998, I presented the first eight cases of PRK for the treatment of keratoconus and was met with a great deal of criticism and controversy. A subsequent patient from this early experience with PRK for keratoconus is the subject of this story.
A Heavy eye rubber
In April 1999, a 23-year-old man arrived at my clinic with his parents. He had been diagnosed with progressive keratoconus and severe allergic conjunctivitis. The patient had tried many times to wear rigid contact lenses but was unsuccessful due to his severe allergy, and his glasses were unable to provide decent visual acuity. Eventually, he had to quit his engineering studies because of his poor vision.
Corneal transplantation had been recommended to the patient—in fact, he was told this was his only option to regain sight. His parents, aware of all the possible complications of the surgery, were terrified. Then a friend who was one of the eight initial keratoconus patients I treated told the family about this new procedure for keratoconus.
My examination and diagnosis were clear: moderate keratoconus in progress due to heavy eye rubbing associated with the ocular allergy. I presented the family with the surgical option of PRK (without CXL at the time). Eye rubbing was forbidden, and heavy treatment for the allergy was initiated.
The patient and his parents accepted and signed a consent form for this treatment with the promise that the worst possible scenario would be no effect after surgery, at which point a corneal transplant would be the only option. They signed with the enthusiasm of Catholic parents who believed in miracles. I was not so sure about a miracle, but after my promising first cases I had good expectations and a lot of hope.
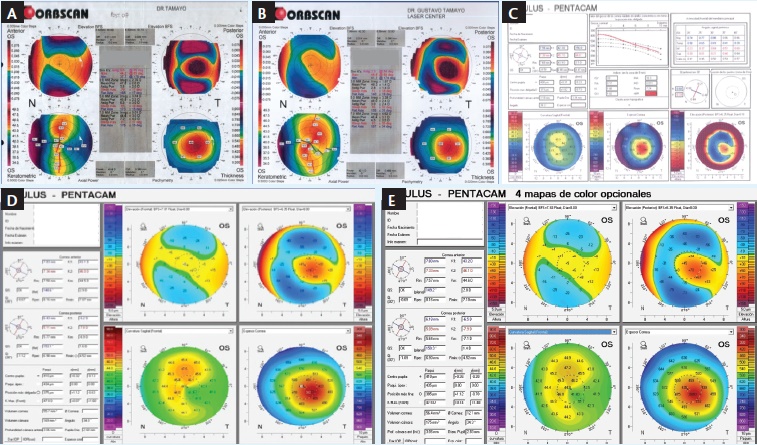
Figure 19. Elevation topography at 1 month (A), 3 years (B), 10 years (C), 14 years (D), and 17 years (E) postoperative.
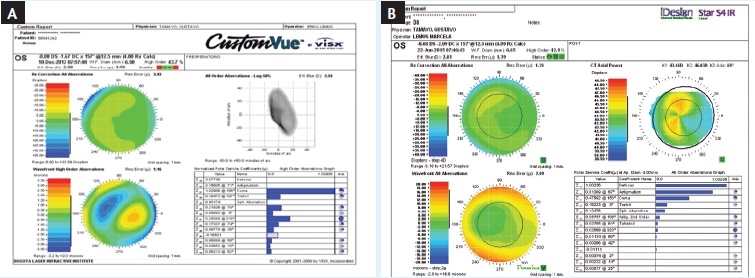
Figure 20. The patient’s wavefronts in 2012 (A) and 2015 (B) demonstrate stability and an excellent refractive result.
THE END RESULT
Seventeen years have passed since that dramatic moment, and this young man achieved his dreams without needing a corneal transplant. He is now employed as an engineer and is married with two beautiful children. He does not wear glasses, despite his vision being only 20/30. During his most recent visit this year, he brought the family along for the examination. He has been stable throughout the years, with periodic visits—and, of course, no eye rubbing.
The topographies shown in Figure 18 illustrate the patient’s cone at the preoperative examination, and Figure 19 shows successive elevation topographies at 1 month (Figure 19A), 3 years (Figure 19B), 10 years (Figure 19C), 14 years (Figure 19D), and 17 years (Figure 19E) postoperative. All of them are clear examples of a good and completely stable result.

Figure 21. Dr. Tamayo and the patient who introduced him as “the doctor who saved my eyes and my life.”
The wavefronts taken in 2012 and 2015 (Figure 20) also show stability and an excellent refractive result. Unfortunately, at his most recent visit, I observed that some aging problems had started to occur, and the patient was informed about the rough road of presbyopia ahead. Nonetheless, he still has a couple more years to enjoy without glasses.
My proudest moment came when the patient introduced me to his kids as “the doctor who saved my eyes and my life.” After I wiped away tears, we took a picture together (Figure 21). I was so proud!

Surgery on an Escaped Captive
By Khiun F. Tjia, MD
My proudest surgical moment in recent years involved a Romanian woman, a retired veterinarian who came to me for help in early 2014. Her story is as bizarre as it is horrific.
Many years ago, this woman sought political asylum in the Netherlands during the Ceausescu dictatorship era. In the process, she was helped by a Dutch woman, with whom she had exchanged Christmas cards ever since. Decades later, she ended up at the Dutch woman’s doorstep somewhere in the rural Netherlands after a frightful journey from Romania, which had just joined the European Union. What happened to her in the time between is a gruesome but unfortunately true tale.
In Romania, the woman was held captive, chained in a dark cellar, by her own son. Forced to withstand terrible circumstances in solitary confinement, she was also repeatedly beaten on her ears and eyes with the goal of rendering her deaf and blind. The son’s reason for doing this was to make his mother helpless and unable to escape from captivity. The motivation behind his terrible actions was that every month he needed his mother to collect her pension in person; the son saw this as an easy source of income and subsequently used all of the pension money for himself.
One day, the woman was sitting alone in the car with her passport and recently collected pension money while her son was running errands, trusting his mother’s blindness to prevent her from running away. Seeing this as an opportunity to escape, the woman got out of the car, grabbed the first bystander she could find, and asked to be brought to a taxi. The taxi took her to a hotel, where she requested to stay for a few days. She then asked for help booking a one-way flight to Amsterdam and for transportation to the airport a few days later.

Figure 22. The woman on the right was held captive for 4 years, during which she was beaten blind and deaf. She had hypermature cataracts in both eyes with large zonulolysis. The bilateral surgery took 2 hours to perform, but the patient is now extremely happy and back in her home country.
Once in Amsterdam, the woman relied on the same tactics of asking people to bring her to the train station and locate a taxi. With the address from the Christmas card in her hand, she finally ended up at the doorstep of her former benefactor from 30 years ago (the woman on the left in Figure 22). The Dutch woman was completely overwhelmed and shocked, but after listening to the woman’s sad story she decided to help her look for a surgeon who was willing to operate on her badly damaged eyes. This was not an easy task, and several hospitals refused to help. Finally, the Dutch woman got in touch with our hospital, and I agreed to see her friend.
a long bilateral SURGical course
Fortunately, the patient was not completely deaf, and I was able to communicate somewhat with her in German. However, she had only light perception in her left and right eyes. At the slit lamp, both eyes showed hypermature intumescent white cataracts. I suspected potential zonular damage because of the traumatic origin of the cataracts.
It took me 2 hours to perform surgery on both eyes. I employed several key strategies, including performing each procedure in a closed anterior chamber overinflated with OVD to flatten the bulging anterior capsule. I decompressed the patient’s swollen lens by aspirating cortex with a 25-gauge needle mounted on a small-volume, fluid-filled syringe. Careful creation of a large capsulorrhexis after trypan blue dye staining was employed to minimize stress on the zonules. I performed careful nucleus disassembly by cracking and/or chopping the nucleus into multiple small fragments.
Because of my suspicion of zonular dialysis, I implanted a capsular tension ring. I injected triamcinolone multiple times to identify vitreous and performed a subsequent two-port anterior vitrectomy to avoid unnecessary retinal traction. In one eye, I implanted a three-piece IOL in the sulcus with optic capture to ensure a stable position of both the IOL and the capsule. Surgery in this eye was successfully completed, and 20/25 visual acuity was achieved.
In the other eye, I failed to maintain the capsulorrhexis, so I decided to leave this eye aphakic in the primary surgery. The retina is undamaged, and secondary IOL implantation is feasible.
CONCLUSION
This patient’s happiness and gratitude during our latest meeting at her 1-week postoperative visit was breathtaking. Experiences like this are what make physicians thrive and enable us to cope with the everyday stress and bureaucracy of practicing medicine. I will never forget this patient’s transition from a desperate, deeply worried, sad person to a merry, proud, and independent individual. She gave me a small personal present after her surgery, but the smile on her face was the greatest gift for me.


When the Microscope Light Turned On
By Antonio Toso, MD; and Simonetta Morselli, MD
Dr. Morselli: In February 2015, Dr. Toso and I saw a 45-day-old baby girl because her pediatrician had concerns about the red reflex of both eyes. We performed our examination with the patient under sedation in order to better understand the baby’s eye problems. Under the microscope, we detected bilateral congenital cataracts. The right eye was smaller than the left, and echographic examination suggested a persistent hyaloid artery.
We worried about how to inform the parents without turning this news into a tragedy. It was a difficult, emotional situation. We tried to clearly explain all of the details to them, including the pros and cons of surgery, our surgical technique, and the postoperative follow-up.
After this first hurdle was overcome (with a lot of emotional stress), we had to solve another problem. The anesthetist told us that we would be unable to operate on the baby at our hospital because the pediatric intensive care unit was not available. We did not want to abandon this baby or discharge her.
Fortunately, we had an idea. We called one of our colleagues in Verona, where a pediatric intensive care unit was available. He confirmed that we would be able to operate on the baby girl at that facility. In just 1 week, we organized the surgery at this other hospital. During that period, we spent a lot of time imagining all possibilities of the surgery and training ourselves to overcome any potential difficulties.
Eventually, the day came. When we entered the operating room, we saw the baby girl ready for surgery. My heart was beating quickly as I prepared the little eye for surgery, but once the microscope light was turned on the surgeon inside me began to concentrate.
The pupil was not well dilated, and I understood the possibility of encountering the hyaloid artery. I started to perform a corneoscleral incision, keeping in mind some concerns that my co-surgeon, Dr. Toso, had expressed about the hyaloid artery remnant. Staining of the capsule with trypan blue dye was prolonged slightly in order to obtain stiffness of the anterior capsule. During hydrodissection, the pupil started to become miotic, and I tried first to use a high-molecular-weight OVD.
At the end of the cataract aspiration, it was impossible to proceed with the pupil that small. I conversed with Dr. Toso about the best way to enlarge the pupil. In the end, we decided not to use a pupillary ring because of the small dimensions of the eye, but chose instead to place four iris hooks. I was concerned about performing four more incisions and, thus, needing four more stitches that would cause more inflammation in the baby’s eye, but I had no other choice.
We then proceeded with the posterior capsulorrhexis, which was a real challenge. The posterior fibrosis was finally removed thanks to Dr. Toso’s smart suggestion to use vitreoretinal forceps, which he describes further below. In emotional situations such as this, it is helpful to work as a team because the first surgeon may be too focused to see other approaches due to the stress of the case. For this reason, it is important to work with someone you trust. Posterior vitrectomy and IOL implantation proceeded in a more relaxed manner, like navigating a downhill winter slope on a nice sunny day.
Dr. Toso: The sympathy we felt for this baby and her family since her first visit continued to increase throughout this case, especially when we entered the operating room. There we saw the baby girl, with her lovely little round face, under general anesthesia. Our fingers touched her soft, warm cheeks. It was difficult to see this tiny human being sleeping not in her cradle but on a cold bed with a laryngeal tube.
We hesitated a moment because of this intensely emotional situation. We wondered if we could handle it. Then, we reminded ourselves that we had the skills, experience, and well-balanced temperaments needed to cope with this challenging task. So we started our job. Dr. Morselli operated first on the right eye, and then I operated on the left.
During surgery on the right eye, Dr. Morselli had to deal with a challenging posterior capsular opacity that could not be removed with standard microincision capsulorrhexis forceps. Thinking of my daily experiences in vitreoretinal surgeries, I suggested that we use 23-gauge curved vitreoretinal scissors to cut the posterior capsule around the opacity and then use two kinds of 23-gauge vitreoretinal forceps, end-gripping and serrated (ie, crocodile), to remove the opacity en bloc.
The surgery on the left eye was slightly less difficult thanks to a softer and more workable posterior capsular opacity. Fortunately, the microincision capsulorrhexis forceps worked well in this eye.
Based on the literature regarding IOL implantation in bilateral congenital cataract surgery, we decided to employ this technique with the goal of preventing posterior opacification and keeping the vitreous from extending anterior to the IOL. This approach also ensures stability and long-term centration of the IOL. The technique requires anterior vitrectomy to be performed using a 23-gauge cutter, which then allows the IOL optic to be captured through the posterior capsulorrhexis opening.
The operations in both eyes lasted about 2 hours, and, in the end, everything was all right. After surgery, we joined the parents and told them that the surgery had been successful. The mother burst into tears of joy and hugged Dr. Morselli, while the father took our hands and exclaimed, “Thank you very much, doctors. I owe you!” I told him, “I took care of your daughter as if she were mine.” n
Ashvin Agarwal, MS
• Senior Consultant, Dr. Agarwal’s Eye Hospital and Eye Research Centre, Chennai, India
• agarwal.ashvin@gmail.com
• Financial disclosure: None
Suven Bhattacharjee, MS, DO, DNB, FRF
• Director and Consultant, Nayan Eye Centre, Kolkata, India
• suvenb@gmail.com
• Financial disclosure: Pending patent applications for the Pupil Expansion Ring and Expandable Pupil Expansion Ring
Detlev R.H. Breyer, MD
• Head of Breyer-Kaymak-Klabe Eye Surgery, Düsseldorf, Germany
• Head of PremiumEyes Laser Eye Surgery, Düsseldorf, Germany
• Member, CRST Europe Editorial Board
• d.breyer@augenchirurgie.clinic
• Financial disclosure: Consultant, Medical Advisory Board Member, Speaker (Alcon, Carl Zeiss Meditec, STAAR Surgical, Oculentis, Abbott Medical Optics, Topcon, Lensar, Ruck)
Lucio Buratto, MD
• Director, Centro Ambrosiano Oftalmico, Milan, Italy
• office@buratto.com
• Financial disclosure: None
Simonetta Morselli, MD
• Head of the Ophthalmology Department, S. Bassiano Hospital, Bassano del Grappa, Italy
• Member, CRST Europe Editorial Board
• simonetta.morselli@gmail.com
• Financial disclosure: None
Sunil Shah, MBBS, FRCOphth, FRCS(Ed)
• Midland Eye, Solihull, West Midlands, United Kingdom
• Birmingham & Midland Eye Centre, Birmingham, United Kingdom
• sunilshah@doctors.net.uk
• Financial disclosure: None
Gustavo E. Tamayo, MD
• Director, Bogotá Laser Refractive Institute, Bogotá, Colombia
• gtvotmy@telecorp.net
• Financial disclosure: None
Khiun F. Tjia, MD
• Anterior segment specialist, Isala Clinics, Zwolle, Netherlands
• Editor Emeritus, CRST Europe
• kftjia@gmail.com
• Financial disclosure: None
Antonio Toso, MD
• Head of the Vitreoretinal Surgery Unit, Ophthalmology Department, S. Bassiano Hospital, Bassano del Grappa, Italy
• antonio.toso@gmail.com
• Financial disclosure: None