First Patient Receives Stem Cell Treatment for wet amd in the United Kingdom
A woman in the United Kingdom has become the first patient to receive a pioneering stem cell treatment designed to restore sight in people with wet age-related macular degeneration (AMD), according to a news release.
The operation, performed at Moorfields Eye Hospital in London, is a major milestone in the London Project to Cure Blindness, which was established 10 years ago with the aim of curing vision loss in patients with wet AMD. It is also the result of a partnership between Moorfields Eye Hospital, the University College London Institute of Ophthalmology, and the National Institute for Health Research, the news release said. Pfizer joined the partnership in 2009 with the goal of helping to turn ideas into potential therapies.
Following the successful operation, a trial of the new stem-cell–derived treatment has commenced at Moorfields Eye Hospital. The trial is investigating the safety and efficacy of transplanting retinal pigment epithelium cells derived from stem cells to treat patients with severe visual loss from wet AMD. These cells are used to replace cells that are diseased in AMD. This is usually done using a specially engineered patch inserted behind the retina in an operation lasting 1 to 2 hours, the news release said.
No complications of the surgery have been reported to date. The patient wishes to remain anonymous, but the team hopes to determine her outcome in terms of initial visual acuity by December 2015.
“There is real potential that people with wet AMD will benefit in the future from transplantation of these cells,” said Lyndon Da Cruz, MD, who is performing the operations at Moorfields Eye Hospital and co-leading the London Project.
The trial will recruit patients over a period of 18 months. Each patient will be followed for 1 year to assess the safety and stability of the cells and whether there is an effect in restoring vision.
Study Shows 1.1 Billion People Need Glasses but Lack Access
OneSight, an independent nonprofit, announced findings from The Global Need for Glasses Study, which was conducted to determine how accessible vision care and quality eyewear are around the world.
The study defined visual impairment by a child’s ability to perform well in school and an adult’s ability to be productive at work. Access was defined by glasses being available at an affordable price, a news release said.
According to the report, 1.1 billion people around the world need glasses but do not have access to vision care. One in seven people globally cannot see clearly because they do not have access to glasses. In certain countries, up to 44% of the population needs glasses but lacks the access to get them. There are 239 million children globally who need glasses but lack access. The crisis is most acute in Sub-Saharan Africa and Southeast Asia, the news release said.
The study found that the ability to forgo wages for a day, pay for transportation, and ultimately pay for vision care was a greater barrier than just the cost of glasses themselves.
“Clear vision can fundamentally change someone’s world and help unlock their full potential. Yet, for one in seven people around the world, clear vision is not accessible. We can begin to solve this issue by bringing awareness to the global vision care crisis,” Jason Singh, OD, OneSight Executive Director, said in the news release. “We know the difference a simple pair of glasses can make in a person’s life. A child may be able to see the blackboard clearly for the first time. A parent may be better able to support their family by producing more at work, all thanks to clear vision. The immediate and long-term impact is powerful.”
OneSight’s mission is to eradicate the global vision care crisis in our lifetime through sustainable and charitable solutions. The company operates charitable clinics around the world to provide communities with eye exams and free glasses.
Fewer Keratoplasties for Keratoconus Performed Since Introduction of CXL
The frequency of keratoplasty for keratoconus has decreased by more than half at a hospital in Norway since the introduction of CXL, according to a study in Cornea.>1
The investigators compared data from a cohort of patients from the Oslo Hospital University corneal transplant registry from 2005 to 2006 (period 1) and 2013 to 2014 (period 2). Patients in period 1 had surgery before the introduction of CXL treatment, and patients in period 2 had surgery after CXL was well established at the hospital. Patient age and sex were registered, and the Amsler-Krumeich classification system was used to grade the degree of keratoconus.
According to the study, the total number of keratoplasties performed during period 1 was 137, and keratoconus was the cause of surgery in 55 eyes (55 patients). For period 2, the total number of keratoplasties was 231, and keratoconus was the cause of surgery in 26 eyes (26 patients). The difference between the numbers of keratoplasties for keratoconus in the two periods was statistically significant (P=.003). There were no significant differences in the distributions of age and sex between the periods. In period 1, 63.6% of the eyes were graded as stage 4 versus 96.2% in period 2 (P=.001).
“Although we performed more keratoplasty procedures in the last period compared with the first period (231 vs 137), the frequency of keratoplasties in patients with keratoconus was more than halved (reduced from 55 to 26 cases),” the study authors wrote. “There is reason to believe that this reduction is in great part a result of the introduction of CXL. This study is the first to report such a decrease. However, large longitudinal studies in patients with keratoconus are warranted to make definite conclusions about this matter.”
1. Sandvik GF, Thorsrud A, Råen M, et al. Does corneal collagen cross-linking reduce the need for keratoplasties in patients with keratoconus? Cornea. 2015;34(9):991-995.
Harmoni IOL Receives CE Mark
ClarVista Medical received the CE Mark for its Harmoni Modular IOL System for the visual correction of aphakia in adult patients in whom a cataract has been removed, according to a company news release.
The modular design of the IOL is intended to enable safe and easy exchange of the optic component to reduce residual postoperative refractive error at the time of the primary surgery or 3 months postoperatively. In addition to the CE Mark, ClarVista also received ISO 13485 certification, an internationally recognized quality standard for medical devices.
IOL Pioneer Jan Worst, MD, Is Remembered
Femto LDV Z8 Approved by Health Canada
The Femto LDV Z8 (Ziemer Ophthalmic Systems) has been approved by Health Canada, according to a company news release.
The Femto LDV Z8 is a mobile femtosecond laser for refractive, cornea, and cataract procedures. The all-in-one system combines low energy with a high repetition rate to provide a precise capsulotomy and lens fragmentation, the news release said.
Oculus Presents DED Software Module for Keratograph 5M
Oculus introduced the Jenvis Dry Eye Report, which sets new standards in screening for dry eye disease (DED) and was developed for the Oculus Keratograph, according to a company news release.
Founded in 2005, Jenvis is an independent research institute located in Jena, Germany. The institute is directly connected to the Department of Optometry at the University of Applied Sciences in Jena, the news release said. The Jenvis Dry Eye Report supports optometrists and ophthalmologists in diagnosing DED.
A complete Jenvis Dry Eye Report summarizes all of the data from a dry eye questionnaire (eg, Ocular Surface Disease Index and McMonnies scores), a slit-lamp measurement of lid parallel conjunctival folds, and four measurements performed with the Oculus Keratograph 5M. These comprise an analysis of the degree of bulbar redness using R-Scan, measurements of tear meniscus height and tear film breakup time, and meibography. Other commonly used screening methods such as eye blink frequency or lipid layer analysis also run on the Keratograph 5M and can be added, the news release said.
Users can add their company logo to the patient’s printout of the Jenvis Dry Eye Report. The report also contains recommendations specific to the patient and an explanation of abbreviations and technical terms.
Presbia Introduces Disposable Microlens Inserter
Presbia introduced a disposable inserter for the Flexivue Microlens at the Australasian Society of Cataract and Refractive Surgeons in Noosa, Queensland, Australia, according to a company news release.
With its simplified action, the inserter further reduces the time required to complete the Flexivue Microlens procedure. The disposable inserter complements Presbia’s reusable inserter, offering surgeons an option to select the instrument that best suits their needs, the news release said.
Ursapharm, Novaliq Announce European Partnership Agreement
The pharmaceutical company Ursapharm and Novaliq, a clinical stage specialty pharma company with an ophthalmic drug delivery technology platform, announced a partnership agreement as well as the European launch of EvoTears, an over-the-counter treatment for DED, according to a joint news release.
As part of the agreement, Ursapharm obtains the exclusive license for the commercialization of EvoTears, Novaliq’s first commercially available ophthalmic product for the treatment of evaporative DED. Ursapharm’s ophthalmic portfolio includes Hylo Eye Care. The company also offers a variety of ophthalmic antibiotic, antiglaucomatous, and antiinflammatory treatments, the news release said.
–Compiled by Steve Daily, Executive Editor, News; and Callan Navitsky, Senior Editor
CLICKWORTHY
Junior Doctors March Over Contract Dispute
Junior doctors have been marching in protest in London at planned changes to their contracts in England.
www.bbc.com/news/health-34561366
Men’s and Women’s Hearts Age Differently
A study shows that the heart ages differently for women and men, suggesting a possible need for gender-specific treatments.
www.webmd.com/heart/news/20151020/mens-womens-hearts-age-differently
Researchers Develop Drug Delivery Technique to Bypass Blood-Brain Barrier
Researchers have prevented the development of Parkinson disease in a mouse model using new techniques to deliver drugs across the naturally impenetrable blood-brain barrier.
www.sciencedaily.com/releases/2015/10/151020145221.htm
No Amount of Alcohol Safe During Pregnancy, Doctors Say
A leading US pediatricians’ group has issued a new warning that no amount of drinking is safe for a woman while she is pregnant.
consumer.healthday.com/general-health-information-16/alcohol-abuse-news-12/pediatricians-no-alcohol-in-pregnancy-is-safe-704306.html
First Case of Cancer Linked to Fukushima Cleanup Work Diagnosed
Four years after the Fukushima Daichi nuclear plant suffered a catastrophic meltdown, Japan has confirmed the first case of cancer stemming from that work.
www.cnn.com/2015/10/20/asia/japan-fukushima-radiation-cancer/index.html
Letters to the Editor
In the recent article titled The New Ocular Biometers: How Do They Stack Up? (July/August 2015, pg 45; http://crstodayeurope.com/2015/07/the-new-ocular-biometers-how-do-they-stack-up/), the authors stated that, “The Galilei G6 (Ziemer), combining optical A-scan, dual-Scheimpflug imaging, and Placido-disc topography, is reported to measure most ocular elements; however, posterior cornea and corneal aphericities do not appear to be available.” As a long-time user and design contributor of the Galilei, it seems to me that the authors did not have experience with this device, as posterior cornea analysis is available and visible in several ways.
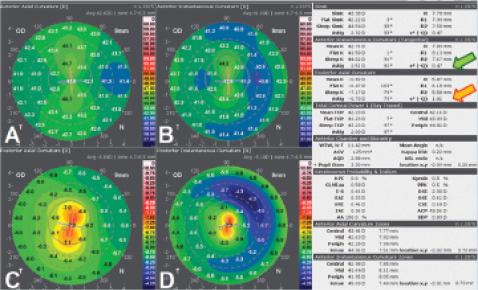
Figure 1. Axial (A and C) and instantaneous (B and D) curvature maps of a cornea with symmetric with-the-rule astigmatism in a normal (q = -0.47; green arrow) aspheric anterior surface (A and B) and symmetric with-the-rule astigmatism in an already steeper than normal (orange steps on maps) hyperprolate (q = -1.82; orange arrow) aspheric posterior surface (C and D).
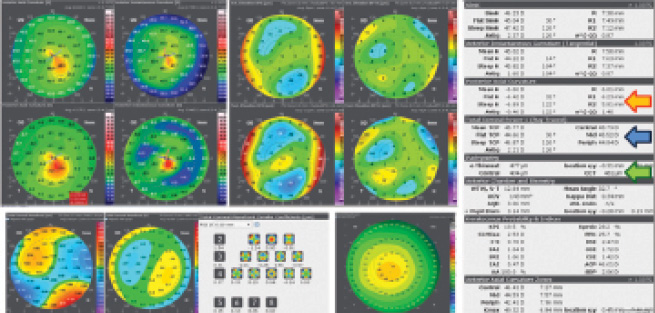
Figure 2. Axial and instantaneous curvature maps (top left), best fit sphere and best fit toric asphere elevation maps (top right), total corneal wavefront and coma maps (bottom left), Zernike pyramid (bottom center), and pachymetry map (bottom right) of the right eye of a young woman with initial keratoconus and a maximum keratometry of 49.32 D. The left cornea was similarly thin but normal. Posterior axial curvature and asphericity (orange arrow), total corneal power (blue arrow), and pachymetry (green arrow) are among the numerical indices.
Since the first Galilei G1, the Alternate Profile I created in 2008 had both the Axial Anterior-Posterior Topography Report and the Instantaneous Anterior-Posterior Topography Report showing posterior curvature maps in diopters with inverted color scales.1 This view of the posterior cornea allowed a simpler comparison with anterior surface maps and faster interpretation of findings (Figure 1).2 Furthermore, all Galilei versions (G1, G2, G4, and G6) have always had available posterior axial and posterior tangential curvature maps, posterior best fit sphere, best fit asphere, and best fit toric asphere elevation maps as well as automatically shown numerical data such as the posterior mean keratometry, flatter and steeper posterior curvature, or posterior cylinder and axis (Figures 1 and 2).
The Galilei G6 is the first optical biometer by interferometry available on the market that includes, without assumptions, a true calculation of the total corneal power by ray tracing and shows the axial length and IOL power calculation3 with links with new ray-tracing IOL formulas, and thickness of the natural lens or an implanted IOL (Figure 3). Reliable total corneal power4 and pachymetry5 were possible because the posterior cornea is detected, assessed, and available in the Galilei.
Multiple studies on this aspect have been published6,7 and presented at international meetings, showing the good repeatability and applications of Galilei posterior corneal surface data. The Charles D. Kelman Innovator’s Lecture at the 2012 ASCRS meeting about corneal optics and toric IOL calculation8 and the Troutman Prize of 2013 about keratoconus screening9 are two of them.
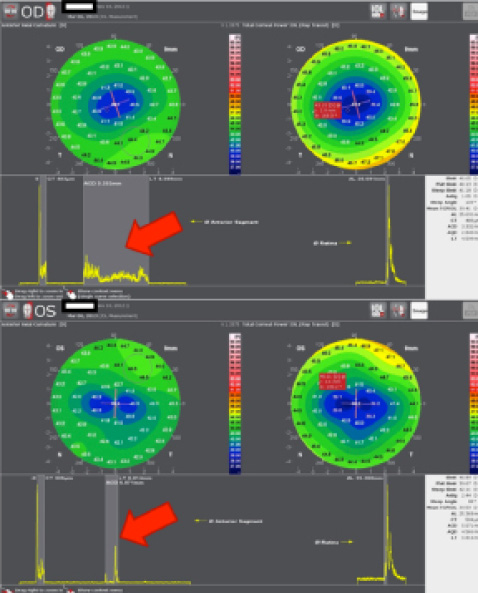
Figure 3. Axial curvature (top left), pachymetry (top right) corneal maps and optical A-scan of both eyes of a woman who underwent myopic LASIK, showing the thickness of her natural lens in the right eye and the position and thickness of the IOL implanted in the left eye.
On the other hand, corneal asphericity is represented numerically by the q factor being q = -e2, where e2 is squared eccentricity. All Galilei versions always calculated and showed these values from both the anterior and the posterior corneal surfaces from an 8-mm-diameter central zone aligned to the vertex (in the G1) or to the first Purkinje (SW 6.0 or newer). Finally, Galilei also determines the anterior, posterior, and total corneal spherical aberration, which is directly related to the q factor (or inverse with the e2; Figures 1 and 2).
Taken together, it is important that physicians understand the scientific and medical benefits of posterior analysis and imaging capabilities on the Galilei. These features offer users an opportunity to make informed treatment choices for patients and ensure that clinical care achieves a level of excellence.
Carlos G. Arce, MD
São Paulo, Brazil
1. Arce CG. Map Interpretation Guide. Software Version 5.2. Ziemer Ophthalmic Systems AG. Port, Switzerland. October 2011.
2. Arce CG. Galilei: Topografia de Plácido e Tomografia do Segmento Anterior com Duplo Scheimpflug. In: Ambrosio Jr R, Chalita MR, Vieira Netto M, Schor P, Chamon W, Machado Fontes B, eds. Wavefront & Topografia, Tomografia e Biomecânica da Córnea. 2nd edition. Rio de Janeiro, Brazil: Cultura Médica; 2013: 165-202.
3. Pajic B, Mueller M, Allemann R, Vestardis I. GALILEI G6 Lens Professional vs. IOLMaster and Lenstar LS900 - A Comparison Study. Ziemer Ophthalmic Systems AG. 2014.
4. Shirayama M, Wang L, Weikert MP, Koch DD. Comparison of corneal powers obtained from 4 different devices. Am J Ophthalmol. 2009;148(4):528-535.
5. Ladi JS, Shah NA. Comparison of central corneal thickness measurements with the Galilei dual Scheimpflug analyzer and ultrasound pachymetry. Indian J Ophthalmol. 2010;58(5):385-388.
6. Savini G, Carbonelli M, Barboni P, Hoffer KJ. Repeatability of automatic measurements perfomed by a dual Scheimpflug analyzer in unoperated and postrefractive surgery eyes. J Cataract Refract Surg. 2011;37:302-309.
7. Fahd DC, Cherfan CG, Raad C, Asouad M, Awwad ST. Assessment of anterior and posterior corneal indices using two Scheimpflug analyzers. Arq Bras Oftalmol. 2014;77:17-20.
8. Koch DD, Ali SF, Weikert MP, Shirayama M, Jenkins R, Wan GL. Contribution of posterior corneal astigmatism to total corneal astigmatism. J Cataract Refract Surg. 2012;38(12):2080-2087.
9. Smadja D, Santhiago MR, Mello GR, Krueger RR, Colin J, Touboul D. Influence of the reference surface shape for discriminating between normal corneas, subclinical keratoconus, and keratoconus. J Refract Surg. 2013;29:274-281.
Dear Dr. Arce,
Thank you for your letter highlighting the blatant error in our article. You are absolutely correct that the statement made by us regarding the Galilei G6 combining optical A-scan, dual-Scheimpflug imaging, and Placido-disc topography omitting to measure posterior cornea and corneal asphericities is simply incorrect.
We are unsure how this error occurred, as the entire team is aware of the capabilities of the Galilei and the work done by Douglas D. Koch, MD, and his team on the posterior corneal toricity and its impact on total corneal toricity using this device. Thank you for your feedback and setting the record straight for our readers. This error should never have occurred, and we collectively apologize for it.
Arthur B. Cummings, MB ChB, FCS(SA), MMed(Ophth), FRCS(Edin)
Dublin, Ireland Michael Mrochen, PhD
Zürich, Switzerland
We have received some calls and emails from surgeons regarding an article published in the October issue, Ones to Watch: Premium IOL Technologies, written by Jorge L. Alió, MD, PhD (pg 33; http://crstodayeurope.com/2015/09/ones-to-watch-premium-iol-technologies/).
In the article, Reviol, which is actually a diffractive bifocal lens, was listed as a refractive IOL. As it is well known today, refractive IOLs are considered an old technology, and many models have been discontinued in the past due to neural adaptation problems and severe patient complaints. In this regard, we would like to remark again that Reviol is established on the principles of diffractive IOL technology, which is so-called active diffractive optics. The IOL is fully pupil independent, provides maximum light transmission, has unique light distribution structure, and gives a high rate of spectacle independence.
Further, Professor Alió did not mention the Tri-ED trifocal IOL in his discussion on trifocal lens technologies. The Tri-ED IOL combines trifocal diffractive technology and extended depth of focus vision, which is the first type of IOL on the market to provide continuous vision under all light conditions. n
Fatih Ergin, PhD
Product Manager, VSY Biotechnology
CRST Europe regrets this error and this omission.
