
Although the anterior segment of the eye is one of the most readily accessible organs of the body, drug delivery to ocular tissues is particularly problematic, as reflected by the notoriously poor bioavailability of topical ocular drug formulations of 5% or less. A standard drop of water of approximately 40 to 50 μL will activate the blinking reflex, which washes away the majority of any topically administered dose within 15 to 30 seconds after instillation.1
AT A GLANCE
• Drug delivery to ocular tissues is problematic, as reflected by the notoriously poor bioavailability of topical ocular drug formulations of 5% or less. To further complicate matters, up to 75% of new chemical entities are considered poorly soluble, even for oral administration.
• The three major issues of safety, bioavailability, and stability must be be addressed for successful ocular formulations. EyeSol, a new ocular drug delivery technology, fulfills these requirements and is based on a semifluorinated alkane platform (F4H5).
• Novaliq is developing a broad portfolio of products across multiple segments based on this concept.
To further complicate matters, up to 75% of new chemical entities are considered poorly soluble, even for oral administration.2-4 In short, a drug is considered poorly soluble if the required dose cannot be dissolved in 250 mL of aqueous medium, and, at 40 to 50 µL, an aqueous eye drop has 5,000 times less volume. This exacerbates the solubility problem by several orders of magnitude.
Three major issues must be addressed for successful ocular formulations: safety, bioavailability, and stability. EyeSol, a new ocular drug delivery technology developed by the German pharmaceutical company Novaliq, fulfills these requirements. EyeSol is based on a semifluorinated alkane (SFA) platform, specifically perfluorobutylpentane (F4H5). SFA is a special class of fluorocarbon compounds that have been used for more than a decade in the posterior segment of eyes in thousands of patients and that have an excellent safety profile.5,6
F4H5 is a colorless liquid with high vapor pressure, and it is physically, chemically, and physiologically inert with no catabolism or metabolism in the human body. Additionally, low surface and interface tension permits dissipation of the drug within minutes from the ocular surface.
The low viscosity and surface tension of SFAs result in droplets of around 15 μL—approximately one-third the size of standard aqueous drops. Thus, SFAs avoid the spillover effect and the subsequent loss of most of the administered dose, which increases their bioavailability. Further, the refractive index of SFAs is similar to that of water, so these formulations do not impair patients’ vision as emulsions and oily drops tend to do.
Due to their amphiphilic nature, SFAs dissolve a number of therapeutically relevant, poorly water-soluble compounds such as cyclosporine A and tacrolimus. Moreover, the aqueous-free environment of this delivery system increases the stability of these drugs by preventing oxidation and hydrolysis.
THE PRODUCT PORTFOLIO
Novaliq is currently developing a broad portfolio of products (Figure 1) across multiple segments, including dry eye disease (DED), glaucoma, and retina. In October 2015, along with its European partner Ursapharm, Novaliq brought to market its first product, EvoTears (NovaTears in the United States), an over-the-counter eye drop for the treatment of mild to moderate evaporative DED. In a recent study,7 EvoTears was found to significantly improve four of five measures associated with evaporative DED. The product was also shown to be safe and well tolerated, and it caused no changes in visual acuity or IOP.7 A mean 21-point decrease of Ocular Surface Disease Index scores exceeded minimal, clinically important differences for mild or moderate and severe disease. A follow-up to this product, EvoTears with omega-3, has entered the approval phase. It is anticipated that EvoTears with omega-3 will receive the CE Mark this year.
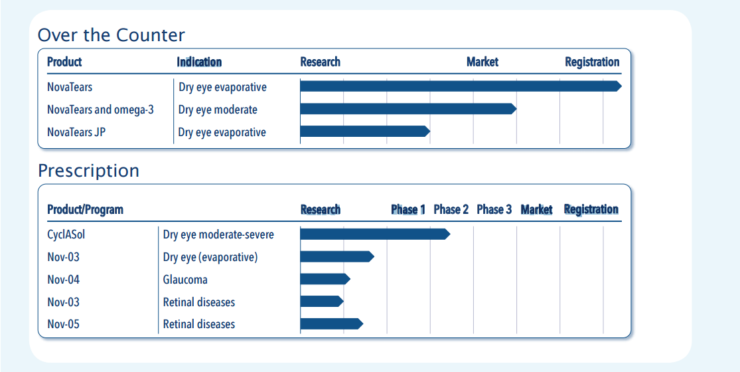
Figure 1. Novaliq’s ophthalmic pipeline.
Novaliq’s prescription portfolio includes CyclASol, a cyclosporin A 0.05% solution formulation in the SFA vehicle that has been granted intellectual property position in the European Union and United States, among other major markets.
CONCLUSION
Novaliq’s technology offers a unique combination of physicochemical properties, including excellent spreading behavior, physical and chemical inertness, and solubility of poorly water-soluble compounds. Additionally, the lower drop volume reduces the blinking reflex and enhances topical drop delivery. The absence of surfactants and irritating preservatives and the avoidance of blurry vision associated with emulsions combine to improve the tolerability and convenience of these nonaqueous products.7
1. Peyman GA, Schulman JA, Sullivan B. Perfluorocarbon liquids in ophthalmology. Surv Ophthalmol. 1995;39(5):375-395.
2. The Food and Drug Administration. The Biopharmaceutics Classification System (BCS) Guidance. April 2009.
http://tinyurl.com/hrlopzh. Accessed February 9, 2016.
3. Löbenberg R, Amidon G. L. Modern bioavailability, bioequivalence and biopharmaceutics classification system. New scientific approaches to international regulatory standards. Eur J Pharm Biopharm. 2000;50(1):3-12.
4. Scherer D, Reidelshöfer A. Softgel capsules solve economic and safety issues posed by poorly soluble anticancer drugs. Pharm Ind. 2012;5(74):745-749.
5. Kirchhof B, Wong D, Van Meurs J, et al. Use of perfluorohexyloctane as a long-term internal tamponade agent in complicated retinal detachment surgery. Am J Ophthalmol. 2002;133(1):95-101.
6. Meinert H, Roy T. Semifluorinated alkanes—a new class of compounds with outstanding properties for use in ophthalmology. Eur J Ophthalmol. 2000;10(3):189-197.
7. Steven P, Scherer D, Krösser S, et al. Semifluorinated alkane eye drops for treatment of dry eye disease—a prospective, multicenter noninterventional study. J Ocul Pharmacol Ther. 2015;31(8):498-503.
Michael O’Rourke
• President and founder, Scotia Vision Consultants
• scotiavc@gmail.com
• Financial disclosure: Strategic advisor (Novaliq)

