
CXL is a treatment modality combining UV-A light and the chromophore riboflavin to arrest the progression of keratoconus, pellucid marginal degeneration, and postoperative ectasia.1-4 The biomechanical effect induced by CXL can be detected in a number of ways. Indirectly, topography can demonstrate the arrest of ectasia progression. High-frequency dynamic Scheimpflug imaging (CorVis; Oculus) and the Ocular Response Analyzer (Reichert Technologies) can also, to a certain extent, detect these changes.5,6 Finally, the depth of the demarcation line after CXL is seen by many as a tool to assess the depth of effective crosslinks during CXL.7,8
AT A GLANCE
• Although the main goal of CXL is to arrest ectasia progression, the patient’s biggest concern is his or her quality of vision.
• The corneas of keratoconus patients do not react uniformly to the stimuli provided by light and chromophore in CXL treatment.
• In the ongoing debate about whether to perform PRK simultaneous with or sequential to CXL, the author prefers the sequential approach; due to the 7% rate of failure with standard CXL, simultaneous PRK may have deleterious effects.
Since the introduction of the Dresden protocol for CXL, an abundance of supposedly more sophisticated CXL protocols have been introduced into the field, causing confusion. These approaches, including accelerated CXL, pulsed CXL, contact lens–assisted CXL, epithelium-on (epi-on) CXL, do provide some biomechanical effect; ultimately, however, the biomechanical effect of these protocols is still inferior to that of epithelium-off (epi-off) CXL.9-14 Our research group has argued that the primary reason for this is a relative lack of oxygen availability with these and other new protocols.15,16 Therefore, the most widely accepted CXL protocol to date is a variant of the classic Dresden protocol: epi-off irradiation with 9 mW/cm2 for 10 minutes, using continuous light.
VISUAL REHABILITATION THROUGH CUSTOMIZED PRK
The main goal of the CXL procedure is to arrest ectasia progression. However, the patient’s biggest concern is his or her quality of vision. A number of procedures may be envisaged for the visual rehabilitation of patients after CXL, such as adaptation of keratoconus contact lenses, implantation of intrastromal corneal ring segments, implantation of phakic IOLs for the symmetric portion of the refractive error, and topography-guided or corneal wavefront-guided PRK. The concept behind each of these procedures varies. This article focuses on customized surface ablation for the treatment of keratoconus.
The principal idea is to use corneal wavefront data to calculate and apply an ablation profile customized for each patient. The main goal of this is to improve BCVA so that the patient may alternate more easily between glasses and contact lenses. The patient must clearly understand the goal of the procedure from the start in order to reduce expectations that the laser procedure will remove his or her refractive error entirely and completely even out the asymmetry of the cornea.
Based on the proposition of Kanellopoulos and colleagues, there is now consensus in removing no more than 50 µm of corneal stroma during this laser procedure.17,18 To remain within this ablation limit, a number of considerations must be taken, including the following:
- What target refraction should be chosen to avoid a too high level of anisometropia when the patient is wearing glasses;
- What size optical zone should be chosen, based on the location of the apex of the cone, the patient’s age, the patient’s pupil size, and the overall thickness of the cornea; and
- What other factors may influence the biomechanical integrity of the cornea in a positive (ie, smoking) or negative (ie, pregnancy, hypothyroidism) manner.19-22
Additionally, for planning the ablation profile, some laser platforms allow users to switch on and off the correction of individual Zernike coefficients. With these platforms, the surgeon may choose to correct certain essential Zernike polynomials, such as coma, and omit others that are less relevant for the sake of tissue sparing.
NOT ALL CORNEAS REACT THE SAME WAY
The human body does not react uniformly to an external stimulus, such as CXL. Generally, the body responds in a Gaussian distribution (Bell curve). Although most patients react the way we expect, others may react either excessively or not at all (Figure 1). The corneas of keratoconus patients are no different: They do not react uniformly to the stimuli provided by light plus chromophore in CXL treatment. In most patients, this treatment simply stabilizes their keratometry (K) readings or shows a moderate flattening effect of 2.00 to 3.00 D on average, extending over months to years.23
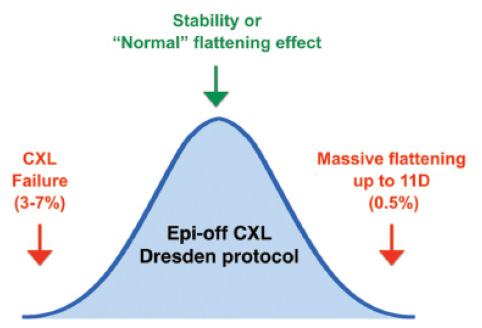
Figure 1. Gaussian distribution of outcomes after Dresden protocol epi-off CXL procedure. Whereas approximately 60% of all cases show either stability or a normal flattening effect with a mean of 2.70 D, failure to arrest ectasia or extreme flattening may be observed in 3% to 7% and 0.5% of cases, respectively.
In CXL, a lack of reaction occurs when there is a failure to stabilize the progressive irregular increase in corneal curvature. Even with the classic Dresden epi-off CXL approach of applying 30 minutes of irradiation at 3 mW/cm2, failure occurs in 7% of cases.24 When epi-on protocols are used, the failure rate increases to more than 20%.25
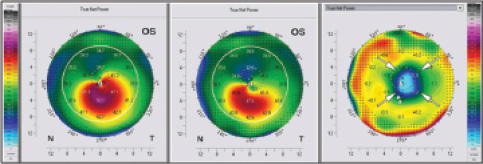
Figure 2. Example of massive flattening following CXL. Scheimpflug analysis of the anterior corneal surface (true net power) before (A) and at 12 months after (B) CXL. The difference image (C) shows a strong reduction of maximum keratometry values of up to 9.00 D.
Other patients overreact to CXL. In an analysis of our first 1,000 cases, we observed an extreme flattening effect in approximately 0.5% of all patients. This flattening effect could be observed just a few weeks after the procedure. In extreme cases, the cornea flattened by up to 11.00 D (Figure 2).26-28
Additionally, the spatial and temporal extent of the healing reaction (haze) may vary substantially between individuals. Whereas haze might be barely detectable in some patients, it can be at debilitating levels in others.
SIMULTANEOUS OR SEQUENTIAl?
There is ongoing debate about whether PRK should be performed simultaneous with or sequential to CXL. Table 1 provides an overview of the benefits and drawbacks of each approach, in my view. Our best practice model is to perform epi-off CXL and wait 12 months to assess the patient’s cornea. The assessment is based on whether the cornea is: (A) stable,
(B) flattening, (C) flattening in an extreme way, or (D) not responding to the CXL procedure. In scenario A, we proceed to perform a sequential approach. In scenarios B and C, we perform more routine clinical controls until the K readings of the cornea have stabilized. In scenario D, we perform a second CXL procedure.
In performing a sequential approach, the surgeon first needs to know whether the excimer laser energy levels used in healthy eyes must be adjusted to accommodate for the increased crosslinks in a CXL-treated cornea. Both the Seiler research group and our group have determined the adjustment in excimer pulse energy needed on different laser platforms. We have both determined that an increase in laser energy of 9% and 12%, respectively, is needed for crosslinked compared with noncrosslinked stroma.29,30
CONCLUSION
We prefer the sequential approach to PRK visual rehabilitation after CXL, mainly because of the failure rate of 3% to 7% associated with the simultaneous approach. This rate is not negligible, and neither is the risk of extreme flattening occurring in 0.5% of patients. However, there are exceptions to this treatment protocol. In two circumstances in particular, we use the simultaneous approach: (1) when the patient’s visual quality is too poor for daily activities, and (2) in the case of contact lens intolerance, when glasses are the only option for the patient.
1. Hafezi F, Kanellopoulos J, Wiltfang R, Seiler T. Corneal collagen crosslinking with riboflavin and ultraviolet A to treat induced keratectasia after laser in situ keratomileusis. J Cataract Refract Surg. 2007;33(12):2035-2040.
2. Raiskup F, Theuring A, Pillunat LE, and Spoerl E. Corneal collagen crosslinking with riboflavin and ultraviolet-A light in progressive keratoconus: ten-year results. J Cataract Refract Surg. 2015;41(1):41-46.
3. Richoz O, Mavrakanas N, Pajic B, Hafezi F. Corneal collagen cross-linking for ectasia after LASIK and photorefractive keratectomy: long-term results. Ophthalmology. 2013;120(7):1354-1359.
4. Wollensak G, Spoerl E, Seiler T. Riboflavin/ultraviolet-A-induced collagen crosslinking for the treatment of keratoconus. Am J Ophthalmol. 2003;135(5):620-627.
5. Spoerl E, Terai N, Scholz F, Raiskup F, Pillunat LE. Detection of biomechanical changes after corneal cross-linking using Ocular Response Analyzer software. J Refract Surg. 2011;27(6):452-457.
6. Steinberg J, Frings A, Mousli A, et al. New Scheimpflug dynamic in vivo curve analyses to characterize biomechanical changes of the cornea after cross-linking for progressive keratoconus. J Refract Surg. 2016;32(1):34-39.
7. Peyman A, Nouralishahi A, Hafezi F, Kling S, Peyman M. Stromal demarcation line in pulsed versus continuous light accelerated corneal cross-linking for keratoconus. J Refract Surg. 2016; 32(3):206-208.
8. Seiler T, Hafezi F. Corneal cross-linking-induced stromal demarcation line. Cornea. 2006;25(9):1057-1059.
9. Brittingham S, Tappeiner C, Frueh BE. Corneal cross-linking in keratoconus using the standard and rapid treatment protocol: differences in demarcation line and 12 month outcomes. Invest Ophthalmol Vis Sci. 2014;54(12):8371-8376.
10. Gatzioufas Z, Richoz O, Brugnoli E, Hafezi F. Safety profile of high-fluence corneal collagen cross-linking for progressive keratoconus: preliminary results from a prospective cohort study. J Refract Surg. 2013;29(12):846-848.
11. Hammer A, Richoz O, Mosquera S, Tabibian D, Hoogewoud F, Hafezi F. Corneal biomechanical properties at different corneal collagen cross-linking (CXL) irradiances. Invest Ophthalmol Vis Sci. 2014;55(5):2881-2884.
12. Kymionis GD, Tsoulnaras KI, Grentzelos MA, et al. Corneal stroma demarcation line after standard and high-intensity collagen crosslinking determined with anterior segment optical coherence tomography. J Cataract Refract Surg. 2014;40(5):736-740.
13. Vinciguerra P. The efficacy of corneal cross-linking shows a sudden decrease with very high intensity UV light and short treatment time. Invest Ophthalmol Vis Sci. 2013;54(2):1181.
14. Wernli J, Schumacher S, Spoerl E, Mrochen M. The efficacy of corneal cross-linking shows a sudden decrease with very high intensity UV light and short treatment time. Invest Ophthalmol Vis Sci. 2013;54(2):1176-1180.
15. Richoz O, Hammer A, Tabibian D, Gatzioufas Z, Hafezi F. The biomechanical effect of corneal collagen cross-linking (CXL) with riboflavin and UV-A is oxygen dependent. Transl Vis Sci Technol. 2013;2(7):6.
16. Kling S, Richoz O, Hammer A, et al. Increased biomechanical efficacy of corneal cross-linking in thin corneas due to higher oxygen availability. J Refract Surg. 2015;31(12):840-846.
17. Kanellopoulos AJ, Binder PS. Collagen cross-linking (CCL) with sequential topography-guided PRK: a temporizing alternative for keratoconus to penetrating keratoplasty. Cornea. 2007;26(7):891-895.
18. Kanellopoulos AJ. Comparison of sequential vs same-day simultaneous collagen cross-linking and topography-guided PRK for treatment of keratoconus. J Refract Surg. 2009;25(9): S812-S818.
19. Gatzioufas Z, Thanos S. Acute keratoconus induced by hypothyroxinemia during pregnancy. J Endocrinol Invest. 2008;31(3):262-266.
20. Hafezi F. Smoking and corneal biomechanics. Ophthalmology. 2009;116(11):2259.e1.
21. Hafezi F and Iseli HP. Pregnancy-related exacerbation of iatrogenic keratectasia despite corneal collagen crosslinking. J Cataract Refract Surg. 2008;34(7):1219-1221.
22. Hafezi F, Koller T, Derhartunian V, Seiler T. Pregnancy may trigger late onset of keratectasia after LASIK. J Refract Surg. 2012;28(4):242-243.
23. Raiskup-Wolf F, Hoyer A, Spoerl E, Pillunat LE. Collagen crosslinking with riboflavin and ultraviolet-A light in keratoconus: long-term results. J Cataract Refract Surg. 2008;34(5):796-801.
24. Koller T, Mrochen M, Seiler T. Complication and failure rates after corneal crosslinking. J Cataract Refract Surg. 2009;35(8):1358-1362.
25. Soeters N, Wisse RP, Godefrooij DA, Imhof SM, Tahzib NG. Transepithelial versus epithelium-off corneal cross-linking for the treatment of progressive keratoconus: a randomized controlled trial. Am J Ophthalmol. 2015;159(5):821-828.e3.
26. Hafezi F, Koller T, Vinciguerra P, Seiler T. Marked remodelling of the anterior corneal surface following collagen cross-linking with riboflavin and UVA. Br J Ophthalmol. 2010;95(8):1171-1172.
27. Koller T, Pajic B, Vinciguerra P, Seiler T. Flattening of the cornea after collagen crosslinking for keratoconus. J Cataract Refract Surg. 2011;37(8):1488-1492.
28. Santhiago MR, Giacomin NT, Medeiros CS, Smadja D, Bechara SJ. Intense early flattening after corneal collagen cross-linking. J Refract Surg. 2015;31(6):419-422.
29. Chen S, Li Y, Stojanovic A, et al. Evaluation of the efficacy of excimer laser ablation of cross-linked porcine cornea. PloS one. 2012;7(10):e46232.
30. Richoz O, Arba Mosquera S, Kling S, et al. Determination of the excimer laser ablation rate in previously cross-linked corneas. J Refract Surg. 2014;30(9):628-632.
Farhad Hafezi, MD, PhD
• Medical Director, The ELZA Institute, Dietikon/Zurich, Switzerland
• Professor of Ophthalmology, University of Geneva, Switzerland
• Clinical Professor of Ophthalmology, Keck School of Medicine, University of Southern California, Los Angeles
• Research Group Leader, Center for Applied Biotechnology and Molecular Medicine, University of Zurich, Switzerland
• farhad@hafezi.ch
• Financial disclosure: Chief Technology Officer (EMAGine SA, a company developing CXL technology at the slit lamp); author of patents related to CXL technology

