


Modern approaches to cataract surgery have been confirmed to be effective and safe in numerous clinical scenarios.1,2 Unfortunately, in the event of IOL explantation, whether in the early or late postoperative period, there are many difficult issues, and no single best method is accepted by all cataract and vitreoretinal surgeons. Reasons for IOL explantation or exchange surgery include decentration, subluxation of the full complex of the IOL and capsular bag, refractive error, IOL opacification, and corneal endothelial cell decompensation.
AT A GLANCE
- As an alternative to mechanical dissection of an IOL for explantation, the femtosecond laser can be used to partially or fully dissect the IOL according to a chosen profile pattern programed into the software. This process is called IOL transection.
- Laser settings must be adjusted according to the material class of the IOL to be transected.
- Low-energy femtosecond laser transection has a small but significant salutary effect on the performance of IOL explantation and is applicable in a wide range of clinical situations.
Numerous surgical techniques have been described for mechanical division of the IOL to avoid the need for a large incision; these include crisscross lensotomy, bisection, trisection, and removal of the optic only (Figure 1).3-7 All of these techniques are effective, but excessive manipulation within the confines of the anterior segment can quickly lead to intraoperative difficulties or prolonged recovery after IOL dissection.
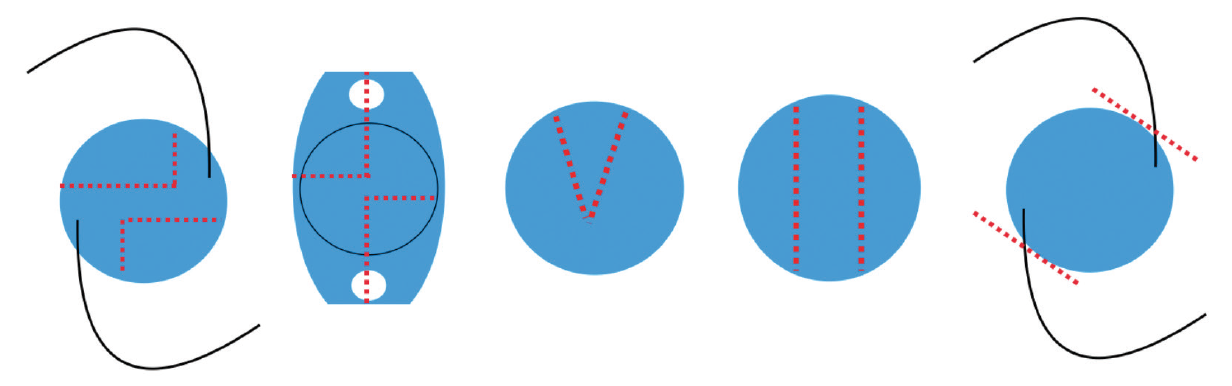
Figure 1. Schematic representation of different surgical methods of IOL disassembly for explantation: crisscross lensotomy (left and second from left); bisection (third from left); trisection (fourth from left); and removal of the optic only (right).
The biomechanical and physical properties of IOLs vary. A high resistance to deformation can make mechanical IOL separation difficult. Unexpected trauma to surrounding tissues can result from the IOL slipping from between the jaws of scissors or rotating during dissection. Unnecessary tissue damage can also occur from the manipulation of the microsurgical instruments themselves.
Since 2009, surgeons have used femtosecond laser technology to assist with or replace some of the mechanical steps in cataract surgery.8 More recently, femtosecond laser transection of the lens has been proposed as an alternative method for IOL removal.9,10 In our experience, we believe that this technology has the potential to replace manual techniques for routine IOL explantation.
LOW-ENERGY IOL TRANSECTION
The term transection characterizes the ability of the femtosecond laser platform to partially or fully dissect the IOL according to the chosen profile pattern programed into the software. (It should be noted that this is an off-label use of ophthalmic femtosecond laser devices.) Although the laser is adaptable to individual surgical technique preferences, there remain limitations.
Compared with mechanical IOL separation with microsurgical scissors, femtosecond laser IOL transection is a no-touch technology that optimizes the force that must be applied to corneal incisions for IOL section removal. Surgically induced astigmatism is affected by the stretching of the corneal stroma and the size of the incision used to explant IOL sections. The simplicity of intraocular manipulations afforded by in situ transection is key to rapid recovery and successful outcomes after explantation.
Fortunately, femtosecond IOL transection is feasible at low energy parameters that are optimal for safe, precise, and reproducible IOL separation.11
IOL MATERIAL AFFECTS TRANSECTION
Laser settings must be adjusted according to the material class of the IOL to be transected. The most frequently explanted IOLs can be classified into three groups based on their ability to be transected. Our unpublished data, based on in vitro investigations conducted prior to clinical application, indicates the following:
Type 1 IOLs. Soft hydrophilic and silicone-based IOLs are easily transected with minimal femtosecond laser parameters requiring little effort for mechanical separation.
Type 2 IOLs. Hydrophobic acrylic IOLs require a more significant mechanical separation force after femtosecond laser transection (Figure 2).
Type 3 IOLs. In hard, rigid PMMA IOLs, femtosecond laser transection is ineffective or does not meet the requirements of clinical safety.
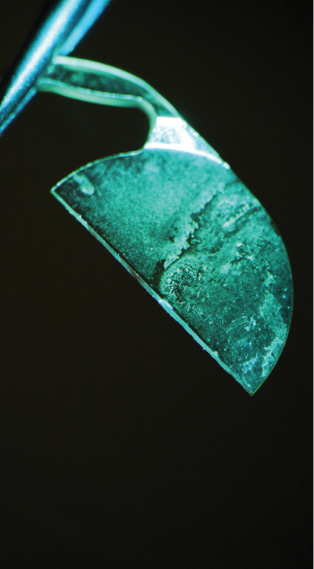
Figure 2. Slit-lamp photograph of hydrophobic acrylic IOL fragment with standard green filter after femtosecond laser low energy transection (bisection) and explantation surgery.
TIPS FOR SUCCESSFUL TRANSECTION
All clinical scenarios require a sterile environment for the femtosecond laser, as docking must take place after the incision is made and the anterior chamber is entered. If the IOL is located in the capsular bag with sufficient zonular support, the lens should be freed from the bag and brought to the iris plane or into the anterior chamber after injection of an OVD. Certain properties define cohesive OVDs as an excellent choice for stabilizing the IOL position: They are more viscous than dispersive OVDs and have higher molecular weights and longer molecular chains.
Horizontal placement of the IOL is crucial, as the profile of the laser cut should cover the IOL only and not the space in which the OVD resides. If the laser exposure interacts with the water molecules of the OVD, gas formation is inevitable. If it forms under the IOL, the lens is pulled up by the tendency of these bubbles to migrate superiorly. This micromotion of the lens leads to incomplete transection, as its superficial layers are pulled out of the profile of the laser cut.
The steps of surgery are described here. After sufficient mydriasis to bring the pupil to approximately the size of the IOL optic size plus 1 mm, a cohesive OVD (Provisc; Alcon) is injected into the anterior chamber and just under the IOL into the capsular bag. The IOL is removed from the bag, lysing all adhesions, and lifted above the anterior capsule. The IOL is positioned either in the pupil plane or in the anterior chamber. The anterior capsule is checked to be certain of its separation from the IOL. The corneal wound is ascertained to be watertight or secured with a single suture.
After the docking procedure is done, the profile of the laser cut is positioned in relation to the pupil diameter and the anterior capsule to allow an adequate safety zone. The length of the laser cut is adjusted to slightly bigger than the optic size. For hydrophobic acrylic IOLs, 6 µJ of energy is selected to avoid excessive alteration of the IOL surface but to be enough to complete IOL separation.
Spot and layer separation size is 7 µm; this size is chosen to avoid spot overlap, which is undesirable and unnecessary. Overlap multiplies the energy impact, causing charring of lens material and exfoliation of chemical products released by pyrolysis. The expansion of gas disseminates the exfoliated material, causing it to be trapped in the corneal wound during irrigation and aspiration. The IOL is then half-transected. (The patterns described here can be changed according to individual preferences and surgical technique.)
The patient interface is then removed, and the patient is switched back under the microscope while sterility is maintained and the anterior chamber is confirmed to be formed. The peripheral part of the IOL optic is grasped with two 25-gauge end-gripping microforceps (Grieshaber revolution DSP ILM forceps; Alcon/Grieshaber) through the preexisting paracenteses and fragmented into two parts by smoothly pulling in a direction perpendicular to the transection line.
The IOL parts are subsequently removed from the anterior chamber though the main incision. After the replacement IOL is implanted, bimanual irrigation and aspiration of the anterior and posterior chambers is done in routine fashion, removing OVD and any cavitation gas bubbles left after laser transection. Finally, the corneal incisions are ascertained to be watertight.
FUTURE DIRECTION
This technique is a novel use of femtosecond laser technology, and its safety with regard to biocompatibility of exfoliated material from laser-lens material interaction requires evaluation by gas quantitative analysis. More trials are needed to determine its clinical efficacy.
Low-energy femtosecond laser transection has a small but significant salutary effect on the performance of IOL explantation and is applicable in a wide range of clinical situations. This technique is an example of the trend toward minimally invasive surgical intervention. Automated IOL separation can decrease ocular trauma and surgical time, resulting in less postoperative inflammation and faster patient recovery.
1. Lundström M. Barry P, Henry Y, Rosen P, Stenevi U. Visual outcome of cataract surgery; study from the European Registry of Quality Outcomes for Cataract and Refractive Surgery. J Cataract Refract Surg. 2013;39(5):673-679.
2. Wang SY, Stem MS, Oren G, Shtein R, Lichter PR. Patient-centered and visual quality outcomes of premium cataract surgery: a systematic review. Eur J Ophthalmol. 2017;27(4):387-401.
3. Osher RH. Crisscross lensotomy: New explantation technique. J Cataract Refract Surg. 2006;32(3):386-388.
4. Karamaounas N, Kourkoutas D, Prekates C. Surgical technique for small-incision intraocular lens exchange. J Cataract Refract Surg. 2009;35(7):1146-1149.
5. Por YM, Chee SP. Trisection technique: a 2-snip approach to intraocular lens explanation. J Cataract Refract Surg. 2007;33(7):1151-1154.
6. Gekka T, Ogawa T, Ohkuma Y, Katagiri S, Shiba T, Tsuneoka H. Trisection technique for the extraction of dislocated intraocular lenses through a small surgical incision. J Cataract Refract Surg. 2015;41(10):2040-2042.
7. Lee SJ, Sun HJ, Choi KS, Park SH. Intraocular lens exchange with removal of the optic only. J Cataract Refract Surg. 2009;35(3):514-518.
8. Nagy Z, Takacs A, Filkorn T, Sarayba M. Initial clinical evaluation of an intraocular femtosecond laser in cataract surgery. J Refract Surg. 2009;25(12):1053-1060.
9. Bala C, Shi J, Meades K. Intraocular lens fragmentation using femtosecond laser: an in vitro study. Transl Vis Sci Technol. 2015;4(3):8.
10. Jonathan C. Lake, MD; Celso Boianovsky, MD; Mário J. Carvalho, MD; Francisco S. Soares, MD. Femtosecond laser-assisted IOL removal. Presented at ASCRS ASOA Symposium and Congress; Feb. 6-10, 2016; New Orleans, USA.
11. Anisimova NS, Malyugin BE, Arbisser LB, et al. Femtosecond laser–assisted intraocular lens fragmentation: low energy transection. J Refract Surg. 2017;33(9):646-648.



