Dr. Nawani talks through how he managed several incision-related complications.
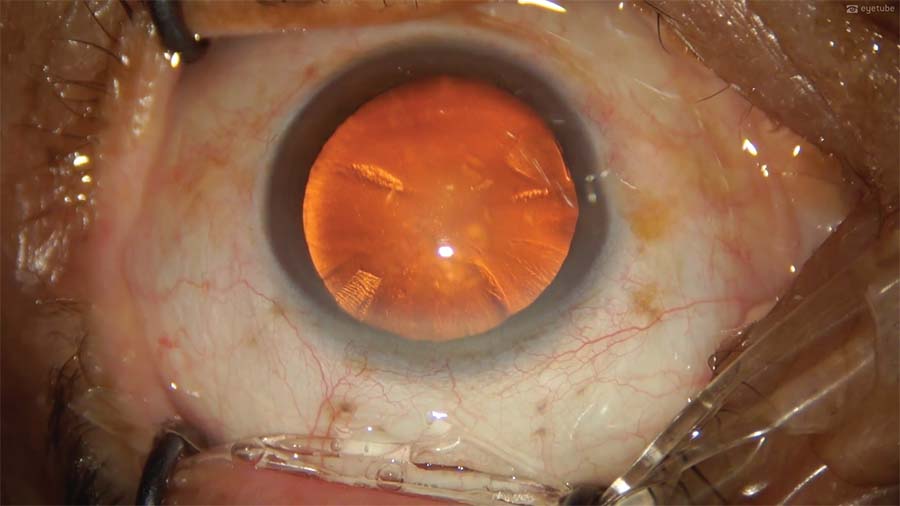
1. Two sideport incisions are created. During the second, the blade enters the cornea tangentially but does not penetrate the anterior chamber. This creates a long intracorneal track and only partial entry into the capsular bag.
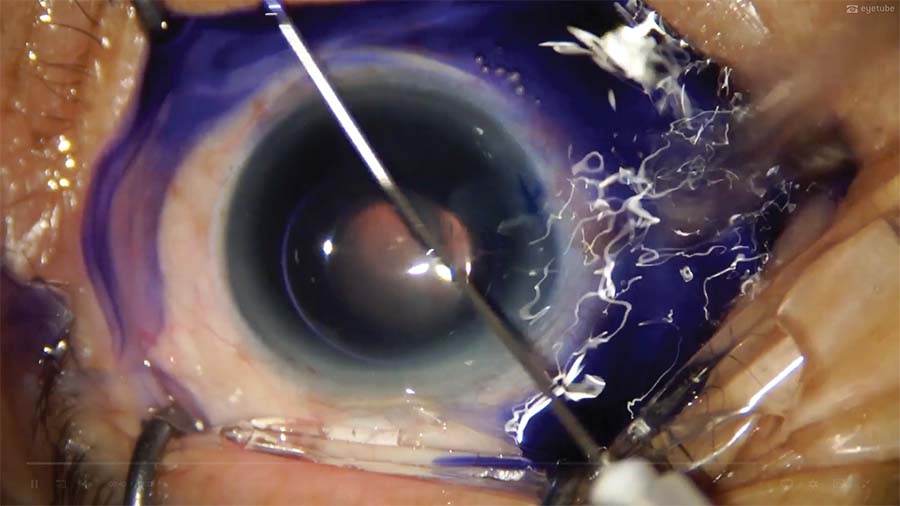
2. The anterior corneal surface is massaged while the capsule is stained with trypan blue dye under an air bubble. This allows the air bubble to spread the dye evenly over the anterior capsule.
3. The problem with the second sideport incision is revisited. Countertraction is applied, and the blade enters the sideport incision from the same place but on a deeper plane.
4. A triplanar 2.8-mm clear corneal incision is attempted. After the first plane is created, however, the blade slides forward, creating a slightly anterior corneal incision and a long intracorneal tunnel.
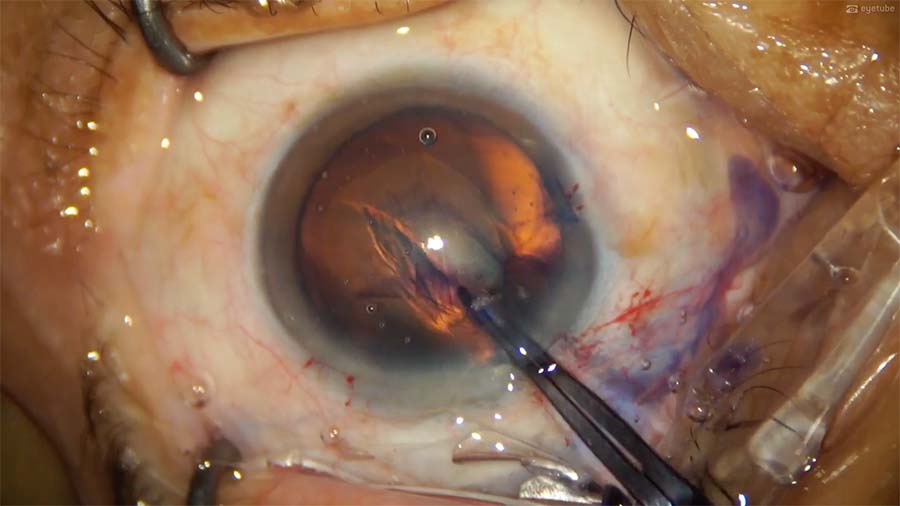
5. The anterior chamber is refilled with hydroxypropyl methylcellulose 2%, and a capsular flap is lifted with a 26-gauge bent cystotome. Utrata forceps are used to grasp the flap and initiate the capsulorhexis. Control of the capsulorhexis is poor, however, and the tear extends toward the temporal side.
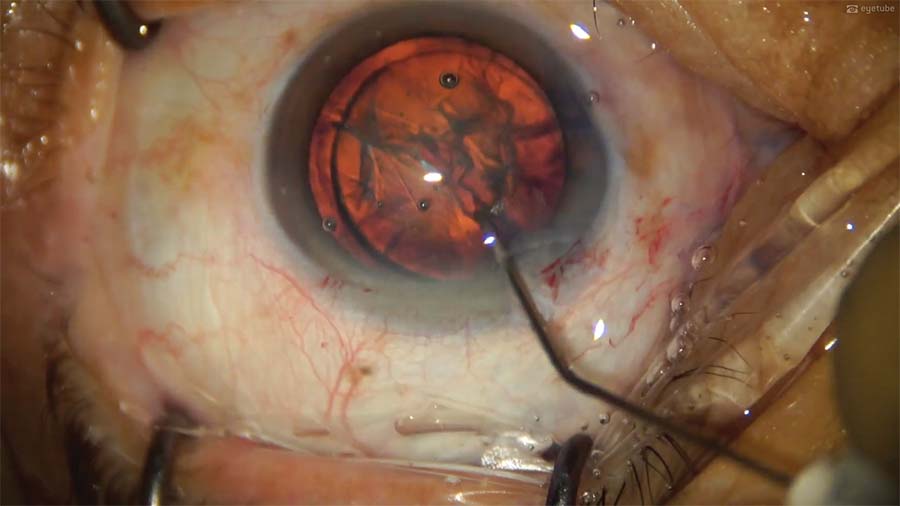
6. Hydrodelineation and hydrodissection are used to free the nucleus from its corticocapsular adhesions and rotate it into a better position. The pros and cons of performing phacoemulsification through the anterior corneal incision are weighed; in the end, superotemporal phacoemulsification is performed.
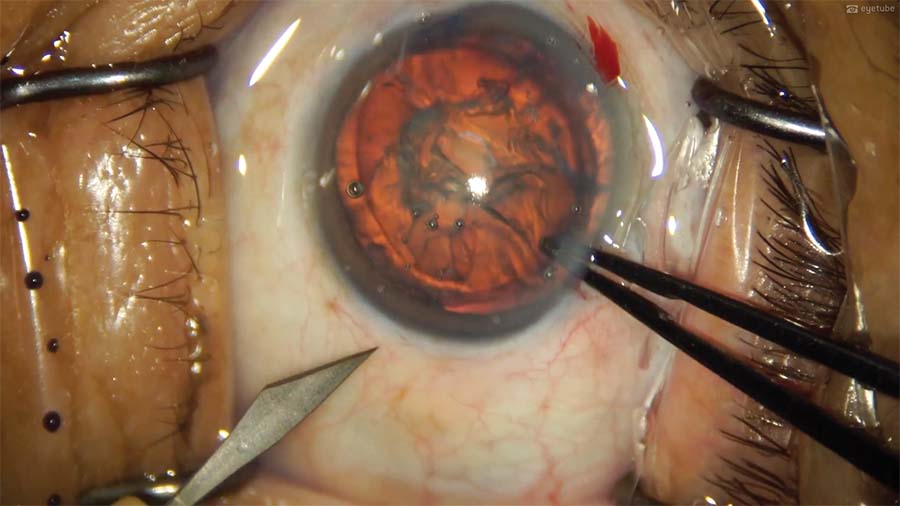
7. A third sideport incision is constructed at the inferotemporal location, and the second sideport incision is converted into the main 2.8-mm clear corneal incision. This is the third time the cornea is entered from that location.
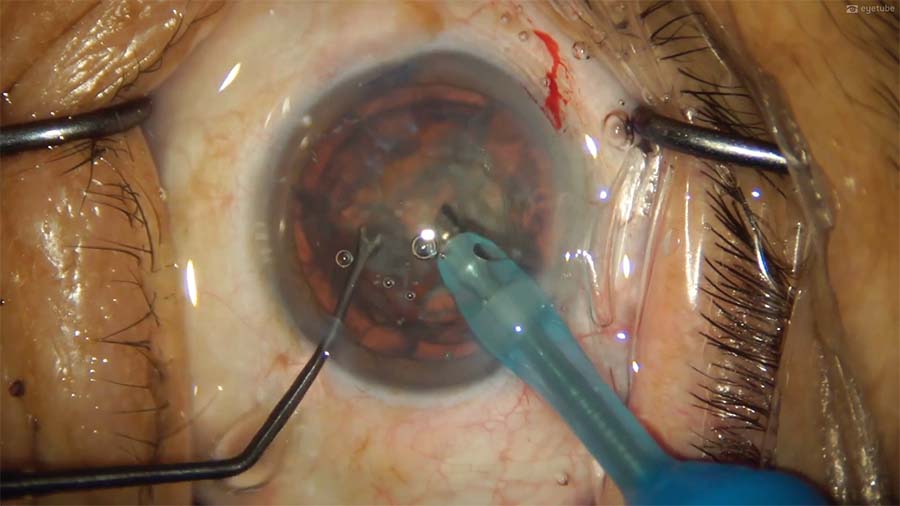
8. The nucleus is fragmented with a direct phaco chop technique using the CataRhex EC phaco machine (Oertli Instrumente). The sharp chopper is removed from the anterior chamber, and a blunt nucleus manipulator is inserted to manipulate nuclear fragments without the risk of tearing capsular structures.
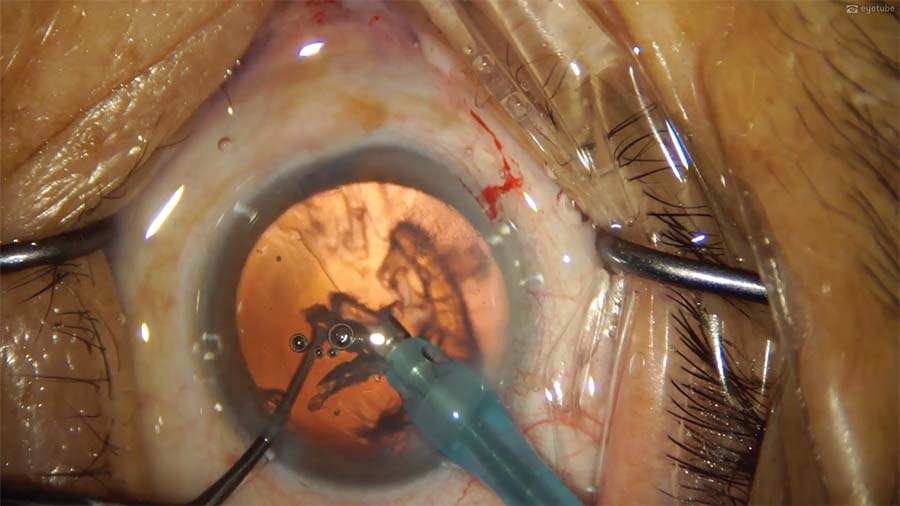
9. The epinucleus is removed. The second instrument nudges the epinucleus from the capsular bag toward the phaco tip, where the tissue is emulsified using low phaco settings (30% phaco power, 250 mm Hg vacuum, and 25 mL/min aspiration flow rate).
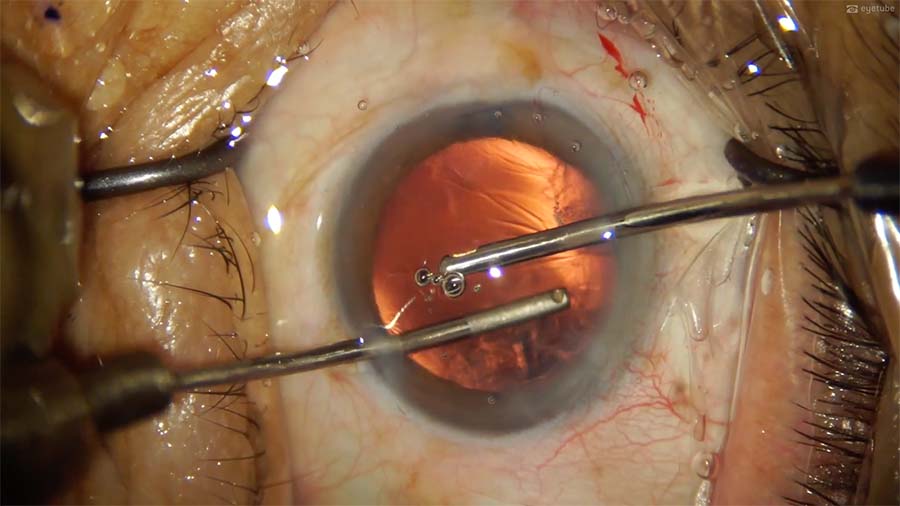
10. A bimanual I/A technique is used. The irrigation cannula is placed in the first 2.8-mm incision. The leaky incision causes anterior chamber instability, so cortical aspiration is performed in a slow and controlled manner. Despite this precaution, corneal folds form in the anterior chamber.
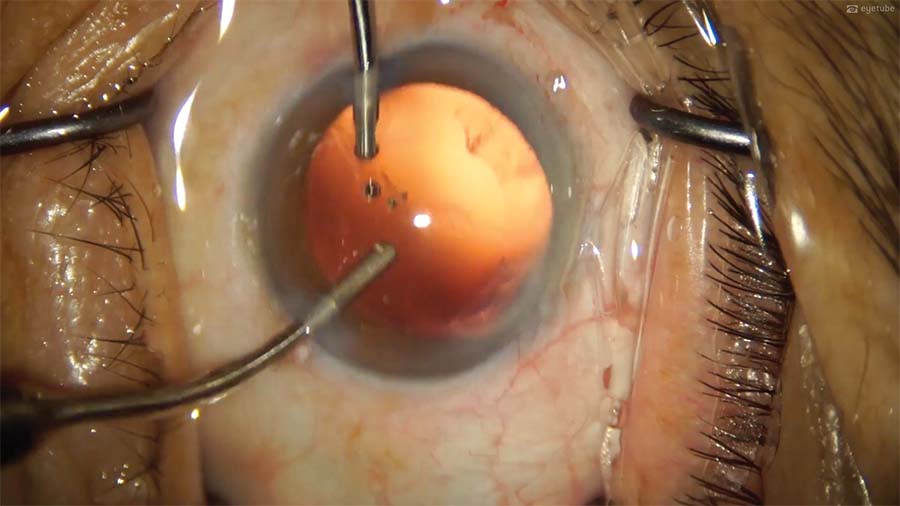
11. The subincisional cortex is removed through the first sideport incision. Plaque on the posterior capsule is polished carefully with a sandblasted aspiration cannula to avoid puncturing the posterior capsule and compromising IOL stability.
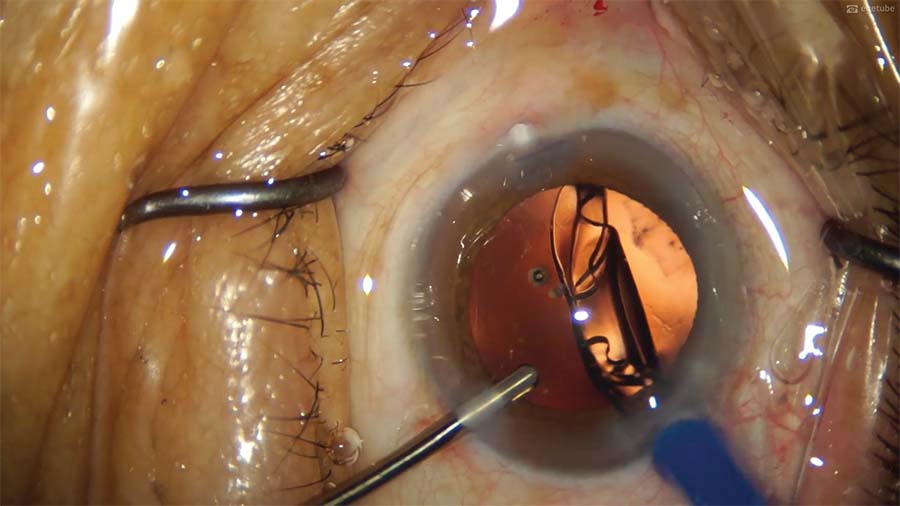
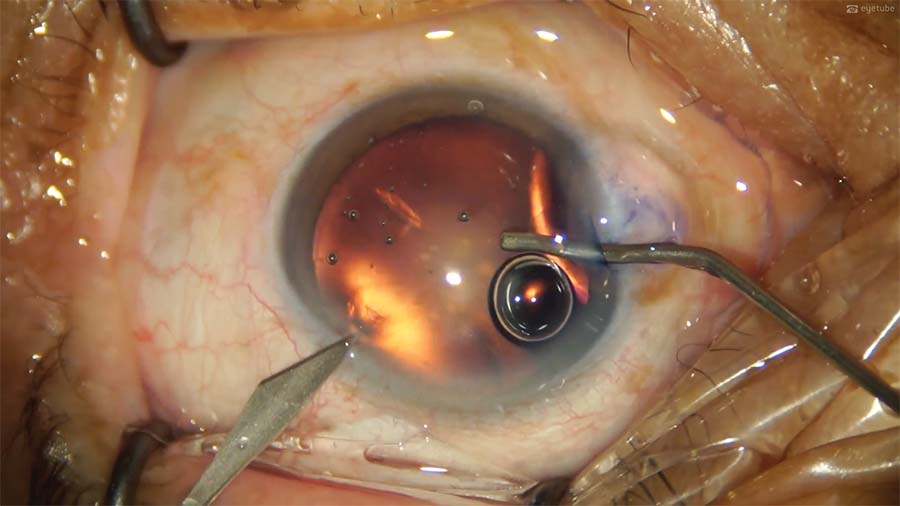
12. A hydrophobic IOL is implanted with a hydroimplantation technique, but insertion does not go smoothly. The IOL is checked for proper centration and stability. A superior subluxation has occurred.
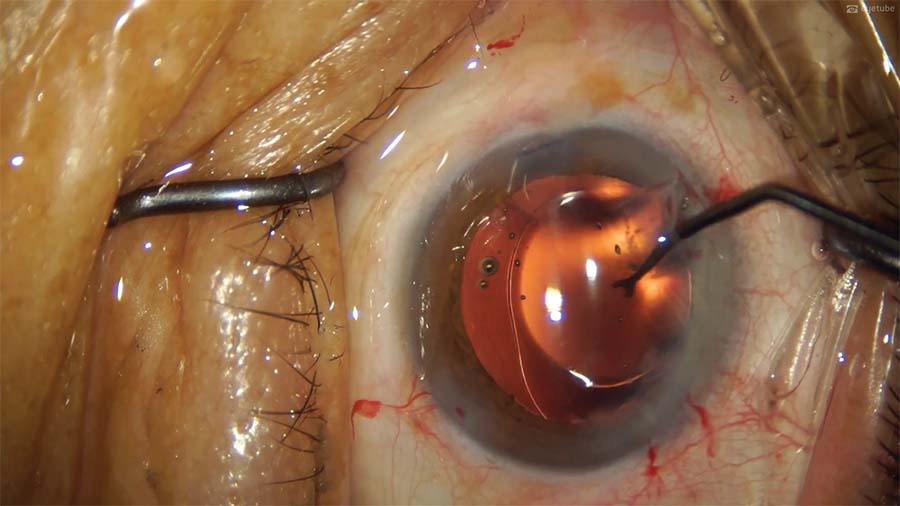
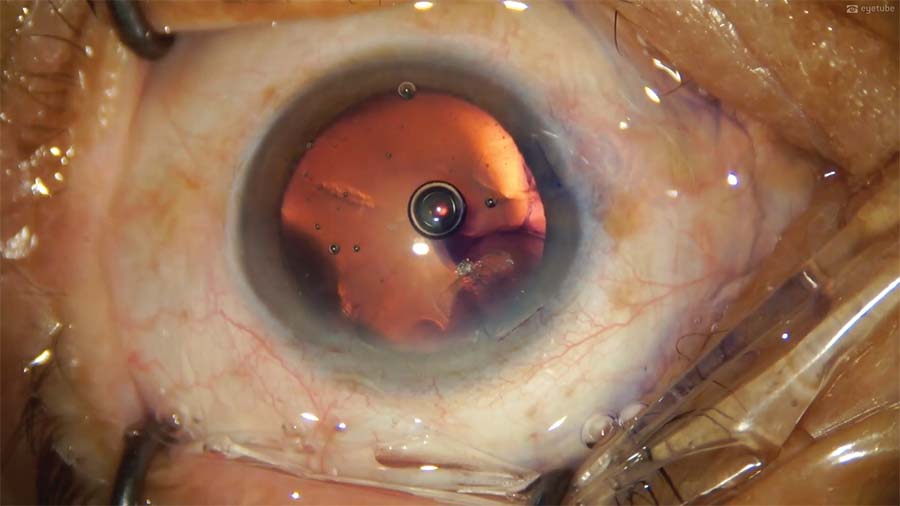
13. An OVD is injected into the anterior chamber and behind the IOL. Then, a Y manipulator is used to push the IOL optic inferiorly to recenter the IOL.
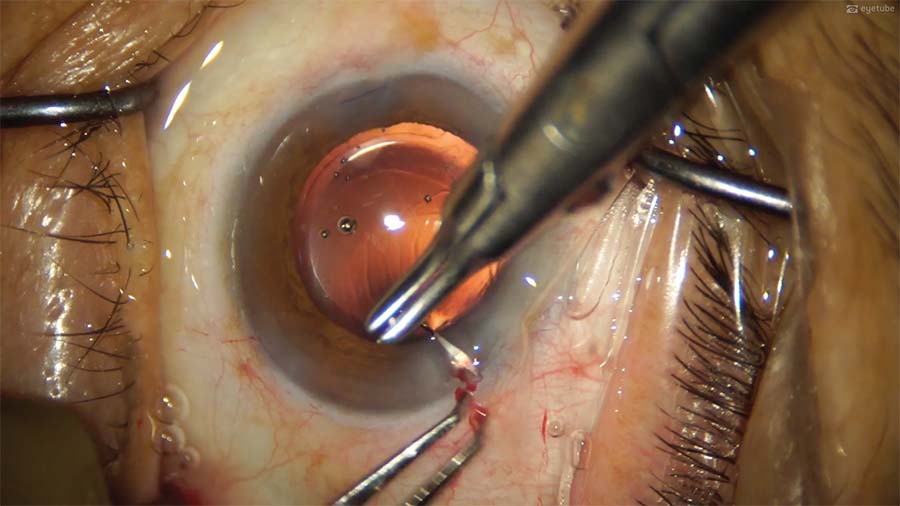
14. The most unstable wound is sutured first, followed by the initial main incision and the remaining incisions. The OVD is aspirated from the anterior chamber but not from behind the IOL to avoid disturbing the well-centered IOL.
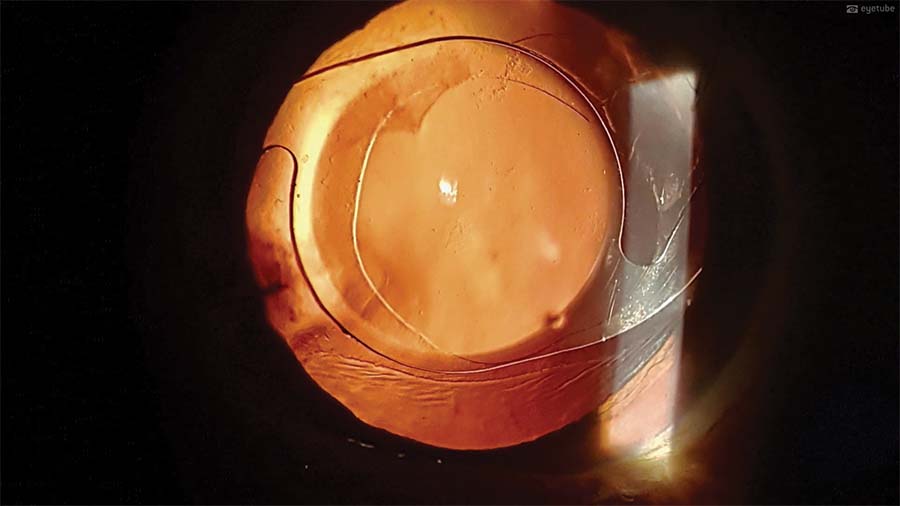
15. An air bubble is injected in the anterior chamber, and the sideport wound is hydrated to ensure tight anterior chamber closure. One week after surgery, the cornea is clear. The sutures, however, are causing about 2.00 D astigmatism. On retroillumination, the IOL is well centered.




