
The optimal location for IOL implantation after successful cataract extraction is within the capsular bag. This placement mimics the location of the natural crystalline lens and allows lifelong stability after the majority of cataract operations. Unexpected events, however, can occur that force cataract surgeons to use alternative strategies for IOL placement. One such option is to implant an IOL in the ciliary sulcus, between the remaining anterior lens capsule and posterior iris. This strategy affords long-term stability and safety in a majority of cases (Figure 1).
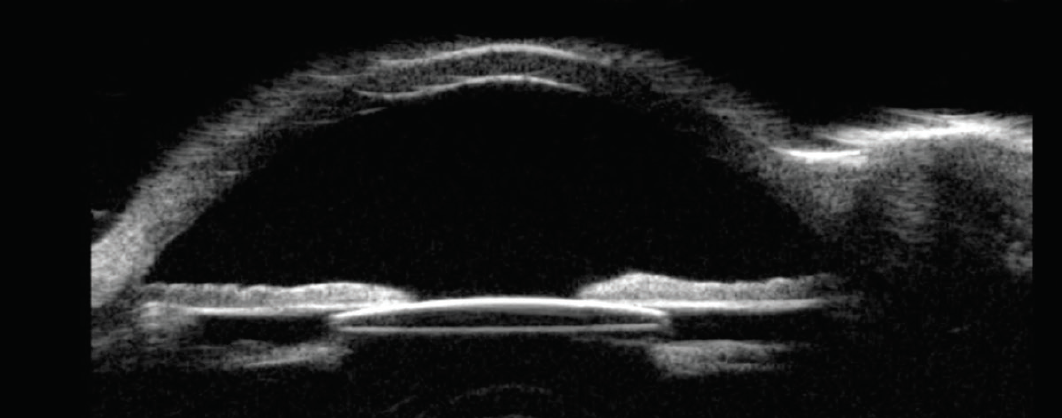
Figure 1. Ultrasound biomicroscopy shows a well-positioned IOL residing in the ciliary sulcus space, anterior to the remaining lens capsule and posterior to the iris.
INDICATIONS
There are three main scenarios in which sulcus IOL implantation may be indicated.1
No. 1: Mild anterior capsular extension. In this situation, it may be possible to place an IOL in the capsular bag if the haptics can be oriented away from the defect and if the area of anterior capsular extension is not large. If these two criteria cannot be met, placing an IOL in the capsular bag risks extending the tear and involving the posterior capsule. The placement of an IOL in the sulcus can be a safe alternative.
No. 2: Ruptured posterior capsule. Placing an IOL in the bag when the posterior capsule has ruptured may also trigger extension of the tear and lead to IOL instability, especially if the capsular defect is large and/or irregular. A ruptured posterior capsule is a common indication for sulcus IOL implantation provided that anterior capsular support is sufficient.
No. 3: Piggyback IOL. The implantation of an IOL in the ciliary sulcus may be considered when a primary IOL is present in the capsular bag and further refractive correction is required.
IOL SELECTION
One-piece acrylic IOLs must not be placed in the ciliary sulcus because friction may develop between the relatively thick, bulky haptics and the overlying posterior iris pigment epithelium, potentially leading to pigment liberation and secondary pigmentary glaucoma.2,3 Suitable lens options for the sulcus include three-piece IOLs with an acrylic optic and PMMA haptics (Figure 2), three-piece silicone IOLs, and one-piece PMMA IOLs. Each of these lens types has advantages and disadvantages.

Figure 2. A three-piece IOL composed of an acrylic optic and PMMA haptics.
Three-piece acrylic lenses. IOLs such as the AcrySof (model MA60AC, Alcon) are generally well tolerated in the ciliary sulcus, and they can be folded and injected through a 2.75-mm corneal incision. The haptics are thin, so care must be taken during insertion to decrease the risks of amputation and breakage. The haptic-to-haptic length is 13.0 mm, which is suitable for most patients but may not be stable in large eyes. The optic has a sharp edge design that may lead to iris chafing.
Three-piece silicone lenses. Some of these IOLs such as the Tecnis Z9002 (Johnson & Johnson Vision) have rounded anterior edges that minimize the risk of iris chafing. A drawback of silicone lenses, however, is that they can limit visualization of the posterior segment if silicone oil fill is performed for retinal pathology in the future.
One-piece PMMA lenses. These IOLs are generally stable when placed in the ciliary sulcus. They are not foldable, however, so a larger incision size is required relative to the aforementioned lens options.
TECHNIQUE
Once the decision has been made to implant an IOL in the ciliary sulcus, an adequate anterior vitrectomy is performed as necessary to decrease the risk of vitreous prolapse and/or traction during IOL insertion. Afterward, an OVD is injected to tamponade remaining vitreous and to deepen and create space between the iris and remaining anterior capsule. The main corneal incision is enlarged as necessary to accommodate the IOL (approximately 2.75 mm for a foldable three-piece IOL and 5.0 to 6.0 mm for a one-piece rigid PMMA IOL).
Three-piece IOLs may be folded and inserted manually or injected with a cartridge delivery system. Alcon’s three-piece IOLs may be injected with the company’s Monarch IOL Delivery System. The larger B cartridge should be selected to accommodate the IOL. After the cartridge has been filled with an OVD, the lens is inserted and advanced into the cartridge without tucking the leading haptic, and the trailing haptic is hooked around the haptic fixation post (Figure 3). The cartridge is then placed and locked into the injector. After entrance into the anterior chamber, the lens is slowly injected into the sulcus space with an initial turn of the cartridge bevel clockwise to promote proper orientation of the leading haptic. The cartridge bevel is then turned counterclockwise while injection continues to allow slow unfolding of the optic in the sulcus space (Figure 4). Forceps or a second instrument may be used to tuck the trailing haptic underneath the proximal iris.
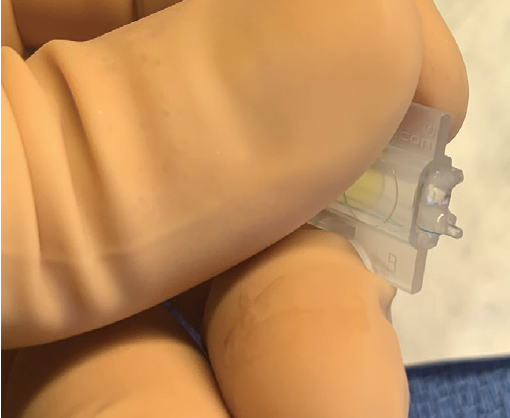
Figure 3. A properly loaded three-piece IOL. The trailing haptic is hooked around the haptic fixation post of a B cartridge.
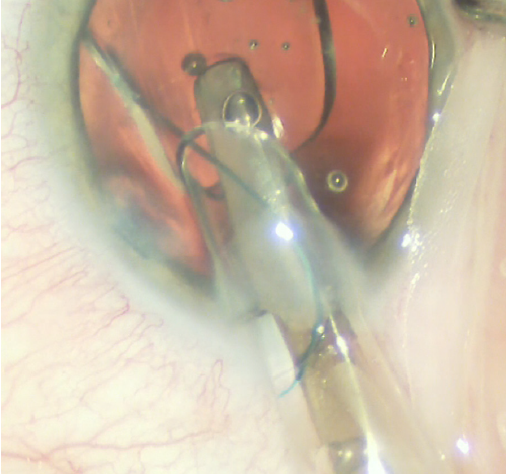
Figure 4. Counterclockwise turning of the lens inserter as the trailing haptic is injected into the ciliary sulcus space.
At this point, if there is enough anterior capsular support, the optic may be captured into the capsular space with the application of gentle pressure posterior to the IOL optic with a second instrument.4 Optic capture can increase the stability of an IOL in the sulcus, especially in large eyes.
IOL POWER CALCULATION
An IOL that is properly positioned within the ciliary sulcus resides anterior to the capsular plane. A modification in power is therefore required to achieve a refractive target similar to that planned with capsular IOL placement.5 In most cases, the lens power can be reduced by 0.50 D compared to the power predicted to reach the target refraction when the same lens is placed in the capsular bag.
It is important to account for the change in A-constant if a different type of IOL will be placed (eg, one-piece lens to a three-piece lens). Simply deducting 0.50 D when changing A-constants will likely lead to a postoperative refraction that is different from the target. If the lens optic is captured in the capsular bag, it is not necessary to deduct 0.50 D, but the change in A-constant is required if the lens type is changing.
Preoperatively determining these calculations helps to reduce the chance of an intraoperative error in lens power selection.
CONCLUSION
Following the guidelines discussed in this article and accompanying sidebar (see Guidelines for Success) and knowing the three main scenarios for IOL placement in the sulcus should help surgeons accomplish safe, effective surgery with excellent postoperative outcomes.
Guidelines for Success
Do
- Do plan to have an appropriately powered three-piece acrylic or silicone IOL or a one-piece PMMA IOL available for possible insertion in the ciliary sulcus.
- Do take into account a change in A-constant when changing the plan from the placement of a one-piece IOL in the capsular bag to the placement of a three-piece IOL in the ciliary sulcus.
- Do use a generous amount of an OVD to tamponade vitreous and create space in the ciliary sulcus before IOL insertion.
- Do enlarge the corneal wound sufficiently to accommodate a large cartridge for the insertion of a three-piece IOL.
- Do use the appropriate cartridge and load the three-piece IOL properly to facilitate accurate orientation.
Don’t
- Don’t place a one-piece acrylic IOL in the ciliary sulcus space because this can lead to secondary pigmentary glaucoma and irreversible vision loss.
- Don’t place an IOL in the sulcus if anterior or posterior capsular support is inadequate.
- Don’t fret if it becomes necessary to implant an IOL in the ciliary sulcus. With proper preparation and technique, an excellent postoperative outcome can be achieved in the majority of cases.
1. Mehta R, Aref AA. Intraocular lens implantation in the ciliary sulcus: challenges and risks. Clin Ophthalmol. 2019:13;2317-2323.
2. Chang DF, Masket S, Miller KM, et al; ASCRS Cataract Clinical Committee. Complications of sulcus placement of single-piece acrylic intraocular lenses: recommendations for backup IOL implantation following posterior capsule rupture. J Cataract Refract Surg. 2009;35(8):1445-1458.
3. Ali MH, Dikopf MS, Aref AA. Late complications of single-piece intraocular lens implantation in the ciliary sulcus. JAMA Ophthalmol. 2018;136(7):825-826.
4. Gimbel HV, DeBroff BM. Intraocular lens optic capture. J Cataract Refract Surg. 2004;30(1):200-206.
5. Suto C, Hori S, Fukuyama E, Akura J. Adjusting intraocular lens power for sulcus fixation. J Cataract Refract Surg. 2003;29(10):1913-1917.



