In 2012, data from the International Council of Ophthalmology (ICO) showed that, although there are more than 200,000 ophthalmologists worldwide, there remains an anticipated global shortfall of ophthalmologists.1 This affects not only developing countries, where there is a well-known shortage of ophthalmologists, but also the developed world. In developed countries, although the number of ophthalmologists is increasing, the percentage of the population aged greater than 60 years is growing twice as fast.
AT A GLANCE
• The challenge for eye care providers in the 21st century is finding innovative solutions to build capacity within services in the face of diminishing resources while maintaining delivery of high-quality care.
• Teleophthalmology is defined as the use of medical information exchanged from one site to another via electronic communications to improve eye health.
• Ophthalmologists’ historical willingness to embrace new technology, recent advances in ocular imaging, and the preponderance of chronic diseases in ophthalmology are conducive to telemedicine.
As such, the challenge for eye care providers in the 21st century is finding innovative solutions to build capacity within services in the face of diminishing resources while maintaining the delivery of high-quality care. An obvious solution to this would seemingly be teleophthalmology—innovative use of technology that allows physicians to see more patients at a lower cost.
Telemedicine is a relatively young field that has changed drastically since its inception 50 years ago, when it was used solely to reach patients in remote locations.2 The telehealth community soon realized that telemedicine may also be applied in the developed world and urban settings. The inceptive access to care model may not be directly applicable in the developed world, where access to care is commonplace; however, the cost savings model—providing alternative, cheaper care delivery methods that reduce cost—and the access to market model—allowing delivery and expansion of services and expertise over a greater distance—are attractive options that could help meet the needs of the aging population of the developed world.
three key barriers
Why, then, has telemedicine not taken off as might be expected? In 2011, The King’s Fund identified key barriers to the adoption of telemedicine, including (1) lack of access to data, (2) commissioning services lacking tools and capabilities to drive change, and (3) the lack of a leadership culture and organizational infrastructure to support such changes.3 More simply, John Pugh MP, Chair of the All-Party Parliamentary Group on Telehealth in the United Kingdom, encapsulated these barriers as a “problem in our relationship with technology.” But surely this cannot apply to ophthalmologists, who operate in a realm of rapidly advancing imaging capabilities, microprecision lasers, and ever-shrinking microsurgical instruments?
Teleophthalmology is defined as the use of medical information exchanged from one site to another via electronic communications to improve a patient’s eye health status.4 More specifically, in teleophthalmology, delivery of eye care may take several forms: through real-time teleconsultation with an eye specialist; via the store-and-forward model, in which digital ocular imaging is acquired and transferred via telecommunications technology to remote eye specialists; or through a remote-monitoring model, which involves tracking of a patient’s health data after he or she has left the hospital (eg, IOP monitoring via a contact lens or implantable device).
Within ophthalmology, the store-and-forward model is most well recognized. A good example of this is diabetic retinopathy screening programs. In England, the National Health Service (NHS) conducts systematic screenings using diabetes registries for diabetic eye disease. Retinal photographs are acquired, read, and assessed for 1.9 million people every year in the NHS Diabetic Eye Screening Program (DESP).5 Patients found to have potentially sight-threatening retinopathy in the DESP are referred to hospital eye clinics. The threshold for referral, defined by the National Screening Committee, is currently equivalent to the Early Treatment Diabetic Retinopathy Study (ETDRS) definition of moderate nonproliferative diabetic retinopathy or any sign of diabetic maculopathy.5,6 This comprises approximately 4.5% of the screening population, and identification of those in need of treatment in this population portends huge savings for the NHS.7
available Evidence and guidelines
Despite the apparent benefits of teleophthalmology, the evidence base indicating its efficiency and effectiveness has been limited. In particular, there is a paucity of evidence from randomized controlled trials in teleophthalmology. Experts in the field have argued that randomized controlled trials may not be the study design of choice to assess the effectiveness of this modality.8 Evidence dictating the standard of care has been either specialty-specific, derived from single-center studies, or based on retrospective data.9-11 Use of teleophthalmology in eye diseases such as diabetic retinopathy, age-related macular degeneration, glaucoma, and retinopathy of prematurity have been reviewed in detail.9,12-14
Examples of published guidelines specific to teleophthalmology have to date been largely confined to diabetic retinopathy; these include the Telehealth Practice Recommendations for Diabetic Retinopathy published by the American Telemedicine Association and the standards and quality assurance documents published by the DESP.15 To date, there is no systematic approach to designing teleophthalmology programs and little guidance on such a process. It is likely that the lack of unified, published standards and quality assurance processes have posed a major barrier for widespread development and uptake of teleophthalmology programs. Nevertheless, we cannot ignore the potential efficiencies that teleophthalmology lends to eye care services. A health economic analysis based on our dataset from Moorfields Eye Hospital at Croydon University Hospital revealed that we can predict a fivefold reduction in cost per patient assessment in a virtual medical retina clinic setting (a store-and-forward model using multimodal retinal imaging with ultra-widefield color photography and OCT) compared with face-to-face consultations with a consultant ophthalmologist. The disparity was further increased to a tenfold cost reduction should a trained optometrist grade the images in place of the consultant ophthalmologist.
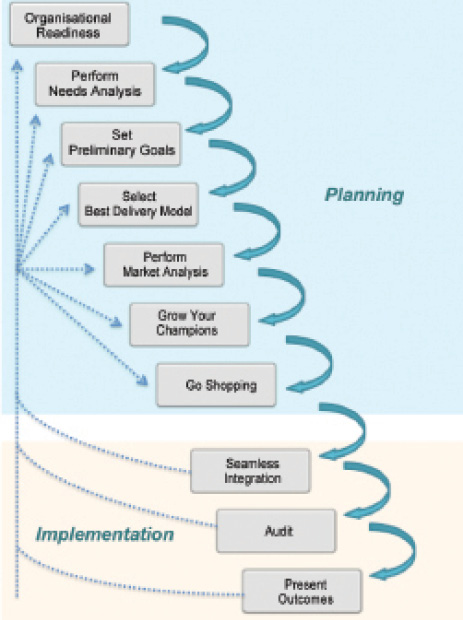
Figure 1. Ten best practices in the process of planning and implementing a telemedicine program.
best practices
For this reason, below we suggest 10 best practices for the development of teleophthalmology programs within existing eye care services in the developed world (Figure 1).
No. 1: Confirm your organization’s readiness. Buying equipment is not the first step. It is paramount that the strengths, weaknesses, opportunities, and threats of your organization are known and reported prior to introducing a new program. The ability to identify leaders equipped with the proper authority to move forward and bring together a team with a shared vision are key features indicating that your organization is ready to design, implement, and sustain a teleophthalmology program.
No. 2: Perform a needs analysis. Identifying key unmet needs will provide a sound foundation for planning. For example, is there sufficient postoperative clinic capacity for patients undergoing cataract surgery? Are the waiting times for glaucoma clinics increasing? The needs analysis must be data-driven, as this will give a clear understanding of the nature and scope of what is required by your organization. This saves both time and costs while avoiding the groupthink trap—a phenomenon that occurs when decisions are made with limited scrutiny because of high cohesion in a group, in which individuals’ desires to conform leads to a failure to consider all available information.
No. 3: Set preliminary goals. Be realistic in this step. Start small and prioritize your service options. Rather than an immediate conversion of every postoperative appointment to a videoconference consultation, start with a pilot clinic and a single surgeon. Ensure that goals are measurable in as many facets as possible, such as patient safety (missed diagnoses), number of unreadable images, patient satisfaction, and staff performance.

No. 4: Select the best delivery model. As noted, ophthalmologists are familiar with the store-and-forward model of teleophthalmology. However, with increasing video transmission speeds, a synchronous real-time model may be more appropriate for non–imaging-based subspecialties and may provide the opportunity to incorporate a multidisciplinary team, including nonmedical ophthalmic technicians, nurses, or optometrists in the community. It is important to examine the infrastructure already in place and the feasibility and cost to install what is required. Information technology infrastructure and bandwidth are the backbones of most telehealth programs and are likely to be the main determinants for which model of practice is ultimately selected.
No. 5: Perform a market analysis. A thorough understanding of the demand for the teleophthalmology model you are considering will help not only to inform the business decision but also to ensure the sustainability of the program. A major hurdle for the longevity of such programs is often a lack of reliable funding. In the United States, attaining reimbursement from insurance companies is a challenge for the majority of teleophthalmology programs. Early engagement of stakeholders such as insurance companies or commissioners and demonstration of data-driven demand will help to ensure the durability of a program.
No. 6: Grow your champions. Champions may be defined as the primary advocates of the telehealth vision. They function as true agents of change for the practice. Having champions at all levels of the program is pivotal: from patients themselves, to administrators checking in the patient for an appointment, imaging technicians engaging the patient and taking photographs, information technology infrastructural support personnel who will ensure that images are securely transferred and that a videoconference does not have an untimely end, and the image readers who risk-stratify patients and diagnose disease.
No. 7: Go shopping. Use technologies you already have, employing validated methods where possible. Evidence from the Joslin Vision Network has shown superiority of ultra-widefield pseudocolor photography compared with standard color photography for the detection of diabetic retinopathy lesions.16 However, not every ophthalmology department can afford to purchase a widefield camera. It is good practice to examine whether a technological upgrade is in line with your primary objective (eg, disease detection versus disease monitoring) to avoid redundancies at the outset. Perhaps less costly or alternative strategies may be more appropriate for your program, such as utilizing multimodal imaging, including the OCT device that is already in your department, and improving the patient journey by placing it next to the fundus camera.
No. 8: Strive for seamless integration. Teleophthalmology services should be designed to complement and integrate into your standard clinic practice and working methods. Performing these services alongside face-to-face clinical activities can provide benefits such as improved clinical support for nonmedical staff and patients, patient safety, flexibility, responsiveness, and the ability to incorporate continual change. A workflow analysis to assess how to incorporate teleophthalmology programs into the eye clinic should be performed at a planning stage.
No. 9: Audit, audit, audit … and audit again. Teleophthalmology protocols and policies should be as close as possible to standard non-telehealth clinical procedures. The same principle applies when developing a quality assurance process. A familiar and standardized quality assurance process saves time, operates on established benchmarks, and ensures continual refinement of the service. It may also help to share learning across subspecialities within the department. All subspecialists in the department may not have considered the benefits of teleophthalmology for their patient groups.
No. 10: Present your outcomes. Feedback both to and from all staff and patients in the program is key. Assist the team to develop short- and long-term performance and strategic goals. Be sure to talk about constant evolution and encourage innovation at all levels.
CONCLUSION
Ophthalmology is well-suited to harness all that the field of telemedicine has to offer. Our historical willingness to embrace new technology, recent advances in ocular imaging, and the preponderance of chronic diseases in our specialty are all factors that should serve to enable our development of telemedicine. An important caveat is the lack of an existing framework to facilitate the translation of telemedicine principles into scalable, replicable, evaluable, evidence-based solutions that can be adapted into real-world eye clinic settings. However, these are not insumountable hurdles. Adaptation of existing clinical guidelines to the teleophthalmology setting using the best practices outlined above can provide a safe mechanism by which to translate evidence and research across domains.
In the future, teleophthalmolgy must not limit itself to the store-and-forward model. In 2015, of the eight top telemedicine companies featured in Telemedicine News, five were developing technologies to support the real-time model, two were in the store-and-forward domain, and only one was working on remote monitoring.17 In contrast, Forbes magazine, which described telemedine as “the new online banking for health,” featured five promising companies that provide telehealth services, four of which were designed for remote monitoring. These companies were diverse, with developments in areas ranging from wearable technology to e-learning psychotherapy programs.18
Regardless of the model that a teleophthalmology program eventually takes or the subspecialty it serves, one must remember that a realistic timeline for implementation is integral to achieving best practices. As the late Steve Jobs famously said, “If you look closely, most overnight successes took a long time.”
Authors’ note: The research presented in this article was supported by The Special Trustees of Moorfields Eye Hospital and the National Institute for Health Research (NIHR) Biomedical Research Centre based at Moorfields Eye Hospital NHS Foundation Trust and UCL Institute of Ophthalmology.
1. Resnikoff S, Felch W, Gauthier TM, Spivey B. The number of ophthalmologists in practice and training worldwide: a growing gap despite more than 200,000 practitioners. Br J Ophthalmol. 2012;96(6):783-787.
2. Beratarrechea A, Lee AG, Willner JM, Jahangir E, Ciapponi A, Rubinstein A. The impact of mobile health interventions on chronic disease outcomes in developing countries: a systematic review. Telemed J E Health. 2014;20(1):75-82.
3. Clark M, Goodwin N. Sustaining innovation in telehealth and telecare. WSD Action Network. http://www.kingsfund.org.uk/sites/files/kf/Sustaining-innovation-telehealth-telecare-wsdan-mike-clark-nick-goodwin-october-2010.pdf. Accessed May 23, 2016.
4. What Is Telemedicine? American Telemedicine Association. http://www.americantelemed.org/about-telemedicine/what-is-telemedicine. Accessed May 23, 2016.
5. Early Treatment Diabetic Retinopathy Study Research Group. Grading diabetic retinopathy from stereoscopic color fundus photographs–an extension of the modified Airlie House classification. ETDRS report number 10. Ophthalmology. 1991;98(5 Suppl):786-806.
6. Mackenzie S, Schmermer C, Charnley A, et al. SDOCT imaging to identify macular pathology in patients diagnosed with diabetic maculopathy by a digital photographic retinal screening programme. PLoS One. 2011;6(5):e14811.
7. Leese GP, Stratton IM, Land M, et al. Progression of diabetes retinal status within community screening programs and potential implications for screening intervals. Diabetes Care. 2015;38(3):488-494.
8. Sharma U, Reed J, Doyle C, Bell D. Challenges in evaluating telehealth through RCT-the problem of randomization. Stud Health Technol Inform. 2012;180:323-327.
9. Strouthidis NG, Chandrasekharan G, Diamond JP, Murdoch IE. Teleglaucoma: ready to go? Br J Ophthalmol. 2014;98(12):1605-1611.
10. Silva PS, Cavallerano JD, Aiello LM, Aiello LP. Telemedicine and diabetic retinopathy: moving beyond retinal screening. Arch Ophthalmol. 2011;129(2):236-242.
11. Lorenz B, Spasovska K, Elflein H, Schneider N. Wide-field digital imaging based telemedicine for screening for acute retinopathy of prematurity (ROP). Six-year results of a multicentre field study. Graefes Arch Clin Exp Ophthalmol. 2009;247(9):1251-1262.
12. Sim DA, et al. The evolution of teleophthalmology programs in the United Kingdom: beyond diabetic retinopathy screening. J Diabetes Sci Technol. 2016;10(2):308-317.
13. Sreelatha OK, Ramesh SV. Teleophthalmology: improving patient outcomes? Clin Ophthalmol. 2016;10:285-295.
14. Sim DA, Keane PA, Tufail A, Egan CA, Aiello LP, Silva PS. Automated retinal image analysis for diabetic retinopathy in telemedicine. Curr Diab Rep. 2015;15(3):14.
15. Telehealth Practice Recommendations for Diabetic Retinopathy. American Telemedicine Association. 2011. http://www.americantelemed.org. Accessed May 23, 2016.
16. Silva PS, Horton MB, Clary D, et al. Identification of diabetic retinopathy and ungradable image rate with ultrawide field imaging in a national teleophthalmology program [published online ahead of print March 1, 2016]. Ophthalmology. doi:10.1016/j.ophtha.2016.01.043.
17. Morris M. 8 telemedicine companies making a difference in 2015: ATA 2015 vendor previews. Telemedicine News. April 10, 2015.
18. Harrison K. 5 companies greening healthcare with telehealth services. Forbes. September 8, 2015.
Naomi Sheeter, BA (Hons), MPhil
• Clinical Operations Manager, Moorfields South, Croydon University Hospital, London
• Financial interest: None acknowledged
Dawn A. Sim, PhD, FRCOphth
• Consultant Ophthalmologist, NIHR Biomedical Research Centre for Ophthalmology, Moorfields Eye Hospital NHS Foundation Trust, and at Moorfields South, St George’s & Croydon University Hospital, London
• Clinical Lead, Southwest London, Diabetic Eye Screening Programme
• Honorary Research Fellow, University College London, Institute of Ophthalmology, London
• dawn.sim@moorfields.nhs.uk
• Financial interest: None acknowledged


