Radial keratotomy (RK; Figure 1) was a popular refractive surgical procedure to correct myopia in the 1970s and 1980s, before the advent of the excimer laser for refractive treatment. This incisional surgery technique was taught by, among others, Professor Svyatoslav Fyodorov in Russia; Leo Bores, MD, and J. Charles Casebeer, MD, in the United States; and Luc Haverbeke, MD, in Belgium.1 Many patients on both sides of the Atlantic opted to undergo this surgery in order to eliminate or reduce the need for spectacles or contact lenses.
There were some limitations to RK, however, and side effects did occur, especially when high corrections were attempted.2 Nevertheless, RK had a great deal of success, and thousands of people underwent the procedure. Today, these patients are at least 20 or 30 years older. Some of them have developed hyperopia or hyperopic astigmatism (hyperopic progression), ocular hypertension, and, increasingly, cataracts.
When cataract surgery is performed in an eye with previous RK, the technique may be basically the same as in a virgin eye, but the IOL calculations are fundamentally different. This article reviews some considerations in performing cataract surgery in patients after RK.
EFFECT OF RK ON CORNEAL SHAPE
Incisional surgery such as RK was performed with a diamond knife; the length of the blade was set according to the pachymetry readings. To have greater effect (ie, to correct more diopters of myopia), the cuts were made from the periphery of the cornea toward the center (Figure 1).1 Whether the incisions were centripetal or centrifugal, they resulted in corneal flattening in the center and corneal steepening, or bulging, in the periphery.
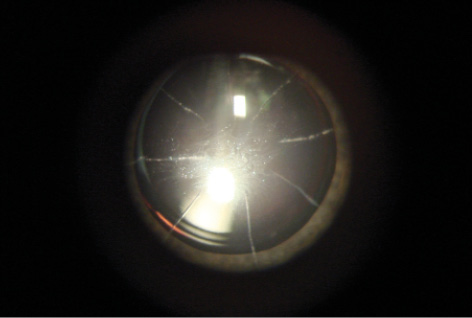
Figure 1. An eye with an eight-incision RK.
In some eyes, this flattening progressed with time, which meant that the keratometry as measured by classical instruments (eg, Javal keratometer) became more and more inaccurate.3 In fact, the clear central optical zone was often only 3 mm, which meant that classical keratometers measuring at the 3.2-mm zone missed the central flatter zone of effective corneal power. Therefore, there was a need to find a new method of keratometry to be used in classical IOL calculations for post-RK eyes.
It should be noted that, when eight incisions were used, the central corneal flattening was more significant than with six or four cuts, thus exaggerating the central irregularity.
MEASUREMENT OF CENTRAL CORNEAL POWER
RK caused relatively proportionally equal flattening of both the front and back surfaces of the cornea, leaving the refractive index unchanged, as demonstrated in model calculations by Hanna.2 By contrast, PRK and LASIK flatten only the front surface, thereby changing the refractive index calculations. Because of the modifications induced by RK, the central corneal power is no longer the real corneal power required for IOL calculation formulas.4
How can the effective corneal power be measured in a post-RK eye? Some instruments can be more helpful than others:
Manual keratometry. As explained previously, classical manual keratometers measure at a 3.2-mm optical zone. This is the least accurate method for measurement in RK eyes. Available instruments, such as the Javal-Schiötz keratometer and similar devices, make too many assumptions, not taking into account irregular corneal astigmatism.5
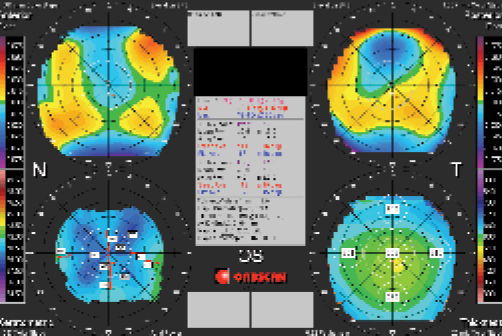
Figure 2. Orbscan map of a 65-year-old patient’s left eye with a previous four-cut RK. It is difficult to assess the correct K from the topography map.
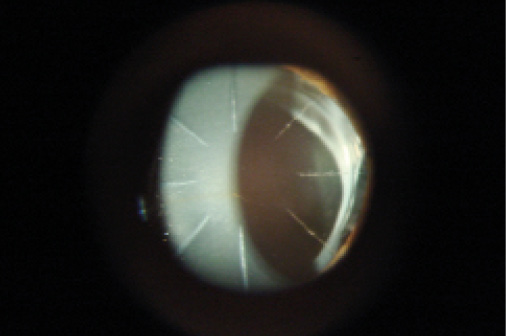
Figure 3. Slit-lamp view of the right eye of the same patient as Figure 2. This eye has an eight-incision RK. The left eye received a four-incision RK to provide monovision.
Videokeratography. Several authors have demonstrated the value of computerized videokeratography, also known as corneal topography, in determining IOL power calculations, especially in surgically altered corneal surfaces.6-8 With the Topographic Modeling System (TMS; Tomey) and similar devices, a simulated keratometry (K) value is determined based on the power of Placido mires 7, 8, and 9 of the videokeratoscope. Maeda and colleagues developed the parameter of average central power, based on the average of corneal powers inside an area demarcated by the entrance pupil on the TMS.6 The Orbscan (Bausch + Lomb) can generate topographic height and power maps of the anterior and posterior cornea (Figures 2 to 4). It is also possible, using the Orbscan’s tangential keratometric map, to draw a circle of 2 mm and take the K reading inside this circle, at 0.5 mm, 1 mm, and 1.5 mm of eccentricity. The algebraic sum of these Ks, divided by the number of measurements, represents a new keratometry that can be used in IOL power calculation formulas (Daniel Gatinel, MD, personal communication).
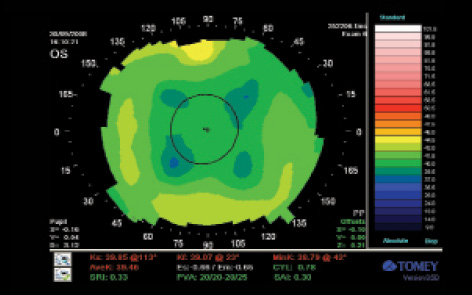
Figure 4. Same patient as in Figure 2, TMS map of left eye. Average K (AveK) of 39.46 is displayed on the map. Interestingly, this AveK is not very different from our calculated K (39.8). This patient is case 8 in Table 2.
Scheimpflug imaging. The Pentacam (Oculus) may offer an improvement over the Orbscan; it eliminates any errors due to eye movements during image capture. The Scheimpflug camera rotates around the visual axis of the eye while taking image slices. The Pentacam measures net power.9
The corneal power estimated by these instruments is not always reliable, however. The critical 3-mm central optical zone is irregular. The topography maps show flattening, but some topographers may miss the flattening in the real optical zone of the eye; the flattening in itself exaggerates the central irregularity.
POST-RK CALCULATION METHODS
Another way to find the real corneal power in post-RK eyes is to use certain calculation methods. The two most important are the historical or clinical method and the contact lens method.
Historical or clinical method. This method depends on access to the eye’s pre-RK refractive data. The effect of the RK—in other words, the myopic correction or reduction—must be deduced from the mean K as measured before RK surgery, but this myopic correction must be adjusted to the corneal plane, not the spectacle plane. The value of the myopic correction must be determined at most a few years after RK was performed, and not when the cataract occurs, because cataract formation in itself induces myopia.10-12
AT A GLANCE
• Before the advent of the excimer laser, thousands of people underwent RK.
• When cataract surgery is performed in an eye with previous RK, the technique may be basically the same as in a virgin eye, but the IOL calculations are fundamentally different.
• Any surgeon examining an RK patient should ask the patient or the RK surgeon for a copy of any records of the surgery.
For example, if a patient was a myope of -6.00 D before RK, and his or her refraction was -0.50 D after RK, the surgical correction is 5.50 D in the spectacle plane. This equates to 5.00 D in the corneal plane.
Let us assume that this patient is now +1.00 D with a K reading of 35.00 D. If the mean K of this patient was 43.00 D before RK surgery, the new K to be used is 43.00 – 5.00 = 38.00. This is certainly different from the 35.00 D that would be measured by the Javal keratometer in this example. If we were to use 35.00 D as the K in the IOL calculation for this patient, we would induce hyperopia, which is the most common problem in cataract surgery after RK. If we use the K as calculated above (38.00 D), the IOL calculations will be correct.5,11-14
Contact lens method. For this method, manifest refraction is first done without a contact lens. Then, a plano hard contact lens of known base curve is placed on the eye. If the manifest refraction does not change, then the central corneal curvature is equal to the base curve of the contact lens. If there is a difference, this difference must be subtracted from the base curve of the contact lens to obtain the corneal power.
However, two problems can occur with this method, as has been pointed out by Hoffer.11 Plano hard contact lenses are not available in every ophthalmologist’s practice, and, more important, the cataract may complicate an accurate refraction.
Other methods. In the double-K method, two K values are used: the pre-refractive surgery value (Kpre), for the calculation of the effective lens position (ELP), and the postrefractive surgery value (Kpost), for the vergence formula that finally gives the IOL power. The Kpost is calculated using the clinical history method.15 However, this method seems to be more useful after PRK and LASIK than after RK because laser ablative surgery changes the relationship between the anterior and posterior corneal radii of curvature, so that the ratio is no longer 7.5/6.3.4,9,10
In the corneal bypass method of Ladas and Stark, the preoperative K is used, and the target is the refractive change.16 In the example above, 43.00 D would be used for the K, aiming at -5.50 D.
IOL CALCULATION FORMULAS
Calculations of IOL power in cataract surgery are based on measurements of keratometric diopters of curvature, axial length, and estimation of postoperative anterior chamber depth. Holladay introduced the notion of ELP, which is defined as the distance from the corneal vertex to the thin plane of the IOL. This value cannot be measured; we have to predict it.10 The Holladay 1, Hoffer Q, Haigis, and SRK/T formulas all base the position of the lens on axial length and keratometry.13,17
The recently developed Haigis-L formula is more helpful for calculations after ablative surgery (PRK and LASIK) than after RK because it corrects the keratometer’s index error, the radius error, and the IOL formula error.17
The determination of optical axial length, from corneal vertex to fovea, is critical. For this purpose, optical biometry with the IOLMaster is more accurate than ultrasound A-scan, especially for long eyes (>26 mm, as defined by Percival).18 Hoffer has shown that the third-generation formulas (Holladay, Hoffer Q, SRK/T, and Haigis) are superior to the empirical SRK 1 and SRK 2 formulas.3,11,12
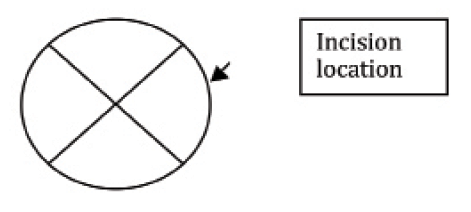
Figure 5. Cataract surgery incision location for a four-cut RK.
The main reason for IOL power miscalculation is mismeasurement of corneal power. In this article, we previously discussed some ways to avoid this. The clinical history method seems to be the most reliable.5
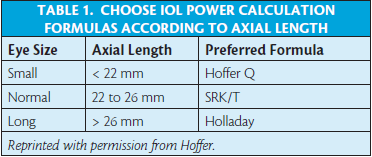
In our hands, the Hoffer Q formula (also available as a computer program from Hoffer) gives the best results. It also helps to choose a formula based on the eye’s characteristics (Table 1), to use more than one formula, to avoid regression formulas, and to choose the highest value for the implant.
For IOL calculations, the surgeon should stay on the safe side. Instead of the classical refraction target of -0.50 D after cataract surgery, we recommend to aim at -1.00 D or even -1.50 D. This allows for a little keratometric miscalculation and provides some reading vision for the patient.
After cataract surgery, a hyperopic shift is often noted. This is due to corneal edema and will disappear with time. However, a highly myopic postoperative refraction will not clear away, and a lens exchange will be required.
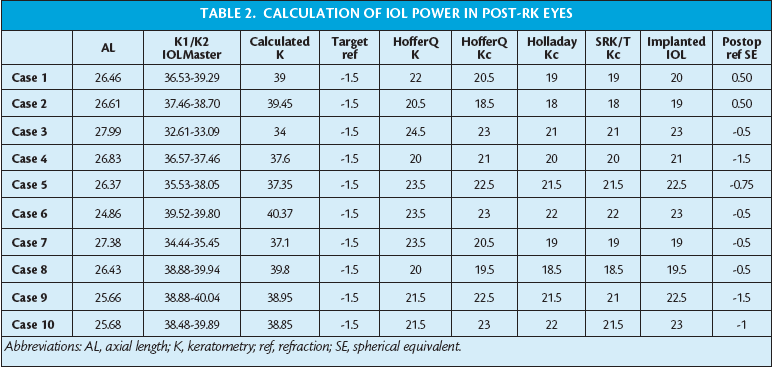
Table 2 presents a few examples from our database of calculations with a number of formulas and the final postoperative result.
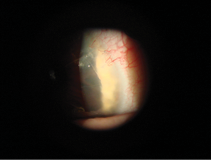
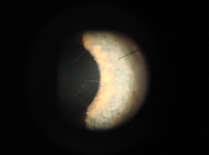
Figure 6. Cataract incision crossing an old RK cut (A),inducing fish-mouth healing. Correct placement of incision, with suture (B).
TIPS FOR CATARACT SURGERY AFTER RK
Many RK patients develop against-the-rule astigmatism while aging. This is, in fact, a normal evolution for many eyes, and it is particularly the case after RK, as progressive corneal flattening seems to induce hyperopic astigmatism. Therefore, it is necessary to choose the cataract incision location according to the patient’s astigmatism. Most RK patients will benefit from a clear corneal incision made in the horizontal axis: in other words, a temporal incision (Figure 5).
However, the corneal incision must never cross the RK cuts. This induces a fish-mouth reaction, with incisions that heal poorly and induction of high astigmatism (Figure 6).
Clear corneal incisions should be sutured to minimize astigmatism. If many RK cuts were done, and it is impossible to create a 2- to 3-mm clear corneal cataract incision, a scleral tunnel should be preferred.
Because IOL power calculations are so challenging in these eyes, it is safer to aim for -1.00 D or -1.50 D of myopia when calculating the IOL power; this leaves a margin of error should the calculations be incorrect. If the patient is more than -1.00 D myopic postoperatively, the surgeon can emphasize to the patient the advantage of the reading vision gained with this myopic refraction. Alternatively, the surgeon can treat the residual refractive error with an excimer laser using a smaller optical zone than usual.
If different methods can be applied to calculate the IOL power, and the results are comparable, then the cataract surgeon can feel more comfortable with the power prediction; it is recommended to use the highest suggested IOL power.
The refraction will usually stabilize by about 3 months after cataract surgery. Most patients experience a hyperopic shift, slowly disappearing, probably as a consequence of biomechanical changes induced by edema in the radial incisions.19 High refractive error after cataract surgery may require a lens exchange. The patient must be informed of this possibility before cataract surgery.
Multifocal IOLs are not indicated after RK surgery; the unusual shape of the cornea (central flattening plus bulging in the periphery: reversed asphericity) combined with the multifocal zones of the IOL can induce halos and glare at night and poor optical performance even during the day.
CONCLUSION
A practical recommendation is that surgeons should give every refractive surgery patient his or her preoperative refractive and keratometric data, along with the correction obtained from the refractive surgery, and patients should keep these data for the future. Any surgeon today examining an RK patient should ask the patient or the RK surgeon for a copy of any records of the surgery, if the medical archives are still available. This information will be very helpful when a cataract develops.
1. Haverbeke L, Levy JP. La kératotomie radiaire. Paris: Masson; 1990.
2. Hanna KD, Jouve FE, Waring GO III. Preliminary computer simulation of the effects of radial keratotomy. Arch Ophthalmol. 1989;107:911-918.
3. Hoffer KJ. The Hoffer Q formula: a comparison of theoretic and regression formulas. J Cataract Refract Surg. 1993;19(6):700-712.
4. Holladay JT. IOL calculations following radial keratotomy surgery. Refract Corneal Surg. 1989;5(3):36.
5. Seitz B, Langenbucher A. Intraocular lens power calculation in eyes after corneal refractive surgery. J Refract Surg. 2000;16:349-361.
6. Maeda N, Klyce SD, Smolek MK, McDonald MB. Disparity between keratometry style readings and corneal power within the pupil after refractive surgery for myopia. Cornea. 1997;16:517-524.
7. Cuaycong MJ, Gay CA, Emery J, Haft EA, Koch DD. Comparison of the accuracy of computerized videokeratography and keratometry for use in intraocular lens power calculation. J Cataract Refract Surg. 1993;19(suppl):178-181.
8. Cellikol L, Pavlopoulos G, Weinstein B et al. Calculations of intraocular lens power after radial keratotomy with computerized video keratography. Am J Ophthalmol. 1995;120:739-750.
9. Holladay JT. IOL calculations after refractive surgery with the Pentacam. CRST Europe. Summer 2007 (suppl) 11-13.
10. Holladay JT. Cataract surgery in patients with previous refractive corneal surgery (RK, PRK and LASIK). Ophthalmic Practice. 1997;15:238-244.
11. Hoffer KJ. Intraocular lens power calculation for eyes after refractive keratotomy. J Refract Surg. 1989;5:203.
12. Hoffer KJ. How to do cataract surgery after RK. Review of Ophthalmology. May 1996:117-120.
13. Lteif Y, Gatinel D. Intraocular lens power calculation after keratorefractive surgery [article in French]. J Fr Ophtalmol. 2008;31(3):326-334.
14. Gimbel H, Sun R, Kaye GB. Refractive error in cataract surgery after previous refractive surgery. J Cataract Refract Surg. 2000;26:142-144.
15. Aramberri J. Intraocular lens power calculation after corneal refractive surgery: double-K method. J Cataract Refract Surg. 2003;29:2063-2068.
16. Ladas JG, Stark WJ. Calculating IOL power after refractive surgery. J Cataract Refract Surg. 2004;30(12):2458; author reply 2458-2459.
17. Haigis W. Intraocular lens calculation after refractive surgery for myopia: Haigis-L formula. J Cataract Refract Surg. 2008;34:1658-1663.
18. Percival P. Lens power calculation–is it necessary? Trans Ophthalmol Soc U K. 1983;103(pt 5):577-579.
19. Alio J, Azar D. Management of Complications in Refractive Surgery. Berlin: Springer Verlag; 2008, 214-221.
Bernard Mathys, MD
• Medical Director and Senior Surgeon, Brussels Eye Specialists, Belgium
• bernard.mathys@skynet.be
• Financial disclosure: None

