
IOL characteristics can influence postoperative cataract surgery outcomes, including the occurrence and frequency of complications such as posterior capsular opacification (PCO). Several studies have described the possible roles of material and design in the prevention of this hurdle. Some of these studies have evaluated material properties, particularly adhesive properties that lead to different patterns of protein adsorption on IOL surfaces.1
ROLE OF DESIGN
The most important IOL feature in PCO prevention with capsular-bag–fixated IOLs was found to be the presence of a square edge on the posterior optic surface, regardless of IOL material (Figure 1). Still, studies evaluating the microstructure of the edges of currently available foldable IOLs have shown that all square edges are not equally square.2 As a group, hydrophilic acrylic lenses were found to have edges that were less square than those of hydrophobic acrylic and silicone lenses.3 Furthermore, animal, cadaver eye, and clinical studies have shown that the square edge should be present for 360° around the lens optic for maximal efficiency in PCO prevention.4
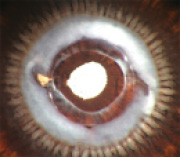
Figure 1. Pseudophakic human eye obtained postmortem implanted with a three-piece silicone lens with square optic edges. Although there is abundant Soemmering ring formation, the posterior capsule is clear. The edge of the capsulorrhexis exhibits fibrosis.
Some IOL designs significantly change the interaction of the lens with the capsular bag, which has an impact on overall capsular bag clarity. Tassignon’s bag-in-the-lens concept involves the use of an IOL designed to fit within the opening of a twin capsulorrhexis, and the surgeon must perform anterior and posterior capsulorrhexes of the same size to hold the lens (Figure 2).5 Recently, studies have suggested that lenses maintaining the capsular bag in an open or expanded state were associated with improved bag clarity.6

Figure 2. Pseudophakic human eye obtained postmortem implanted with the Tassignon bag-in-the-lens (Morcher) showing an overall clear bag with localized patches of Soemmering ring formation.
ROLE OF MATERIAL
The material used in the manufacture of IOLs also has an association with specific changes in the lens optics that can be observed postoperatively. As a group, hydrophilic acrylic or hydrogel materials have an increased tendency to calcify in the late postoperative period, in comparison with other IOL materials (Figure 3). This appears to be a multifactorial problem, and the possibility of host environment (patient-related) factors cannot be ruled out.7 Recent studies have described a specific localized pattern of calcification of different hydrophilic acrylic lenses in eyes undergoing procedures involving repeated intracameral injection of air or gas. The same calcification pattern seen in these lenses has also been observed after other types of secondary procedures, including posterior segment surgery.8
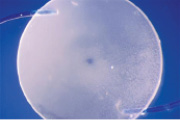
Figure 3. Explanted three-piece hydrophilic acrylic lens exhibiting calcification.
Silicone lenses have been shown to calcify in eyes with asteroid hyalosis. In these cases, studies demonstrated deposits only on the posterior optic surface of the lenses (Figure 4). The deposits could be partially removed with an Nd:YAG laser, but there was reaccumulation of deposits after the laser procedure because the asteroid bodies are rich in calcium and phosphates.7 The interaction of silicone oil, used in vitreoretinal surgery, with standard silicone IOLs is a well-documented clinical complication. Patients with vitreoretinal problems that may require use of silicone oil should not be implanted with silicone IOLs, as the oil will attach to the lens surfaces and cause optical irregularities.7

Figure 4. Explanted plate-haptic silicone lens exhibiting calcification on the posterior optic surface.
Two hydration-related phenomena have been described in the literature in IOLs made of various materials, particularly hydrophobic acrylic materials: glistenings and surface light scattering.9 All hydrophobic acrylic lenses on the market do not have the same propensity to exhibit these changes, however. Glistenings are fluid-filled microvacuoles that form within the IOL optic when the lens is in an aqueous environment. They are usually distributed throughout the entire bulk of the IOL optic, and their size usually ranges from 1 to 20 µm. Surface light scattering is a whitening appearance of the lens surface when light is directed at the IOL with an angle of incidence of 30° or greater during slit-lamp examination. It is also apparent during image capture Scheimpflug photography at an angle of 45°. Studies have shown that this scattering was predominantly caused by phase separation of water (from aqueous humor) as subsurface nanoglistenings.9
The condition called snowflake degeneration is usually observed in three-piece PMMA lenses made in the 1980s and early 1990s, which were generally manufactured by injection molding. This is a slowly progressive condition represented by PMMA degradation caused by long-term ultraviolet light exposure. The intraoptic spherical lesions observed in these cases are thought to correspond to foci of degenerated PMMA material (Figure 5).7
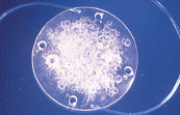
Figure 5. Explanted three-piece PMMA lens exhibiting snowflake degeneration.
SUMMARY
Although IOL design appears to have a more significant impact on overall postoperative capsular bag clarity compared with optic material, specific changes in IOL optics may be associated with the material used in the manufacture of the lens.
1. Linnola RJ, Werner L, Pandey SK, et al. Adhesion of fibronectin, vitronectin, laminin, and collagen type IV to intraocular lens materials in pseudophakic human autopsy eyes. Part 1: histological sections. J Cataract Refract Surg. 2000;26:1792-1806.
2. Werner L, Muller M, Tetz M. Evaluating and defining the sharpness of intraocular lenses. Microedge structure of commercially available square-edged hydrophobic lenses. J Cataract Refract Surg. 2008;34:310-317.
3. Werner L, Tetz M, Feldmann I, et al. Evaluating and defining the sharpness of intraocular lenses. Microedge structure of commercially available square-edged hydrophilic lenses. J Cataract Refract Surg. 2009;35:556-566.
4. Ness P, Werner L, Maddula S et al. Pathology of 219 human cadaver eyes with 1-piece or 3-piece hydrophobic acrylic intraocular lenses: capsular bag opacification and sites of square-edged barrier breach. J Cataract Refract Surg. 2011;37:923-930.
5. Werner L, Tassignon M, Zaugg B et al. Clinical and histopathologic evaluation of six human eyes implanted with the bag-in-lens. Ophthalmology. 2010;117:55-62.
6. Kramer GD, Werner L, Mamalis N. Prevention of postoperative capsular bag opacification by intraocular lenses and endocapsular devices maintaining an open or expanded capsular bag. J Cataract Refract Surg. In press.
7. Werner L. Causes of intraocular lens opacification or discoloration. J Cataract Refract Surg. 2007;33:713-726.
8. Werner L, Wilbanks G, Nieuwendaal CP, et al. Localized opacification of hydrophilic acrylic intraocular lenses after procedures using intracameral injection of air or gas. J Cataract Refract Surg. 2015;41:199-207.
9. Werner L. Glistenings and surface light scattering in intraocular lenses. J Cataract Refract Surg. 2010;36:1398-1420.
Liliana Werner, MD, PhD
• Associate Professor and Co-Director, Intermountain Ocular Research Center
• John A. Moran Eye Center, University of Utah, Salt Lake City, Utah
• liliana.werner@hsc.utah.edu
• Financial disclosure: None

