Differentiating the Effects of Design Versus Material
Differences in IOL function can be seen even within material categories.

By Randall J. Olson, MD
When IOLs were first introduced, the only lens material used was polymethylmethacrylate (PMMA). Differences among materials were largely irrelevant until glass IOLs came along. Glass had some adherents until it became clear that Nd:YAG laser capsulotomy could shatter this material in the eye, at which time these products disappeared almost overnight.1
Later, with the introduction of foldable IOLs, first with silicone and then hydrophobic and hydrophilic acrylic materials, the issue of material impact became significant. At first, the general impression was that material was extremely important, and conclusions were readily drawn in regard to perceived variances in different materials. By and large, it was taken as a fact that silicone IOLs were more inflammatory in the eye and induced much more capsular opacification, both posterior and anterior, than hydrophobic acrylic IOLs (Figure 1).
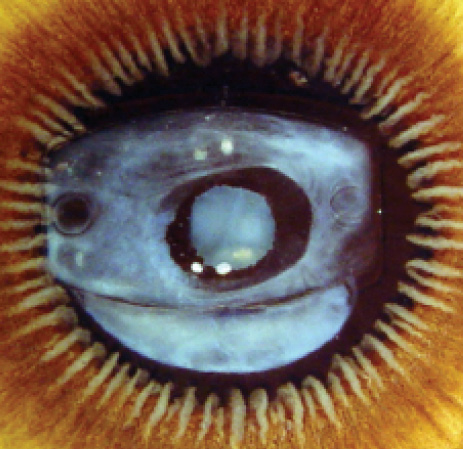
Figure 1. A Miyake view of a first-generation plate silicone IOL with capsular contracture and a previous laser capulotomy.
It is now understood that the physical properties of IOLs were also important regarding how they behaved in the eye. The best example, and the principle that is now best understood, has to do with the truncated optic edge, which can act as a barrier to lens epithelial cell migration and result in decreased posterior capsular opacification (PCO).2,3
When this barrier effect was observed, it was suddenly not so clear that PCO prevention was necessarily related to the hydrophobic acrylic material; it could just as easily have been related to the mechanical edge effect. Furthermore, although studies showed an increase in inflammatory signs such as giant cell deposition with early generations of silicone IOLs,4 with later generations this tendency was less clear.5
This was also the case in regard to anterior capsular contraction and opacification. These phenomena appeared to be related to silicone IOLs, but newer variations of silicone did not necessarily share this same characteristic.6
AT A GLANCE
• An IOL is a total package. That is, both its physical characteristics and its material are important in how the lens functions in the eye.
• There are differences within each material category (silicone, hydrophobic acrylic, hydrophilic acrylic), and one material will not necessarily act like another in the same category.
• Despite a long evolution, no IOL material has all the ideal properties; every material has advantages and disadvantages.
• Hybrid materials hold the promise of combining the best qualities of hydrophobic and hydrophilic, or hygroscopic, materials..
WHAT WE HAVE LEARNED
What have we learned about optic materials overall with time? The following principles have been well elucidated and understood:
• An IOL is a total package. That is, its physical characteristics and the IOL material itself are important in how the lens functions in the eye. It is important to distinguish what is a material effect and what is a physical effect. This can often be difficult to differentiate.
• We like to simplify the material categorization of IOLs so that one size fits all. However, we now know that there are differences in each material category, so that one material is not necessarily going to act like another in regard to, for instance, inflammatory signs or capsular contracture. As another example, the propensity for glistenings in an optic material can be vastly different between two lenses that are both categorized as hydrophobic acrylic.7 Also, even though calcification has largely been a problem associated with hydrophilic acrylic IOLs, we now know that some hydrophilic acyclic lenses seem to be very, if not completely, resistant to this phenomenon. We must be careful about lumping lenses together in a single category.
As time goes on, it is to be hoped that we will get a better handle on the best characteristics of each material, in combination with the best physical characteristics, in order to improve the overall quality of IOLs in the future.
DOCUMENTED MATERIAL DIFFERENCES
Considering all of the foregoing, what can we say today about the IOL materials themselves in regard to their qualities as components of an IOL? The best summary of the latest information is as follows:
Silicone. Silicone IOLs, if they have a truncated edge, can be associated with an excellent and possibly even superior PCO-prevention profile. Some of the latest silicone materials have been associated with no more inflammation than hydrophobic acrylic IOL materials and no greater anterior capsular contracture.5,6
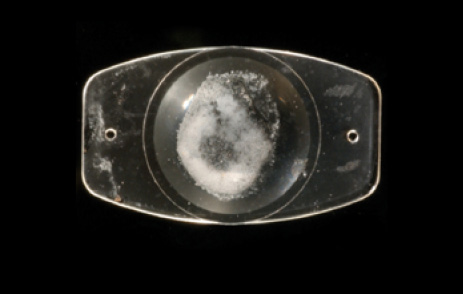
Figure 2. Calcification of a plate silicone IOL from an eye with asteroid hyalosis.
Unique problems are associated with silicone IOLs. Silicone oil tends to obscure the view of the posterior segment with all silicone IOLs; this continues to be a consideration for why such lenses should not be used in some patients.8 Silicone IOLs also have been uniquely associated with calcification in any eye with asteroid hyalosis (Figure 2). Although this is an uncommon problem, it is a relative contraindication to use of silicone IOLs.9 In addition, due most likely to the material’s lower refractive index, silicone IOLs seem to be more forgiving in regard to pseudophakic dysphotopsia than many acrylic IOLs.10
Hydrophobic acrylics. In the United States at least, hydrophobic acrylics are far and away the most common IOL materials used today. These are very well-accepted materials, and, with a truncated edge design, these IOLs have been very resistant to PCO. Some IOL materials in this category have shown a propensity for glistening formation inside the lens optic; however, other materials have not.7 This is likely due to the low water content of these hydrophobic materials; the lower the water content when the IOL is fully saturated, the greater the likelihood of glistenings.
In my opinion, these materials have generally been associated with more pseudophakic dysphotopsia than other materials; however, this may have been related more to their higher refractive index and flatter anterior curvature, as well as untreated, truncated IOL edges.
The hydrophobic materials tend to be brittle, and, if inappropriately handled, they can experience surface damage and even develop cracks in the optic; however, modern insertion devices have essentially eliminated this problem clinically.
Long-term studies of hydrophobic acrylic IOLs with similar physical characteristics have shown no difference in PCO development, but differences in anterior capsular opacification (ACO) have been seen.11 ACO has become more relevant due to increasing concerns about capsular contracture and late dislocation of IOLs still in the capsular bag. Some IOLs in this category have also shown a surface roughening phenomenon that appears to worsen with time, while other hydrophobic acrylic IOLs seem to avoid this problem.12 As with glistenings, the impact of this development on visual quality remains controversial.13
Hydrophilic acrylics. For hydrophilic acrylic IOLs with truncated edge designs, PCO is quite limited. However, all else being equal, it appears that PCO is more common with hydrophilic acrylic IOL materials than with other material categories.14
This group of materials has also shown the tendency to develop calcification.15 It seems that the latest generation of hydrophilic acrylic IOLs is less likely to experience this problem; however, new cases of calcification of hydrophilic acrylic materials keep appearing. The latest risk appears to be associated with the use of intraocular gases.16,17
These materials are certainly the most forgiving in regard to maintaining a good view when silicone oil is used in the eye,8 and they are probably the most biocompatible of all of the materials for use in eyes with chronic uveitis.18
PMMA. PMMA lenses are rarely used today in the United States, although they are still commonly used elsewhere in the world. These rigid materials have an excellent long-term track record, and, with a truncated edge design, they are very protective against PCO.
Some injection-molded PMMA lenses develop very severe glistening.19 By and large, however, this material has been well accepted with its only downside being the need for a larger incision because PMMA is not foldable.
CONCLUSION
We are still learning about differences in IOL materials. We must do a better job of differentiating the effects of physical characteristics of the IOL design from effects due to the material. We also must also be very careful not to assume that one lens in any category is going to behave the same as others in that category; we have seen significant differences over time as these materials have been refined.
1. Fritch C. Ne-YAG laser damage to glass intraocular lens. Ann Ophthalmol. 1984;16(12):1177.
2. Nishi O, Yamamoto N, Nishi K, Nishi Y. Contact inhibition of migrating lens epithelial cells at the capsular bend created by a sharp-edged intraocular lens after cataract surgery. J Cataract Refract Surg. 2007;33:1065-1070.
3. Nishi O, Nishi K, Osakabe Y. Effect of intraocular lenses on preventing posterior capsular opacification: design vs material. J Cataract Refract Surg. 2004;30:2120-2126.
4. Hollick E, Spalton D, Ursell P, et al. Biocompatibility of poly(methyl methacrylate), silicone, and AcrySof intraocular lenses: randomized comparison of the cellular reaction on the anterior lens surface. J Cataract Refract Surg. 1998;24:361-366.
5. Samuelson T, Chu Y, Krieger R. Evaluation of giant cell deposits on foldable intraocular lenses after combined cataract and glaucoma surgery. J Cataract Refract Surg. 2000;26:817-823.
6. Vock L, Crnej A, Findl O, et al. Posterior opacification in silicone and hydrophobic acrylic intraocular lenses with sharp-edged optics six years after surgery. Am J Ophthalmol. 2009;147:683-690.
7. Gregori N, Spencer T, Mamalis N, Olson R. In vitro comparison of glistening formation among hydrophobic acrylic intraocular lenses. J Cataract Refract Surg. 2002;28:1262-1268.
8. Apple D, Federman T, Krolicki J, et al. Irreversible silicone oil adhesion to silicone intraocular lenses: a clinicopathological analysis. Ophthalmology. 1996;103(10):1555-1561.
9. Foot L, Werner L, Gills J, et al. Surface calcification of silicone plate intraocular lenses in patients with asteroid hyalosis. Am J Ophthalmol. 2004;137:979-987.
10. Daynes T, Spencer TS, Doan K, Mamalis N, Olson R. Three-year clinical comparison of 3-piece AcrySof and SI-40 silicone intraocular lenses. J Cataract Refract Surg. 2002;28:1124-1129.
11. Kahraman G, Amon M, Ferdinaro L, et al. Intraindividual analysis of capsule opacification after implantation of 2 hydrophobic acrylic intraocular lens models: three-year follow-up. J Cataract Refract Surg. 2015;41:990-996.
12. Werner L, Morris C, Liu E, et al. Light transmittance of 1-piece hydrophobic acrylic intraocular lenses with surface scattering removed from cadaver eyes. J Cataract Refract Surg. 2013;39:114-120.
13. Henriksen B, Kinard K, Olson R. Effect of intraocular lens glistening size on visual quality. J Cataract Refract Surg. 2015;41:1190-1198.
14. Hollick E, Spalton D, Ursell P, et al. Posterior opacification with hydrogel, polymethylmethacrylate, and silicone intraocular lenses: two year results of a randomized prospective trial. Am J Ophthalmol. 2000;129:577-584.
15. Kleinman G, Mamalis N, Apple D, Assia E. Opacification of the Acryl C 160 hydrophilic acrylic intraocular lens. J Cataract Refract Surg. 2006;32:367-368.
16. Ventura B, Ventura M, Werner L, et al. Calcium deposits on hydrophilic acrylic IOLs. J Cataract Refract Surg. 2013;39:142-143.
17. MacLean K, Apel A, Wilson J, et al. Calcification of hydrophobic acrylic intraocular lenses associated with intracameral air injection. J Cataract Refract Surg. 2015;41:1310-1314.
18. Abela-Formanek C, Amon M, Schauersberger J, et al. Uveal and capsular biocompatibility of two foldable acrylic lenses in patients with uveitis or pseudoexfoliation syndrome. J Cataract Refract Surg. 2002;28:1160-1172.
19. Wilkins E, Olson R. Glistening with long-term follow-up of the Surgidev B20/20 polymethylmethacrylate intraocular lens. Am J Ophthalmol. 2001;132:783-785.
Qualities of the Ideal IOL Material
Progress in materials engineering has enabled advances in cataract surgery.


By Manfred R. Tetz, MD; and Matthew R. Jorgensen, PhD
The first modern use of a designed foreign object to replace the natural human lens during cataract surgery was by Sir Harold Ridley.1 The operation came following the suggestion of an intern, who asked why he was not replacing the lens he had just removed. The choice of material for replacement was directed by Ridley’s experience during wartime removing benign PMMA shrapnel from pilots’ eyes.
.jpg)
Figure 3. One of the first flexible IOLs implanted by Edward Epstein in 1968(human cadaver eye).
That episode began a long and exciting search for materials that could be utilized in IOLs. As understanding of the relationship between IOL material and performance has matured, the engineering of materials with targeted properties has flourished. Choice of materials affects the design, manufacture, insertion, and performance of IOLs, making it one of the central decisive factors in successful IOL implantation.
Following Ridley’s early attempts, the years of trial and error in IOL design were difficult, leading many ophthalmologists to abandon the idea altogether. However, the practice of IOL implantation was revolutionized in the 1980s with the introduction of phacoemulsification; this allowed much smaller incisions and motivated the use of IOL materials that could be folded.2,3 Thomas Mazzacco designed and then began implanting a foldable plate-haptic silicone IOL, the so-called Mazzocco taco (Figure 3).4
Since that time, the range of available IOL materials has expanded to include hydrophilic (more appropriately called hygroscopic) acrylics, hydrophobic acrylics, and, most recently, hybrid materials with specially designed chemistry.
THE IDEAL MATERIAL
Ideally, an IOL material should be biocompatible and flexible; should have a high refractive index, a high surface hardness, and an equilibrium of water and salt content that mirrors the ocular environment; and it should be capable of manufacture with a sharp optic edge.5 The gold standard would, of course, achieve the optimum values of all of these properties; however, this ideal has not yet been reached, as every available material has advantages and disadvantages. Nevertheless, recent progress in material sciences has brought IOLs closer than ever to this goal.
Silicone lenses are flexible, have a very hydrophobic surface that resists posterior capsular opacification (PCO), and have recently been shown to have the sharpest edges.6,7 Unfortunately, early silicone IOLs were reported to have issues with discoloration and calcification associated with certain eye conditions.8,9 Although more modern silicone IOLs appear to have addressed these problems, their track record in the literature has created a lasting aversion.
Flexible hydrophilic or hygroscopic acrylic lenses more closely mimic biologic tissue due to their high water content, which ranges from 18% to 34%.10 Although they are hygroscopic, they may, like silicone, have hydrophobic surfaces that lead to low PCO rates.11 Due to their high water content, they regrettably have lower refractive indices and therefore are thicker lenses. On the other hand, they also reduce glare and other aberrations. Early hygroscopic acrylic lenses were too soft, which led to complications including internal and external calcification and secondary cataract formation.12 Because of these past complications, trust in the performance of hygroscopic acrylic materials has been damaged.
Hydrophobic acrylic lenses were developed at least in part to address the issues that had arisen with hygroscopic lenses, and these are the most widely used IOLs today. One-piece and three-piece hydrophobic lenses can be inserted through very small incisions without damage. Although they are not as good as silicone or hydrophilic materials at resisting PCO, their performance is still much better than that of PMMA.13
These characteristics make modern hydrophobic lenses a tribute to the benefits of innovation in the fields of materials science and cataract surgery. Although the state of the art is good, further improvements can still be made to improve PCO rates, increase surface hardness, and reduce glistenings.
HYBRID MATERIALS
The newest IOL materials combine the best features of hydrophobic and hygroscopic lenses using hybrid materials. Hygroscopic components are included to produce the desired equilibrium of water content (important to reduce glistenings), while hydrophobic elements allow flexibility and control the refractive index.5 One example, the enVista (Bausch + Lomb), appears to have eliminated the formation of glistenings while improving surface hardness.14,15 It is expected that other lenses with superior properties are in development.
CONCLUSION
Since the development of the first IOLs, progress in materials engineering has enabled advances in cataract surgery that continue to improve patient vision. As new surgical technologies are pioneered, and the gold standard definition of the ideal IOL evolves, the development of IOL materials will hold a central role in the future of cataract surgery.
1. Ridley H. Intra-ocular acrylic lenses: a recent development in the surgery of cataract. Br J Ophthalmol. 1952;36(3):113.
2. Choyce DP. Discussion to Barraquer: anterior chamber plastic lenses. Results of and conclusions from five years experience. Trans Ophthalmol Soc UK. 1959;79.
3. Apple DJ, Escobar-Gomez M, Zaugg B, Kleinmann G, Borkenstein AF. Modern cataract surgery: unfinished business and unanswered questions. Surv Ophthalmol. 2011;56(6):S3-S53.
4. Epstein E. History of intraocular lens implant surgery. Thorofare, NJ: Slack Inc; 1986.
5. Tetz M, Jorgensen MR. New hydrophobic IOL materials and understanding the science of glistenings. Curr Eye Res. 2015;40(10):969-981.
6. Cook CS, Peiffer Jr RL, Mazzocco TR. Clinical and pathologic evaluation of a flexible silicone posterior chamber lens design in a rabbit model. J Cataract Refract Surg. 1986;12(2):130-134.
7. Buehl W, Menapace R, Findl O, Neumayer T, Bolz M, Prinz A. Long-term effect of optic edge design in a silicone intraocular lens on posterior capsule opacification. Am J Ophthalmol. 2007;143(6):913-919.
8. Koch DD, Heit LE. Discoloration of silicone intraocular lenses. Arch Ophthalmol. 1992;110(3):319-320.
9. Stringham J, Werner L, Monson B, Theodosis R, Mamalis N. Calcification of different designs of silicone intraocular lenses in eyes with asteroid hyalosis. Ophthalmology. 2010;117(8):1486-1492.
10. Tehrani M, Dick HB, Wolters B, Pakula T, Wolf E. Material properties of various intraocular lenses in an experimental study. Ophthalmologica. 2004;218(1):57-63.
11. Cunanan CM, Tarbaux NM, Knight PM. Surface properties of intraocular lens materials and their influence on in vitro cell adhesion. J Cataract Refract Surg. 1991;17(6):767-773.
12. Werner L, Apple DJ, Escobar-Gomez M, et al. Postoperative deposition of calcium on the surfaces of a hydrogel intraocular lens. Ophthalmology. 2000;107(12):2179-2185.
13. Abela-Formanek C, Amon M, Schild G, Schauersberger J, Heinze G, Kruger A. Uveal and capsular biocompatibility of hydrophilic acrylic, hydrophobic acrylic, and silicone intraocular lenses. J Cataract Refract Surg. 2002;28(1):50-61.
14. Packer M, Rajan M, Ligabue E, Heiner P. Clinical properties of a novel, glistening-free, single-piece, hydrophobic acrylic IOL. Clin Ophthalmol. 2014;8:421.
15. Mentak K, Martin P, Elachchabi A, Goldberg E. Nanoindentation studies on hydrophobic acrylic IOLs to evaluate surface mechanical properties. Paper presented at: XXV Congress of the European Society of Cataract and Refractive Surgeons; September 8-12, 2007; Stockholm.
Matthew R. Jorgensen, PhD
• Material Scientist, Group Leader, Institute for Integrative Nanosciences, Dresden, Germany
• matthewrayjorgensen@gmail.com
• Financial disclosure: None
Randall J. Olson, MD
• Chairman, Department of Ophthalmology and Visual Sciences
• CEO, John A. Moran Eye Center
• randallj.olson@hsc.utah.edu
• Financial disclosure: None
Manfred R. Tetz, MD
• Director, Eye Center-Spreebogen, Berlin
• Scientific Director, Berlin Eye Research Institute
• Member, CRST Europe Editorial Board
• info@augentagesklinik-spreebogen.de
• Financial disclosure: None
