Consider Multiple Factors Before Explanting an IOL
The surgical approach should avoid inducing high postoperative astigmatism and should respect the endothelium.


By Simonetta Morselli, MD; and Antonio Toso, MD
We take several factors into account when we think that an IOL should be explanted. The patient is our first consideration: age, occupation, refraction of the eye in question, refraction of the contralateral eye, and the patient’s own visual expectations and needs. The anatomic condition of the eye is a second consideration. This includes IOL centration, visual acuity, aberrometric analysis, endothelial cell count, integrity of the capsular bag, timing of IOL implantation, IOL position in the eye, and status of the retina.
In our experience in these cases, if an IOL must be explanted, it should be replaced with another IOL. We consider that the best option if it is decided that explantation is necessary. There are many potential justifications for explanting an IOL, including the following:
• Incorrect power calculation
• Opacification of the IOL optic
• Intolerable glare or halos
• IOL tilting due to contraction of the capsular bag
• Intraoperative explantation during vitreoretinal surgery
• Capsular bag dislocation
INCORRECT IOL POWER
In cases of incorrect power calculation, there are two scenarios:
Unanticipated incorrect power immediately after surgery. This issue is due to effective position of the IOL in the eye. In some cases, due to anatomic factors in the eye, the capsular bag is not in the usual position. Because of this, the A-constant is different, and the results of standard IOL power calculations will not provide the expected result.
Anticipated incorrect IOL power after refractive corneal surgery. In patients who have previously undergone corneal refractive surgery, even if some preoperative refractive corneal data were known, most of the time the IOL power after cataract surgery is incorrect. This can happen even if we use all the formulas that are now available to calculate IOL after PRK, LASIK, or LASEK.
THE BEST OPTION?
When halos and glare with multifocal IOLs or hyper-aspheric IOLs affect the patient’s (and the surgeon’s) psychological status, the best option is to explant the IOL, even if the patient’s visual acuity is 20/20. In case of IOL optic opacification, there is no other option. If, during posterior segment surgery or combined anterior-posterior segment surgery, the posterior capsule is damaged—by, for example, the vitrector—there is no other option but to explant the posterior chamber IOL and insert a sulcus- or iris-fixated IOL.
NOT THE BEST OPTION?
Explantation is not the best option when we are unable to explain to the patient the consequences of explanting the IOL, or when the patient does not understand that the surgery is very risky. Some patients think that an IOL exchange is easy, and they would like to exchange their IOL due to a very low incorrect power or some occasional glare. In this case, and especially in the presence of very good visual acuity in the eye in question, we try to not encourage the IOL explant.
SURGICAL TECHNIQUE
In the event of IOL explantation, we pay attention to several factors in planning surgery. We try not to induce high postoperative astigmatism, and we also try to respect and protect the corneal endothelium as much as possible.
In order to accomplish these goals, the incision must comfortably accommodate IOL explantation. In our opinion, the best approach is to create a sclerocorneal 5-mm frown incision. This technique provides enough space to explant the IOL without damaging tissues, while still inducing a very low amount of astigmatism.
We also make sure to have more than one viscoelastic substance available: a cohesive OVD to protect tissues and a high-molecular-weight OVD to be used as an instrument to open the capsular bag and separate the IOL from capsular bag adhesions.
The surgeon should also have on hand 20-gauge vitreoretinal scissors when fibrosis makes it necessary to cut the IOL, leave the haptic loops in the capsular bag, and explant only the IOL optic.
The timing of explantation depends on the nature of the problem, but, if explantation is necessary, we prefer to operate as soon as possible.
CONCLUSION
Before explanting an IOL, we try to consider as many factors as possible in order to achieve the best results for our patient, and we tend to weigh the pros and cons before explanting and replacing an IOL.
Best Approach Depends on Clinical Situation, Patient Factors
Careful technique is mandatory when the IOL is strongly adhered to the capsule.

By Nic J. Reus, MD, PhD
In making the decision whether to exchange an IOL, one should take into account a number of factors, including the following:
• The complaints and expectations of the patient;
• The expected benefits of IOL exchange;
• The potential complications of intraocular surgery (eg, endophthalmitis, choroidal hemorrhage, and cystoid macular edema);
• The presence of a posterior capsulotomy (conveying an increased risk of vitreous loss); and
• The possibility of using any surgical or nonsurgical alternatives to IOL exchange (eg, laser refractive surgery or wearing glasses or contact lenses in case of a refractive surprise).
WHEN TO DO IT
In general, I favor explanting an IOL when it is subluxated or dislocated. This malposition may have occurred due to loss of zonular support in the presence of pseudoexfoliation or trauma. Subluxation may also be the result of insufficient capsular support for a foldable IOL due to a capsule tear, the severity of which was underestimated during surgery.
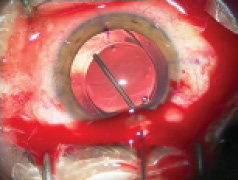
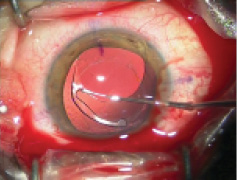
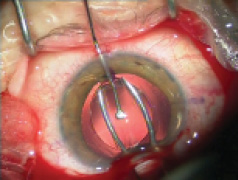
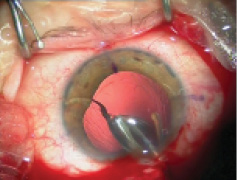
Figure 1. Explantation of a one-piece foldable IOL from thebag: Separate the lens capsule from the IOL carefully using OVD (A). Once it is free from the capsule, luxate the IOL into the anterior chamber (B). Insert a spatula though an extra sideport at the 6-o’clock position and fold the IOL with IOL insertion forceps with biconvex tips over the spatula (C). Rotate the IOL sideways and remove it from the eye (D).
Another reason to remove a lens is the inappropriate implantation of a one-piece foldable IOL in the ciliary sulcus, which might cause uveitis, glaucoma, and hyphema if left in place.
Explantation of an IOL may also be indicated when a patient is severely dissatisfied with the type of IOL implanted. Although most patients become accustomed to dysphotopsias associated with multifocal lenses over time, some patients prefer an exchange of the multifocal IOL for a monofocal one.
In case of myopic or hyperopic refractive surprise, exchange of the IOL is also an option, especially when there is insufficient space for implantation of a nontoric IOL in the sulcus.
Some patients may be dissatisfied with their postoperative cylinder after having received a nontoric IOL. If they are not eligible for laser refractive correction, exchange for a toric IOL in the capsular bag is a viable alternative.
WHAT TO DO
In case of insufficient capsular support, I favor implantation of an iris-fixated IOL over scleral fixation of a one-piece foldable IOL. Iris-fixated lenses are readily clipped to the iris, either prepupillary or retropupillary, and their implantation technique is easy to learn.
In the case of a foldable IOL with good capsular support, I often choose to exchange it for a foldable IOL in the bag. To this end, the anterior lens capsule has to be separated from the IOL optic by injecting a dispersive OVD between the two layers (Figure 1). If the anterior capsule is adhered too tightly to the optic, a 23-gauge needle or microvitreoretinal blade can be used to mechanically lift the capsule from the optic. The OVD can then be injected underneath the capsule to separate the layers.
This procedure must be done gently but thoroughly. The ends of the haptics, particularly, may be encapsulated tightly because of capsular fibrosis. In this case, one may have to scrape off the capsule from the haptic using a spatula while fixating the optic with a second instrument. Once the IOL can be freely rotated in the bag, the IOL is luxated into the anterior chamber.
I then often prefer to fold the IOL in the anterior chamber, allowing it to be explanted through an incision of approximately 3.2 mm. To this end, with the main incision at the 12-o’clock position, I create an extra sideport incision at 6-o’clock. Next, I insert a spatula through the sideport incision and IOL insertion forceps with biconvex tips through the main incision. While holding the spatula underneath the center of the IOL, I fold the optic over the spatula using the IOL insertion forceps. Next, I rotate the IOL sideways and remove it from the eye. The new IOL is then inserted into the bag, and all OVD is removed from the anterior chamber. Of course, one has to make sure the incisions are watertight; otherwise, a suture is placed on any leaking incision.
CONCLUSION
There are many reasons for explanting an IOL. The need for this may be minimized with adequate preoperative counseling of the patient, careful surgery, and discussing possible alternatives to IOL exchange with the patient. However, if one does need to perform IOL exchange, there are numerous surgical options that may be performed safely and provide a good outcome for both patient and doctor.
The Best Approach Depends on Time Since Implantation
Whatever the reason for IOL explantation, patient motivation is key to success.

By Abhay R. Vasavada, MS, FRCS
IOL explantation and/or exchange may be performed for a variety of indications:
• A poorly placed IOL: Often, during primary surgery, there may be anterior or posterior capsule ruptures that may result in asymmetric bag-sulcus fixation or a grossly decentered IOL (Figure 2).
• Late IOL dislocation or decentration: This is a common problem with progressive zonulopathies, such as pseudoexfoliation syndrome and Marfan syndrome, or with trauma in an otherwise normal eye.
• Inaccuracy in IOL power calculation: Postoperative refractive surprises are a major reason for IOL explantation.
• Dissatisfied multifocal IOL patient: This can occur despite a perfect technical outcome.
Whatever the reason for IOL explantation, certain factors must be considered. The primary preoperative consideration is whether the patient is motivated to undergo the procedure. In-depth counseling regarding possible surgical plans, the purpose of surgery, alternative management options, and possible complications is a must. Preoperative examinations should include pseudophakic biometry, preferably optical; corneal astigmatism assessment including the posterior cornea (with Scheimpflug imaging); and a retinal evaluation, especially posterior segment OCT.
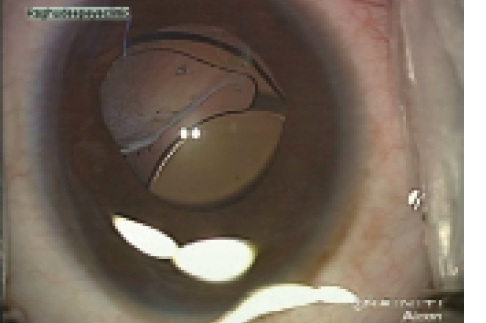
Figure 2. Dislocated IOL due to inadequate capsular support.
Two methods of testing that I find particularly valuable in these cases are wavefront aberrometry and ultrasound biomicroscopy (UBM). UBM is very useful, especially in cases in which sulcus IOL placement, with or without optic capture, may be planned. UBM will reveal the amount of capsular support and the extent of peripheral Soemmering ring (Figure 3), which is important, as this can reduce the sulcus space and cause chronic rubbing of the IOL against the posterior surface of the iris. Aberrometers that measure internal eye aberrations are valuable, especially when patients are dissatisfied with their vision.
Another important consideration for IOL explantation is the integrity of the capsular bag. In cases with anterior capsular tears or even preexisting laser posterior capsulotomy, I am wary of advising IOL explantation, and I would counsel the patient about potential risks if he or she opts for the procedure. I also think it is imperative to arrange
to have a backup three-piece IOL and PMMA IOL for scleral fixation in the appropriate power and a vitrector available in the eventuality of a posterior capsule rupture for every case.
Postoperatively, patients should be monitored for peripheral retinal breaks and for glaucoma. With sulcus-placed, scleral-fixated, or intrascleral-fixated IOLs, monitoring for glaucoma and uveitis is especially important.
.jpg)
Figure 3. Ultrasound biomicroscopy in an aphakic eye, which shows adequate sulcus space to plan sulcus fixation of an IOL.
BEST- and WORST-CASE SCENARIOS FOR IOL EXPLANTATION
Eyes that are less than 4 weeks out from surgery and have an intact capsular bag with a moderately sized anterior capsulorrhexis are ideal, as these cases allow easy explantation of IOLs. Patients who have refractive surprises are the easiest to explant and to satisfy.
By contrast, patients who have either a posterior capsular rupture or a large Nd:YAG laser capsulotomy are the most difficult, as they may require vitrectomy. Quite often, a grossly decentered or dislocated lens in the vitreous requires comanagement with a retinal surgeon.
TIMING
Explantation should be performed as early as possible after primary surgery. An ideal time would be within
6 weeks of primary surgery, so that very severe anterior capsule fibrosis has not set in. Therefore, it is crucial for the surgeon to examine the patient within 2 weeks postoperative, at the latest.
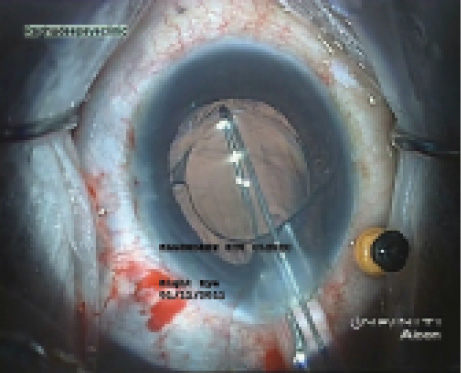
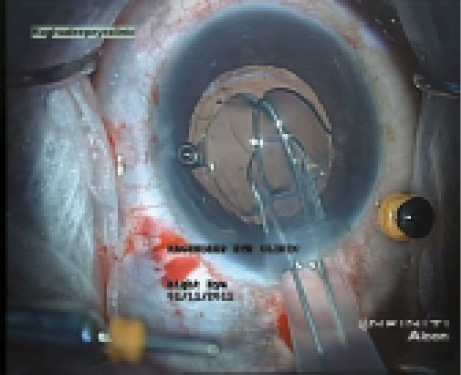
Figure 4. A snare device devised by Arup Bhaumik, MD, hooksan IOL that is to be explanted (A); the IOL is folded and drawn into an A cartridge (B), which allows IOL explantation without enlarging the original 3.2-mm incision.
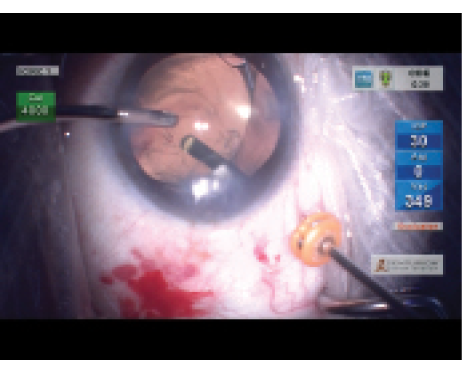
Figure 5. Pars plana vitrectomy is performed in an eye with a dislocated IOL.
There is a word of caution, however, in the case of unhappy multifocal IOL patients. Often, these patients adapt to dysphotopic phenomena after a few months. Also, it should be kept in mind that, if IOL explantation is contemplated, one should not hurry to perform a YAG capsulotomy, as this will complicate the corrective surgery.
TIPS FOR IOL EXPLANTATION
Scenario A: Intact Capsular Bag. If explantation is undertaken in the early postoperative period, it is easy to open the original incision and to dissect the capsular bag using a blunt spatula. If the capsulorrhexis margin is firmly adherent to the IOL optic, I find that a 26-gauge unbent cystotome needle is very efficient for opening the bag space. It is imperative to use either a dispersive (eg, Viscoat, Alcon) or a pseudodispersive (eg, Healon 5, Abbott Medical Optics) OVD to keep the capsular bag and the corneal endothelium protected during maneuvers.
After the capsule margin is opened, OVD is injected gently in multiple quadrants, and the entire capsular bag is opened up. The IOL is brought out from the bag into the anterior chamber. I find the use of a modified snare device (designed by Arup Bhaumik, MD, of Kolkata, India) very useful for this maneuver. It allows a one-piece foldable IOL to be folded inside an A cartridge and explanted through a 3.2-mm incision without cutting the IOL (Figure 4).
Scenario B: Capsular Bag Not Intact/Dislocated Bag-Lens Complex. As time passes, we have been seeing more instances of IOL–capsular bag complex dislocation 10 or more years after cataract surgery. Some of these have visible pseudoexfoliation, while others do not. In these cases, the surgical strategy is very different from cases that need an explant or exchange for refractive surprises or unwanted visual phenomena. It is good practice to discuss the surgical options with colleagues and with a retinal surgeon. If it is feasible, comanaging with the retinal surgeon (Figure 5) should be done, as this strategy reduces posterior segment complications and the risk of glaucoma.
It is most important to perform an adequate vitrectomy, and I recommend using a pars plana approach, as this allows adequate vitrectomy. In the event of a dislocated three-piece IOL, the same IOL may be kept inside the eye, and, with the help of microincision instrumentation, intrascleral glued IOL fixation may be performed without the need to create large incisions. One-piece foldable IOLs, on the other hand, must be explanted and exchanged with an appropriate three-piece scleral-fixated IOL.
CONCLUSION
Patients who undergo vitrectomy, whether limbal or pars plana, during IOL exchange, should have the retina checked by a retina specialist the next day. All of these patients should be carefully monitored postoperatively every 6 months or annually.
Eliminate All Exacerbating Factors Before Discussing Surgery
If explantation is a must, technique depends on type of IOL.

By Jan Venter, MD
In some circumstances, it is quite obvious that lens exchange is inevitable, such as when an IOL is severely opacified, or when a decentered or displaced IOL is unsuitable for iris or scleral fixation. In such situations, surgeons can most often make the correct judgment call easily. In my experience, the worst cases in which to make an explantation decision are those in which the patient reports severe visual phenomena from a multifocal IOL, but the IOL itself is perfectly centered and clear.
We previously published outcomes of 9,366 eyes with a refractive segmented multifocal IOL.1 We found that, in a small percentage of patients (55 eyes, 0.59% of the whole study cohort), visual phenomena were bothersome to the point that the patients could no longer function, and the IOL had to be exchanged for a different design. In our practice, we have performed a total of 47,662 multifocal IOL implantations with various designs (refractive and diffractive), and IOL explantation due to quality of vision issues was necessary in 0.67% of cases.
I normally encourage patients to wait at least 6 to 12 months for neural adaptation to take place, but I also emphasize that the final decision should be made within the first 2 postoperative years; otherwise, lens exchange may become too difficult. Prior to lens exchange, I look for all possible factors that could be exacerbating visual phenomena and quality-of-vision issues, such as residual refractive error, capsular opacification, position of the IOL relative to the pupil, cystoid macular edema, and corneal or ocular surface disease. If all of these are eliminated or treated and the symptoms still persist, I start discussing lens exchange. Capsular opacification is one of the worst factors influencing treatment decisions, as it can severely affect quality of vision, but, if treated with Nd:YAG laser posterior capsulotomy, it can also significantly complicate future lens exchange.
PREOPERATIVE CONSIDERATIONS
Before lens explantation, the original IOL should be thoroughly examined. Attention should be paid to the type of the IOL, haptic design, size of the anterior capsulotomy and the degree to which it covers the posterior chamber, pseudophacodonesis (which can suggest zonular weakness), health of the corneal endothelial cells, and whether Nd:YAG capsulotomy has been performed.
SURGICAL TECHNIQUE
When IOL exchange is performed, the pupil should be maximally dilated. I normally create 1-mm stab incisions at the 6-, 9-, 12-, and 3-o’clock positions and fill the eye with an OVD. I then open the capsular bag with an OVD on a blunt viscoelastic cannula. It is important to pay attention to the peripheral insertion of the haptics into the capsular fornix; a dispersive OVD should be used to open this area. If a haptic is heavily fibrosed, I sometimes cut it and leave it in place after removal of the optic.
When exchanging a C-loop multifocal IOL after YAG capsulotomy, I normally displace the IOL into the anterior chamber and insert a three-piece monofocal IOL into the capsular bag while the original IOL is still in the anterior chamber. This technique helps to reduce the risk of vitreous loss. Once the monofocal lens is well positioned, I grasp the original C-loop IOL from the side using microforceps (D.O.R.C.). Then, using Osher IOL cutting scissors (Storz Ophthalmics), I cut the lens through 90% of its length. Next, I grasp the haptic and rotate half of the almost-bisected lens out of the wound. The lens is then rotated 90°, after which the trailing half of the lens can be rotated out of the eye.
Explanting a multifocal IOL with a plate haptic, I first displace the inferior haptic into the anterior chamber, leaving the superior haptic in the bag. Using the D.O.R.C. microforceps and the Osher IOL cutting scissors, I cut the plate lens through 100% of its length. The two pieces are then removed from the eye one at a time.
CONCLUSION
Patients with premium IOLs have high expectations for complete spectacle independence. Exchanging one of these lenses due to poor quality of vision is never an easy decision. Thorough counseling is a necessity in these cases, and all factors that can possibly be exacerbating the visual phenomena should be eliminated before committing to a lens exchange.
1. Venter JA, Pelouskova M, Collins BM, Schallhorn SC, Hannan SJ. Visual outcomes and patient satisfaction in 9366 eyes using a refractive segmented multifocal intraocular lens. J Cataract Refract Surg. 2013;39(10):1477-1484.
Simonetta Morselli, MD
• Head of the Ophthalmology Department, S. Bassiano Hospital, Bassano del Grappa, Italy
• Member, CRST Europe Editorial Board
• simonetta.morselli@gmail.com
• Financial disclosure: None
Nic J. Reus, MD, PhD
• Ophthalmologist, Amphia Hospital, Department of Ophthalmology, Breda, Netherlands
• nreus@amphia.nl
• Financial disclosure: Consultant (Alcon), Research support (i-Optics))
Antonio Toso, MD
• Consultant in the Vitreoretinal Surgical Department, Ophthalmology Department, San Bassiano Hospital, Bassano del Grappa, Italy
• antonio.toso@gmail.com
• Financial disclosure: None
Abhay R. Vasavada, MS, FRCS
• Director, Raghudeep Eye Clinic, Iladevi Cataract & IOL Research Centre, Gujarat, India
• icirc@abhayvasavada.com
• Financial Disclosure: Research support (Alcon)
Jan Venter, MD
• Emeritus Medical Director, Optical Express, London
• drjanventer@gmail.com
• Financial disclosure: None

