Why I Prefer Toric IOLs Over LRIs
If all steps of the evaluation, calculation, and surgery are optimized, great results can be achieved every time.


By Edoardo Ligabue, MD; and Cristina Giordano, OD
The correction of astigmatism during cataract surgery has always been an area of interest for our practice. From 1988 to 1995, Dr. Ligabue performed cataract surgery with an incision size of 5.5 mm to implant rigid PMMA IOLs. At that time, postoperative astigmatism was not considered to be of significant importance; the incision was simply placed on the steepest corneal axis.
In 1995, the use of foldable lenses allowed reduction of the width of the incision, first to 3.2 mm and then to 2.2 mm. Dr. Ligabue also started to perform corneal limbal relaxing incisions (LRIs) using the nomogram developed by Louis D. “Skip” Nichamin, MD.1 The technique was attractive given that, apart from corneal surgery with the excimer laser, there were no real alternative techniques for astigmatism reduction until the introduction of toric IOLs in 2004.
LRI NOMOGRAMS
LRI nomograms take into account the amount and type of astigmatism (with the rule or against the rule) and the patient’s age. Perfect execution of the corneal incisions using appropriate markers is required. After a learning curve, every surgeon adapts his or her technique to the nomogram. Still, varying corneal response due to a number of factors—including tissue elasticity, corneal thickness, and limbus size—yield results that are not always repeatable or reliable.
Patients who receive LRIs often complain of ocular foreign body sensation, of their eyes bothering them, or of discomfort in general.2 Typically, these complaints are in addition to those related to possible tear film decompensation and dry eye sensations that patients sometimes feel after cataract surgery.
With time, the relaxing effect of LRIs can increase, making the results unreliable in the long term. This instability is more evident with curved astigmatic incisions in clear corneas than with limbal incisions.3 The ability to perform these astigmatic curved incisions with a femtosecond laser has certainly improved their performance in terms of placement, shape, and depth; however, issues related to the unpredictability of corneal tissue response, frequent patient discomfort, and variations in corrective effect over time remain.
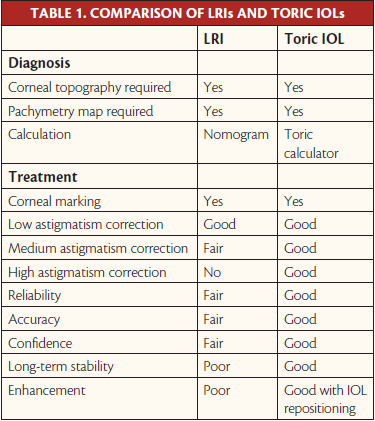
For these reasons, we welcomed the arrival of toric IOLs onto the market, as they allow us to correct astigmatism directly in the eye, resulting in the best visual quality. Since 2004, the use of toric IOLs has grown, to the point that they are now routinely implanted in patients undergoing premium cataract surgery. In our experience, toric IOLs offer several advantages over LRIs (Table 1) as long as patient evaluation, IOL calculation, and implantation are properly performed.
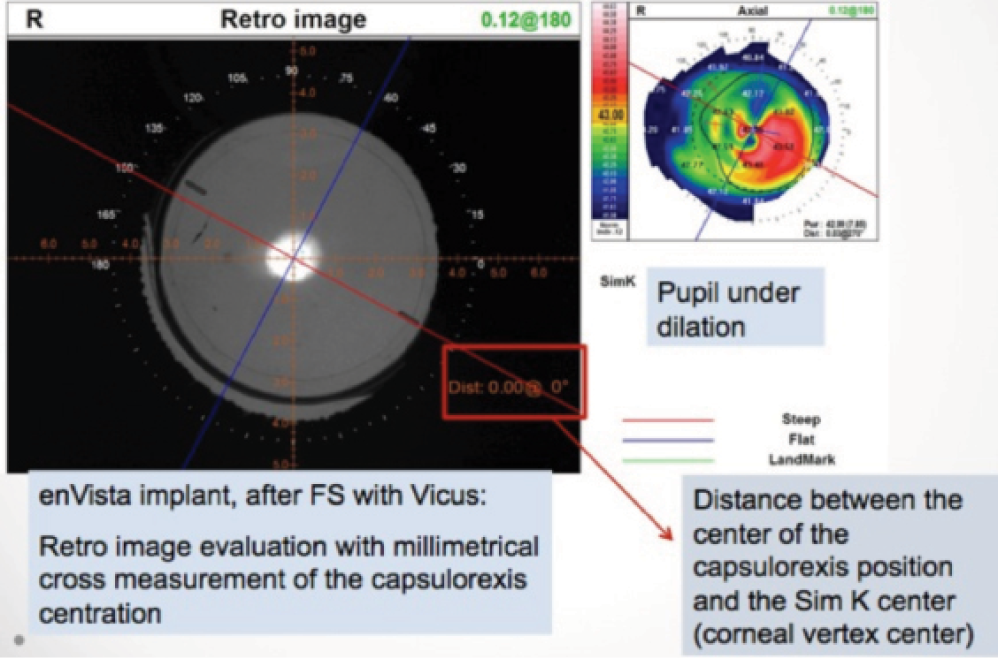
Figure 1. Evaluation of alignment and of capsulotomy centration.
INSIGHTS AND ADVANTAGES
Preoperative examination. In the routine preoperative examination of toric IOL candidates, careful evaluation of corneal topography and astigmatism is required. These measurements must be as accurate as possible in order to precisely determine the positioning axis.
.png)
Figure 2. An enVista Toric IOL behind a femtosecond lasercreated capsulotomy.
It is advisable to perform Scheimpflug corneal tomography, which allows assessment of the posterior surface of the cornea. Proper evaluation of the tear film and assessment for the presence of contact lens warpage are key in order to avoid artifacts and unreliable data collection.4
Marking. Marking is crucial for both LRI positioning and toric IOL alignment. Our routine marking system is to identify the axis 0° to 180° at a slit lamp with a chin rest, using a marker with bubble horizontal reference (E. Janach; axial marker JSL9000) before treatment with the femtosecond laser. The axis of alignment is then marked intraoperatively using a modified Mendez ring (E. Janach; model J2294.5).
Calculations. For LRIs, the nomogram used is always approximate, and it is impossible to take into account all of the variables involved, such as corneal response, actual cutting depth, distance of the corneal incisions from the center, etc.
For toric IOLs, I prefer to use the online calculator provided by the relevant lens manufacturer. I estimate only the keratometry reading of the anterior surface of the cornea because I believe the posterior cornea has little influence on outcomes. In reality, we still do not have a reliable calculation formula that also takes into account the posterior cornea. The available calculators are accurate if the refractive target is emmetropia; however, if the target is myopia, the calculation will result in an undercorrection of about 0.50 D or more.
Correction. Toric IOLs allow correction of 0.75 D to 10.00 D of astigmatism, depending on the lens and manufacturer. LRIs produce good results up to about 2.00 D of astigmatism. Beyond this value, the correction is variable, and it is difficult to exceed 3.00 D.
Reliability. In terms of reliability, accuracy, and confidence in results, toric IOLs are, in my experience, superior to LRIs because they require less approximation and craftsmanship of execution to achieve good results. In regard to long-term stability, toric IOLs are also in the lead. In fact, the rotational stability of these IOLs in the capsular bag is maintained even after 5 years.5
Cost. From a cost perspective, LRIs are less expensive than toric IOLs, but a high level of surgeon experience is required to achieve acceptable results. In contrast, toric IOLs are more expensive, but they can be used successfully by any ophthalmic surgeon.
Enhancements. In the event of an unsatisfactory result, LRIs can be extended in the case of an undercorrection; nothing can be done to fix an overcorrection. Toric IOLs can be repositioned in the event of a misalignment. If an enhancement is necessary, evaluation with an aberrometer such as the OPD Scan III (Nidek) is useful, as this can indicate exactly how many degrees the IOL is out of alignment6 (Figure 1).
.jpg)
Figure 3. Aberrometric evaluation of a perfect toric IOL implantation.
THE FEMTOSECOND LASER
A major advantage for premium and toric lens implantation came with the introduction of the femtosecond laser. Since 2012, we have been using the Victus femtosecond laser (Bausch + Lomb) for all premium cataract surgeries.
The preoperative routine includes evaluation of the crystalline lens density and the corneal curvature gradient. These measurements allow me to determine and set the appropriate energy for the laser treatment.
.jpg)
Figure 4. Rotational stability at 1 year with laser-assisted versus manual cataract surgery.
We developed a nomogram, which has been programmed into the laser software, to take into account the percentage of the lens density. This nomogram allows us to use as little energy as possible, reducing the bubbles generated during the laser treatment and increasing effectiveness. With this nomogram, we can identify cataracts that can be treated using the ZeroPhaco handpiece (Bausch + Lomb), as this instrument allows us to aspirate the nucleus of the lens without using ultrasound.
The use of the femtosecond laser to perform corneal incisions, capsulotomy, and lens fragmentation undoubtedly offers many advantages, including the following:
• Perfect incisions with standardized surgically induced astigmatism;
• A centered and perfectly circular capsulotomy;
• Stable and well-centered IOL positioning without lens tilt (Figure 2);
• Superior rotational stability in the capsular bag; and
• Excellent refractive predictability.
We compared 45 eyes implanted with the enVista Toric IOL (Bausch + Lomb) using the Victus femtosecond laser and 45 eyes implanted with the same lens using standard manual phacoemulsification. Aberrometric evaluation (Figure 3) of coma, point spread function, and modulation transfer function was slightly better in the femtosecond laser group, as was rotational stability at 1-year follow-up7 (Figure 4).
CONCLUSION
Today, cataract surgery requires superior refractive outcomes in the correction of astigmatism, and, in our opinion, only toric IOLs can guarantee these results.
However, in order to achieve success with toric IOLs, it is imperative that all of the necessary precautions be followed before and during implantation. Each measurement and each step can generate a small percentage of error. If all steps of the evaluation, calculation, and surgery are optimized, great results can be achieved every time. These outcomes will likely only continue to become more optimized and standardized with the incorporation of the femtosecond laser.
1. Nichamin LD. Corneal relaxing incisions. Cataract & Refractive Surgery Today. August 2009.
2. Loncar VL, Vickovic IP, Ivekovic R, Mandic Z. Limbal relaxing incision during cataract surgery. Acta Clin Croat. 2012;51(2):289-292.
3. Titiyal JS, Khatik M, Sharma N, et al. Toric intraocular lens implantation versus astigmatic keratotomy to correct astigmatism during phacoemulsification. J Cataract Refract Surg. 2014;40(5):741-747.
4. Ligabue E, Giordano C. Spotlight on enVista Toric, observing real life outcome. Paper presented at: ESCRS Winter Meeting, B+L Symposium; February 2013; Warsaw, Poland.
5. Charters L. Toric intraocular lenses, limbal relaxing incisions benefit astigmatism. Ophthalmology Times. August 2011.
6. Ligabue E, Giordano C. The results in visual quality of capsulorhexis position and centration with FLACS in toric IOL implant. Paper presented at: ESCRS Winter Meeting; February 2013; Warsaw, Poland.
7. Ligabue E, Giordano C. Optical quality and rotational stability of a new toric IOL in femtosecond cataract surgery. Paper presented at: the ESCRS Meeting; September 2013; Amsterdam, Netherlands.
Clinical and Economic Factors Favor Toric IOLs
Toric IOLs offer a safer and more effective way to achieve increased freedom from spectacles or contact lenses than corneal incisions.
By Vinod Gangwani, MD, FRCS, MRCOphth
Cataract patients with astigmatism present a unique challenge for surgeons. Astigmatism has been estimated to account for approximately 13% of all refractive errors. The prevalence of astigmatism varies widely according to ethnicity; East Asian people and Native Americans show a particularly high prevalence.1
Astigmatism has been estimated to affect up to 40% of cataract patients,2 many of whom express the desire to be spectacle-free after surgery. Consequently, surgeons increasingly recognize that correcting the spherical equivalent of refractive error alone is not enough; preexisting corneal astigmatism must also be addressed if patients are to achieve spectacle independence.
EARLY TORIC IOLS
Until relatively recently, existing corneal astigmatism was managed with spectacles or contact lenses, or patients could undergo corneal curvature–altering procedures such as peripheral corneal relaxing incisions (PCRIs), which can cause corneal damage and result in unsatisfactory refractive outcomes.3 Perhaps unsurprisingly then, since their conception in 1992,4 toric IOLs have become a popular solution for enhancing postoperative UCVA in cataract patients with astigmatism.
Nevertheless, early toric IOLs were not without issues. For example, the first toric lens, designed by Shimizu and colleagues in 1992, required an incision of 5.7 mm and was associated with poor rotational stability. Approximately 20% of these IOLs rotated 30° or more, and almost 50% rotated more than 10°.4 Axis misalignment of more than 10° reduces the efficacy of astigmatic correction by 33%, while axis misalignment of more than 30° actually causes astigmatism—the very condition a toric lens aims to manage.4 Two years later, Grabow described clinical outcomes after implantation of a foldable silicone toric IOL. Unfortunately, although 23% of patients achieved distance UCVA of 20/25 or better, as with the Shimizu lens, rotational stability was poor.5-8
CURRENT TORIC IOLS
Rotational stability issues associated with early toric IOLs were likely caused by a number of factors, including haptic design and the biomaterials used, the latter of which influences IOL adhesion to the capsular bag. Also, the lack of precise axis marking techniques led to a compound effect with both errors of misalignment and poor rotational stability. All of the aforementioned factors resulted in poor astigmatic correction.2
Fortunately, the toric IOL landscape has evolved since the early 1990s. Surgeons now have access to an array of lenses, including the T-flex aspheric toric (Rayner Intraocular Lenses), the Acrysof IQ Toric (Alcon), the Tecnis Toric (Abbott Medical Optics), and many more. Each lens differs in material type, design, diameter, and incision size required, but all are available in a wide range of spherical and cylindrical powers, permitting accurate and predictable visual outcomes in astigmatic patients. Multifocal toric IOLs and supplementary toric IOLs are also now available.
Today, toric IOLs have largely overcome the problems associated with earlier lenses. For instance, because the total diameter of the IOL and the design of the haptics are key factors in preventing IOL rotation, most torics have a diameter ranging from 11.0 mm to 13.0 mm with haptics specifically designed for rotational stability.9-11 Current toric lenses are also typically composed of highly biocompatible materials and may be implanted through incision sizes of 2.8 mm and less, facilitating rapid visual rehabilitation and helping to reduce surgically induced astigmatism.12
In addition to improvements in toric IOL design, we also have access to newer techniques to improve the accuracy of toric IOL alignment, including intraoperative wavefront aberrometry systems such as Ora (Alcon WaveTec) and Holos IntraOp (Clarity Medical Systems), as well as the iris fingerprinting technique developed by Osher and colleagues.2,13,14
AT A GLANCE
•Toric IOLs are a safe alternative to peripheral corneal relaxing incisions for the correction of corneal astigmatism during cataract surgery.
• Modern toric IOLs have overcome the problems of poor rotational stability associated with earlier models.
•Clinical evidence indicates that toric IOL implantation leads to more effective and predictable outcomes than the use of peripheral corneal relaxing incisions, resulting in greater spectacle independence.
TORIC IOLs VERSUS PCRIs
PCRIs have been associated with numerous complications including corneal damage and dry eye, and they may yield unpredictable outcomes. Nevertheless, they remain an important tool in the cataract surgeon’s armamentarium. Many surgeons prefer to treat cataract patients with low to moderate corneal astigmatism (< 2.00 D) using PCRIs, perhaps because the procedure is simple to perform and is minimally invasive.15
One of the challenges faced by cataract surgeons is a lack of guidance regarding when to treat astigmatism using PCRIs versus toric lenses. That is, at what degree of astigmatism should one or the other be used—or should toric IOLs always be offered over PCRIs, given the latter’s unpredictability. The Royal College of Ophthalmologists’ 2010 Cataract Surgery Guidelines state that “although incisional surgery [eg, PCRIs] may be used to correct corneal astigmatism, this may be less predictable in both effect size and stability than toric implants.”16
Although toric IOLs are regularly used in some centers to correct astigmatism of less than 1.00 D, a study by Poll et al suggests that toric IOLs really come into their own in patients with higher levels of astigmatism. In a retrospective review of 192 eyes, 77 eyes received a toric IOL, and 115 eyes received PCRIs. Although there was no statistically significant difference between the two treatment options in terms of the amount of surgically induced astigmatism or residual astigmatism, eyes with 2.26 D or more of cylinder were more likely to achieve 20/40 distance UCVA with toric IOLs than with PCRIs.17 These data suggest that patients with higher degrees of astigmatism would benefit from treatment with toric IOLs rather than PCRIs.
Other clinical studies also suggest that toric IOL implantation leads to superior outcomes compared with PCRIs in patients with moderate astigmatism. For example, a study undertaken by Hirnschall and colleagues that included 60 eyes of 30 patients with corneal astigmatism ranging from 1.00 to 2.50 D found that patients who were implanted with the T-flex toric lens demonstrated a statistically significant reduction (P = .042) in mean astigmatism vector. The reduction was 1.74 ±0.64 in the toric IOL group and 1.27 ±0.76 D in the PCRI-plus-monofocal-IOL group. Furthermore, the study highlighted the predictability of toric IOLs over PCRIs, as astigmatism in the PCRI group showed a statistically significant increase (P < .001) in the astigmatism vector from 1 month to 6 months postoperatively.17
A study by Gangwani and colleagues with the M-flex T multifocal toric IOL (Rayner Intraocular Lenses) once again demonstrated the effectiveness of toric technology. In this study, which included 58 eyes of 29 patients with corneal astigmatism ranging from 1.00 to 2.50 D, there was no rotation greater than 6° in any eye, and the mean rotation was 2.52 ±1.97° at the 3-month postoperative visit. Furthermore, once again, the toric correction of the lens established a statistically significant reduction (P = .042) in the mean astigmatism vector; the mean absolute vector difference (mean absolute error) was 1.76 ±0.47 D in the toric IOL group and 1.56 ±0.63 D in the PCRI group.18
A study by Mingo-Botín and colleagues, in which eyes with cataract and corneal astigmatism (1.00 to 3.00 D) had toric IOL implantation or PCRIs, showed that, although refractive astigmatism decreased in both groups, toric IOL implantation was more effective and predictable, resulting in greater spectacle independence.19
ECONOMIC FACTORS
Aside from the clinical aspects of toric IOLs and PCRIs, surgeons must also consider economic factors. This is especially true for those working in cash-strapped European health care systems. Because of the costs associated with toric IOL implantation, many centers are not able to offer this technology routinely in the UK National Health Service; patients who desire a toric IOL are required to pay for the lens and any peripheral costs through private health care options.
One way of reducing the cost of toric IOLs is to use toric lenses of predetermined cylinder (2.00 D), a concept currently being evaluated by Bruno Zuberbuhler of Guy’s and St. Thomas’ NHS Foundation Trust. This evaluation is aimed to determine whether correcting some off-the-shelf level of astigmatism is better for all astigmatic patients, rather than customizing the cylinder corrections offered by manufacturers for each patient.20
Looking at the issue another way, a study undertaken by Robert Pineda and colleagues suggests that, although many patients may be reluctant to pay for toric IOLs, these lenses actually help to reduce lifetime economic costs by reducing the need for glasses or contact lenses following cataract removal.21
The Importance of Posterior Corneal Curvature
Regardless of whether a surgeon chooses to reduce astigmatism using PCRIs or toric IOLs, estimating the degree of posterior corneal curvature is vital in order to avoid unwanted refractive surprise. Many surgeons still measure only anterior corneal astigmatism prior to performing PCRIs or toric IOL implantation. However, several studies have shown that astigmatism also affects the posterior cornea, increasing with higher anterior with-the-rule astigmatism and remaining mainly constant with higher amounts of against-the-rule astigmatism.1,2 Consequently, overlooking posterior corneal curvature may lead to incorrect measurement of total corneal astigmatism, in turn leading to overcorrection in eyes that have with-the-rule astigmatism and undercorrection in eyes that have against-the-rule astigmatism.1,2 Although there is still some debate as to the best method of assessing posterior corneal curvature, the Baylor nomogram, based on population averages, has been used with some success. Future methodologies may include the use of OCT and Scheimpflug imaging. These developments would help us in predicting the total corneal astigmatism and, in turn, providing better outcomes.
1. Koch DD, Jenkins RB, Weikert MP, Yeu E, Wang L. Correcting astigmatism with toric intraocular lenses: effect of posterior corneal astigmatism. J Cataract Refract Surg.2013;39(12):1803-1809.
2. Koch DD, Ali SF, Weikert MP, Shirayama M, Jenkins R, Wang L. Contribution of posterior corneal astigmatism to total corneal astigmatism. J Cataract Refract Surg.2012;38(12):2080-2087.
FUTURE DIRECTIONS
Perhaps one of the main reasons some surgeons are reluctant to offer toric IOLs to their patients, preferring to offer PCRIs instead, is perceived difficulties with lens alignment. However, new technologies such as intraoperative aberrometry, which permits real-time confirmation of sphere and cylinder, thus allowing greater precision in toric IOL alignment, should help to allay these fears. It is also worth remembering that, if necessary, toric IOLs offer the possibility to go back and rotate the lens into the correct axis. In contrast, once a PCRI has been made, it cannot be undone.
Overall, published evidence thus far indicates that, compared with PCRIs, toric IOLs offer excellent visual outcomes and spectacle independence in patients with astigmatism.2 Moreover, as more surgeons become familiar with toric IOL implantation, it is likely that outcomes will improve further. As lens technology continues to develop and we gather more clinical evidence regarding the safety, efficacy, stability and cost-effectiveness of toric IOLs, it seems inevitable that they will gain greater popularity among health systems and surgeons, allowing a greater number of patients to reap the benefits of toric lens technology.
1. BMJ Best Practice. Astigmatism. http://bestpractice.bmj.com/best-practice/monograph/762/basics/epidemiology.html. Accessed April 23, 2015.
2. Visser N, Bauer NJ, Nuijts RM. Toric intraocular lenses: historical overview, patient selection, IOL calculation, surgical techniques, clinical outcomes, and complications. J Cataract Refract Surg. 2013;39:624-637.
3. Pre-existing astigmatism correction combined with cataract surgery: corneal relaxing incisions or toric intraocular lenses? J Fr Ophtalmol. 2012;35(7):539-545.
4. Shimizu K, Misawa A, Suzuki Y. Toric intraocular lenses: correcting astigmatism while controlling axis shift. J Cataract Refract Surg. 1994;20:523-526.
5. Grabow HB. Early results with foldable toric IOL implantation. Eur J Implant Refract Surg. 1994;6:177-178.
6. Sun X-Y, Vicary D, Montgomery P, Griffiths M. Toric intraocular lenses for correcting astigmatism in 130 eyes. Ophthalmology. 2000;107:1776-1781; discussion by RM Kershner, 1781-1782.
7. Ruhswurm I, Scholz U, Zehetmayer M, Hanselmayer G, Vass C, Skorpik C. Astigmatism correction with a foldable toric intraocular lens in cataract patients. J Cataract Refract Surg. 2000;26:1022-1027.
8. Leyland M, Zinicola E, Bloom P, Lee N. Prospective evaluation of a plate haptic toric intraocular lens. Eye (Lond). 2001;15:202-205.
9. Patel CK, Ormonde S, Rosen PH, Bron AJ. Postoperative intraocular lens rotation: a randomized comparison of plate and loop haptic implants. Ophthalmology. 1999;106:2190-2195; discussion by DJ Apple, 2196.
10. Chang DF. Early rotational stability of the longer STAAR toric intraocular lens; fifty consecutive cases. J Cataract Refract Surg. 2003;29:935-940.
11. Shah GD, Praveen MR, Vasavada AR, Vasavada VA, Rampal G, Shastry LR. Rotational stability of a toric intraocular lens: influence of axial length and alignment in the capsular bag. J Cataract Refract Surg. 2012;38:54-59.
12. Musanovic Z, Jusufovic V, Halibasica M, Zvornicanin J. Corneal astigmatism after micro-incision cataract operation. Med Arh. 2012;66(2):125-128.
13. Osher RH. Iris fingerprinting: new method for improving accuracy in toric lens orientation. J Cataract Refract Surg. 2010;36:351-352.
14. Packer M. Effect of intraoperative aberrometry on the rate of postoperative enhancement: retrospective study. J Cataract Refract Surg. 2010;36:747-755.
15. Bronner A. Steep competition: LRIs vs. toric IOLs. Review of Ophthalmology. March 2012.
16. Royal College of Ophthalmologists: cataract surgery guidelines. September 2010. Available at https://www.rcophth.ac.uk/wp-content/uploads/2014/12/2010-SCI-069-Cataract-Surgery-Guidelines-2010-SEPTEMBER-2010.pdf, Accessed December 22, 2015.
17. Hirnschall N, Gangwani V, Koshy J, Maurino V, Findl O. Correction of moderate corneal astigmatism during cataract surgery: toric intraocular lenses versus peripheral corneal relaxing incisions. J Cataract Refract Surg. 2014;40(3):354-361.
18. Gangwani V, Hirnschall N, Findl O, Maurino V. Multifocal toric intraocular lenses versus multifocal intraocular lenses combined with peripheral corneal relaxing incisions to correct moderate astigmatism. J Cataract Refract Surg. 2014;40:1625-1632.
19. Mingo-Botín D, Muñoz-Negrete FJ, Won Kim HR, Morcillo-Laiz R, Rebolleda G, Oblanca N. Comparison of toric intraocular lenses and peripheral corneal relaxing incisions to treat astigmatism during cataract surgery. J Cataract Refract Surg. 2010;36(10):1700-1708.
20. UK Clinical Research Network. Off-the-shelf use of toric intraocular lenses for NHS cataract patients. Available at: http://public.ukcrn.org.uk/search/StudyDetail.aspx?StudyID=11518. Accessed December 16, 2015.
21. Pineda R, Denevich S, Lee WC, Waycaster C, Pashos CL. Economic evaluation of toric intraocular lens: a short- and long-term decision analytic model. Arch Ophthalmol. 2010;128(7):834-840.
Vinod Gangwani, MD, FRCS, MRCOphth
• Consultant Ophthalmic Surgeon, Ashford and St. Peter’s Hospitals NHS Trust, United Kingdom
• dr_vinod_g@hotmail.com
• Financial disclosure: None
Cristina Giordano, OD
• Optometrist and laser key operator, Centro Diagnostico Italiano, Milan, Italy
• applysrl@virgilio.it
• Financial disclosure: None
Edoardo Ligabue, MD
• Chief, Cataract and Refractive Department, Ophthalmic Center at the Centro Diagnostico Italiano, Milan, Italy
• edoliga@tiscali.it
• Financial disclosure: None)
