Getting to Know the Patient
Several years after the development of LASIK-induced epithelial ingrowth in her right eye, a 65-year-old woman presented for cataract surgery with bilateral nuclear opalescent cataracts (right eye, grade 3.8 LOCS III; left eye, grade 3.6 LOCS III). Her BCVA had deteriorated to 20/40-1 OD over the past 8 months, and BCVA was also reduced at 20/30-2 OS. She was hoping for improved vision in both eyes. Ideally, she said, she would like to avoid spectacle correction, but she also communicated that, “after all that had happened to my right eye, I would understand if glasses were a necessity.” More than anything, the patient said she would like to avoid additional surgical intervention that would further disturb the LASIK flap of the right eye.
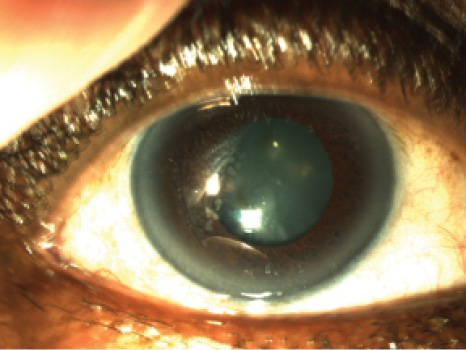
Figure 1. Slit-lamp photograph of the patient’s right eye, which has a previous microkeratome nasal-hinged flap with temporal epithelial ingrowth. The ingrowth persisted despite flap lifts with mitomycin-C applications and sutures at the margins, performed at two encounters 6 months apart. An Nd:YAG laser treatment of the central epithelial islands was also performed.
In 2008, microkeratome-assisted LASIK was performed in the patient’s right eye only, with the goal of monovision. Unfortunately, this was subsequently complicated by epithelial ingrowth under the flap (Figure 1). Six months after the procedure, the flap was lifted, scraped, and treated with antimetabolite therapy. The treatment was repeated 4 months later, to no avail. As recently as 2013, Nd:YAG laser was employed to prevent the centrally progressing island of epithelial ingrowth, a technique described by Ayala et al.1
1. Ayala M J, Alio JL, Mulet ME, DeLaHoz F. Treatment of laser in situ keratomileusis interface epithelial ingrowth with neodymium:yytrium-aluminum-garnet laser. Am J Ophthalmol. 2008;145(4):630-634.

Performing cataract surgery in an eye with previous corneal refractive surgery is inherently challenging under the best of conditions. Two recent developments have helped to improve the predictability of refractive outcomes of IOL power calculations for these eyes: the availability of the web page www.iolcalc.org, supported by the ASCRS Foundation and Alcon,1 and the introduction of intraoperative aberrometry.2
Corneal ectasia and other forms of corneal irregularity are additional sources of reduced refractive predictability in patients requiring cataract surgery.3,4 Previously, authors have reported successes with IOL power calculations based on axial power mapping, total optical power mapping, and subsequent contact lens over-refraction for patients with irregular astigmatism due to corneal problems.5
In the case presented above in Getting to Know the Patient, I was confronted with the unique situation of a post-LASIK patient further complicated by recalcitrant epithelial ingrowth. For this case, outside-the-box thinking was required.
PLANNING AND COUNSELING
Preoperative corneal analysis, aberrometry, and biometry were performed (Figure 2). Our initial suggestion was to attempt a flap lift, scraping, antimetabolite application, and sutures with ReSure Sealant (Ocular Therapeutix) before treatment of the cataract. The patient was not interested in trying this approach.
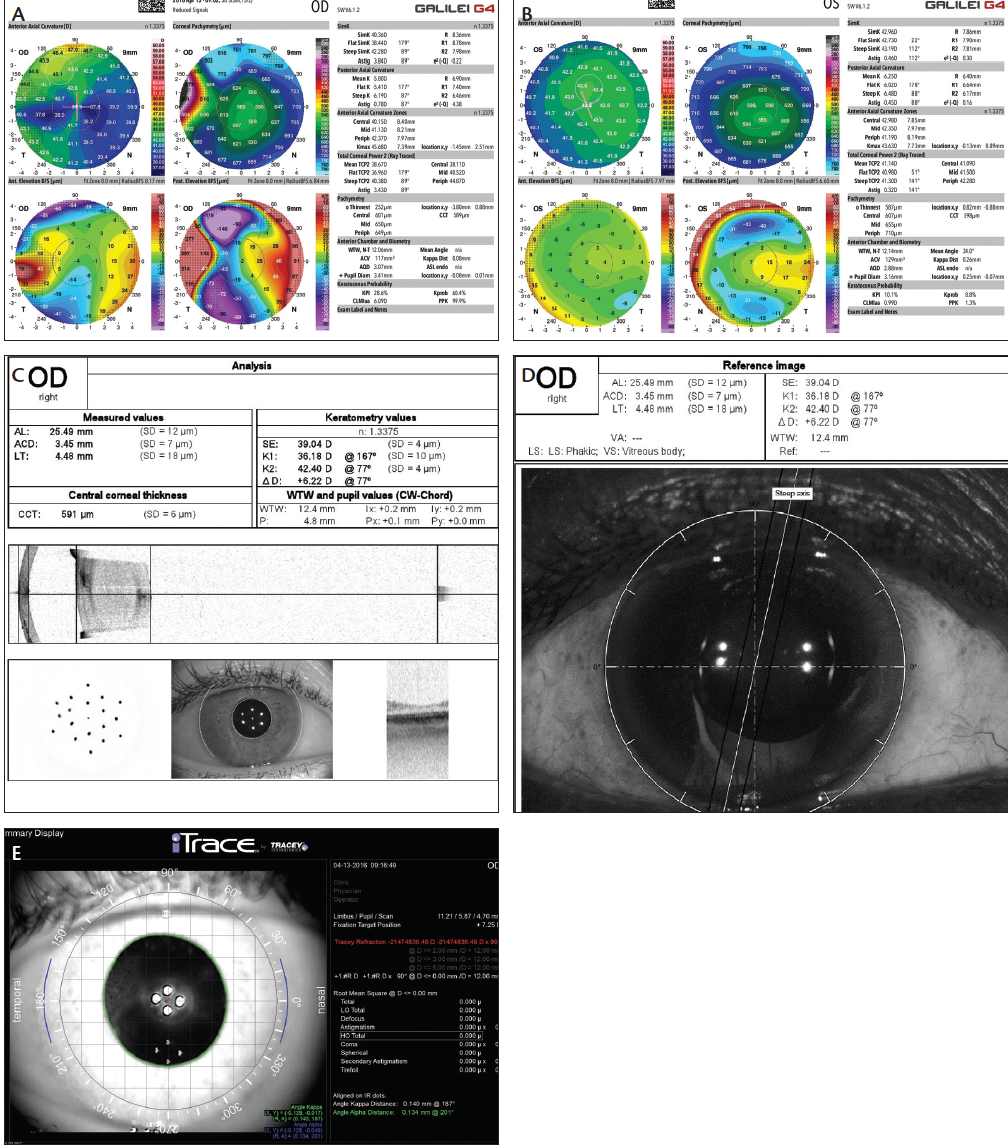
Figure 2. Topographies of the patient’s right eye, demonstrating high irregular with-the-rule cylinder and temporal elevation with displacement of the angle kappa distance (A), and left eye demonstrating normal symmetric curvature, elevation, and line of sight (B). Swept source OCT biometry raw data and imaging, demonstrating the temporal smearing of the keratometry projections and the interface reflectivity within the LASIK bed in the patient’s right eye (C). Wavefront guidance indicating the axis for toric IOL alignment in the patient’s right eye (D). Imaging of the right eye, illustrating temporal reticulopathy near the corneal vertex consistent with the iron line and clinical appearance of the epithelial ingrowth (E).
In further reviewing the patient’s old records, it became evident that, in the intervening years between her last corneal treatment and the cataract development, her UCVA was 20/20-2 OD. On further examination, her spectacle correction was +0.75 -0.75 X 155° OD despite the fact that analyses on several devices demonstrated anywhere from 3.43 to 8.31 D astigmatism, either at the cornea or via wavefront refraction. In observing the patient viewing distance targets, there appeared to be a nasal displacement of the line of sight, akin to what can be seen in corneal ectasia when there is multifocality of the corneal surfaces.6
After a thorough review and discussion of the risks and benefits, we agreed to proceed with cataract removal with wavefront-guided surgery and a goal of emmetropia using a monofocal IOL. We discussed at length the complexity of IOL power calculations due to the previous LASIK procedure and the possibility of a refractive surprise. We also discussed the potential need for enhancement due to the unique aspects of the opacity generated by the epithelial ingrowth. The patient agreed to a contingency plan to proceed with manipulation of the flap bed should her visual outcomes not meet her needs for activities of daily living or if we began to notice reactivation of the islands of epithelial cells in the interface.
Our surgical planning took into consideration the preoperative refractive cylinder, and IOL calculations were performed based on an average keratometry (K) that was centered over the line of sight, much the same way that topography can be used to find the power in a keratoconic eye prior to cataract surgery. The iolcalc.org website, using no historical data, suggested an IOL power range of 22.00 to 23.00 D.
INTRAOPERATIVE ABERROMETRY, POSTOPERATIVE RESULTS
Surgery was uneventful, with the exception of a notable angle kappa when intraoperative aberrometry was performed. The patient was instructed to look at the fixation target to align the vertex of the cornea for initial aphakic measurements, due to the significant negative angle kappa, in order to confirm the line of sight. Then she was asked to view the fixation target where it was most clear (Figure 3A). The aphakic measurement was in agreement with the preoperative corneal topography (Figure 3B). A toric IOL was selected based on the refraction, as opposed to the aphakic refraction from the vertex, and alignment (Figure 3C and 3D) was performed based on the line of sight (Figure 3E).
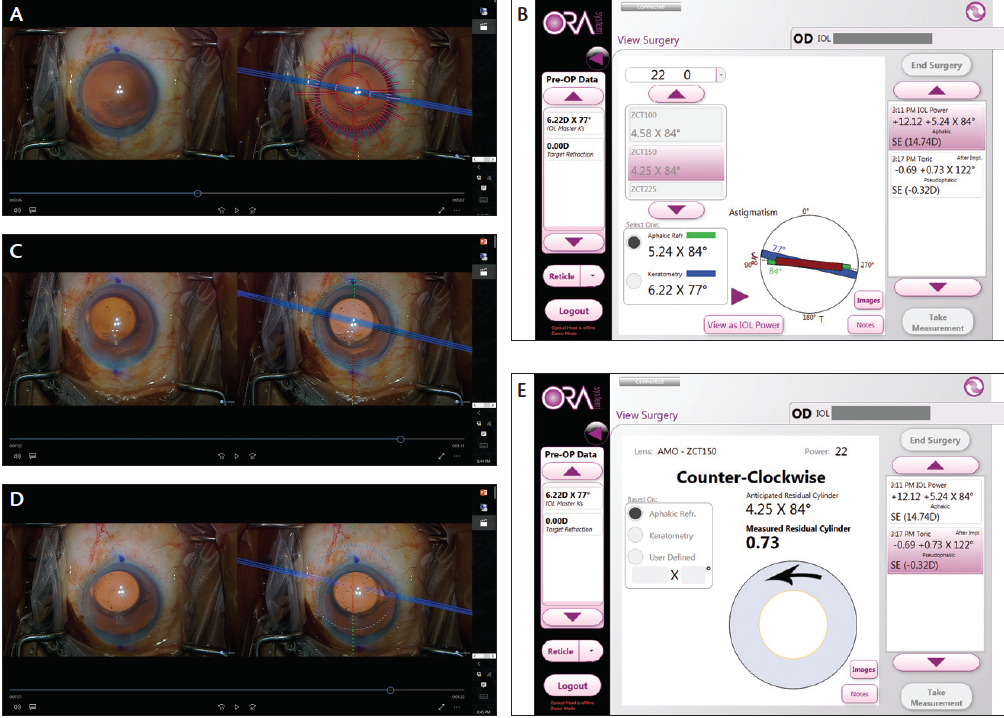
Figure 3. Intraoperative 3-D heads-up image showing Callisto eye (Carl Zeiss Meditec) and ORA System with VerifEye (Alcon) reticules outlining the steep axis alignment (A). ORA measurement and suggested placement of toric IOL (B). Intraoperative guidance for a ZCT150 Tecnis Toric IOL (Abbott Medical Optics) along the steep axis across the vertex (C). Intraoperative guidance for the same IOL based on line of sight (D). ORA measurement of final pseudophakic refraction, line of sight (E).
Postoperatively, the patient returned with a significant improvement in vision and was happy with her level of functional improvement, particularly with the increase in brightness. I was worried, however, about the possibility of the signal-to-noise ratio being imbalanced due to a perception of dysphotopsia. This apparently was not the case.
CASE RECAP
WHO
65-year-old woman with bilateral nuclear opalescent cataracts and a history of LASIK surgery in her right eye, complicated by recalcitrant epithelial ingrowth
What
Ideally, achievement of spectacle independence, but the patient’s main concern was the avoidance of additional surgery that would further disturb the LASIK flap
How
After declining flap lift, scraping, and application of both antimetabolite and ocular sealant, the patient agreed to cataract removal with wavefrontguided surgery and implantation of a toric monofocal IOL; she was happy with her postoperative refraction of -0.25 -0.50 X 175º yielding a 20/25+2 UCVA
Her refraction was -0.25 -0.50 X 175°, yielding BCVA of 20/25+2 and consistent with the intraoperative measurement. She is comfortable with her acuity and feels vindicated in her decision not to manipulate the LASIK flap. I am left scratching my head, but pleased with the results nonetheless. A number of questions linger, along with her corneal pathology. Yet we never opened Pandora’s Box, and hopefully we will not see reactivation of the epithelial cells as she continues to heal from her cataract procedure.
TAKEAWAYS
In retrospect, all the preoperative calculations in the world would not have prepared me for the patient’s postoperative response, but the various devices working together in the office and in the operating room did provide confidence that there was synergy and seamless communication in the pursuit of another complex, complicated lens-based refractive procedure.
With the surgery behind us, her attention is now directed toward correcting her left eye, and she has her sights set on a multifocal IOL. We are currently in discussions.
1. IOL power calculation in eyes that have undergone LASIK/PRK/RK. http://iolcalc.org/. Accessed May 18, 2016.
2. Hemmati HD, Gologorsky D, Pineda R. Intraoperative wavefront aberrometry in cataract surgery. Semin Ophthalmol. 2012;27(5-6):100-106.
3. Langenbucher A, Haigis W, Seitz B. Difficult lens power calculations. Curr Opin Ophthalmol. 2004;15(1):1-19.
4. Tamaoki A, Kojimoa T, Hasegawa A, Nakamura H, Tanaka K, Ichikawa K. Intraocular lens power calculation in cases with posterior keratoconus. J Cataract Refract Surg. 2015;41(10):2190-2195.
5. Cua IY, Qazi MA, Lee SF, Pepose JS. Intraocular lens calculations in patients with corneal scarring and irregular astigmatism. J Cataract Refract Surg. 2003;29(7):1352-1357.
6. Mihaltz K, Kranitz K, Kovacs I, Takacs A, Nemeth J, Nagy ZZ. Shifting of the line of sight in keratoconus measured by a Hartmann-Shack sensor. Ophthalmology. 2010;117:41-48.
Jonathan D. Solomon, MD
• Surgical/Refractive Director of Solomon Eye Associates, Bowie, Maryland
• jdsolomon@hotmail.com
• Financial interest: None acknowledged

