CASE PRESENTATION
A 38-year-old man with a history of high myopia presented 3 days after a sudden reduction in vision in his left eye. He was seen by his primary eye care doctor, who referred the patient for surgical evaluation.
At the time of referral, the visual acuity in his right eye was 20/20 with a contact lens and count fingers in his left eye, improving to 20/200 with pinhole. The reactivity of the pupils was normal, but the left pupil was slightly miotic.
A slit-lamp examination of the patient’s right eye showed several nylon sutures under the conjunctiva. The cornea was clear. The iris had two peripheral iridotomies, and the eye was aphakic. The patient’s left eye had a normal conjunctiva and cornea. The iris had two large peripheral iridotomies, and the crystalline lens was severely dislocated into the anterior vitreous. No vitreous was visible in the anterior chamber, and the surgeon noted no abnormalities on dilated retinal examination (Figures 1–3).
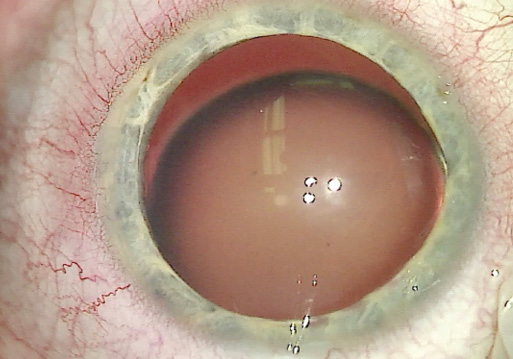
Figure 1. A clinical examination of the patient’s left eye showed inferior subluxation of the lens.

Figure 2. Biometry measurements of the patient’s left eye.
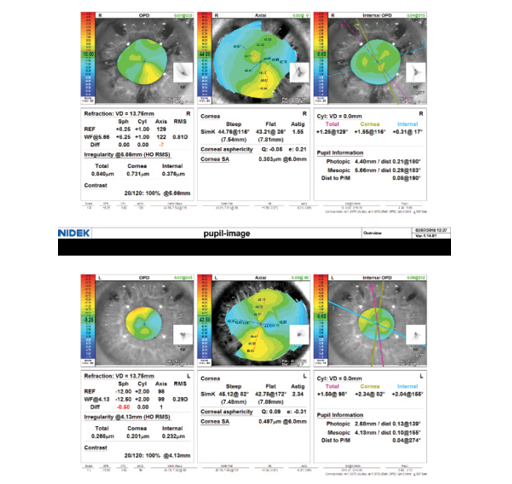
Figure 3. Topography of the patient’s left eye.
The patient’s past ocular history was significant for the placement of Visian ICLs (STAAR Surgical) several years earlier for the treatment of high myopia. Shortly after implantation, the right ICL dislocated, requiring removal, at which time the cataract was also extracted and a posterior chamber IOL (PCIOL) was placed. Several months after the cataract surgery, the PCIOL dislocated, requiring removal and a pars plana vitrectomy, leaving the right eye aphakic. More recently, the left ICL was removed secondary to dislocation.
Understandably, the patient is deeply concerned about his rapid change in vision. How would you manage his left eye?
—Case prepared by Brandon D. Ayres, MD

KJELL U. SANDVIG, MD, PhD
Most likely, this patient had subluxated/dislocated crystalline lenses preoperatively, and Marfan syndrome and homocystinuria should be ruled out. He therefore should never have had ICLs implanted or a PCIOL placed without extra support. Possible causes of his reduced visual acuity other than severe lens dislocation should at least be excluded by OCT imaging of the macula and the optic nerve head. I would recommend removing the severely dislocated lens in the patient’s left eye in the most controlled manner: phacovitrectomy via the pars plana. Because I am an anterior segment surgeon, I would refer the patient to a vitreoretinal specialist.
The next questions are should an IOL be implanted, and, if so, which one? The patient’s right eye is currently aphakic, but vision is successfully corrected with a contact lens. Bilateral aphakia is impractical, however, because it makes putting contact lenses in difficult. I would therefore recommend implantation of an IOL in his left eye. There are three options with relatively long-term follow-up: an anterior chamber IOL (ACIOL), an iris claw lens, or a sutured PCIOL. The patient’s relatively young age contraindicates an ACIOL because of the unacceptably high risk of endothelial cell loss and glaucoma. His age is also an argument against a sutured PCIOL because I would expect the sutures to degrade in a few decades. The best option would therefore be an iris-fixated—preferably retropupillary—IOL targeted for slight myopia (mini monovision). If necessary, the patient’s residual astigmatism could later be corrected with anterior surface excimer ablation.

SARAH BROWN WEISSBART, MD
Preoperatively, I would perform refraction, a dilated fundus examination, and testing with a potential acuity meter to determine the patient’s visual potential and risk of posterior segment complications. Because of his history of multiple intraocular surgeries, I would also obtain an endothelial cell count to determine the risk of corneal decompensation. Additionally, given the asymmetric astigmatism in the patient’s right eye, I would perform corneal tomography to rule out ectasia. I would explain to the patient the complex nature of the surgical intervention as well as the potential for complications and additional surgery in the future.
Figure 1 shows inferior subluxation of the crystalline lens, which is likely secondary to zonular loss. I would be prepared to use capsular hooks, sutured capsular tension segments, and a capsular tension ring to stabilize the bag. I would inject trypan blue dye before making the capsulorhexis or consider using a femtosecond laser to perform the capsulotomy. I would also have anterior vitrectomy equipment available because of the potential for vitreous prolapse. If I were able to stabilize the capsule adequately, I would implant a one-piece toric IOL (depending on the tomography) or a monofocal IOL. If a capsular tear occurred, I would opt for a scleral-fixated IOL. I would avoid an iris-sutured IOL because of the two peripheral iridotomies, a multifocal IOL because of the possibility of future dislocation and additional ocular pathology, and an ACIOL because of the potentially unhealthy corneal endothelium.
Finally, the combination of high myopia, bilateral ectopia lentis, and asymmetric corneal astigmatism raises my concern about connective tissue disease. I would therefore refer the patient to his medical doctor for further evaluation.

WHAT I DID: BRANDON D. AYRES, MD
I had a long and comprehensive discussion of the risks and benefits of surgical intervention with the patient. I explained the increased complexity of his case and the option of genetic testing. I also sent him for surgical retinal evaluation because there was a high likelihood of his needing a pars plana vitrectomy. After carefully considering his options, the patient decided in favor of proceeding with cataract surgery and against genetic testing.
The first challenge was creating the continuous curvilinear capsulorhexis (CCC). The extreme zonular instability prevented a cystotome or forceps from penetrating the anterior capsule. A 22.5º sharp blade was required to puncture the capsule. I completed the CCC via a bimanual technique in which I used one pair of forceps to stabilize the lens and another for the tear. Next, I placed capsular retracting hooks to keep the lens in position and allow its removal (Figure 4).
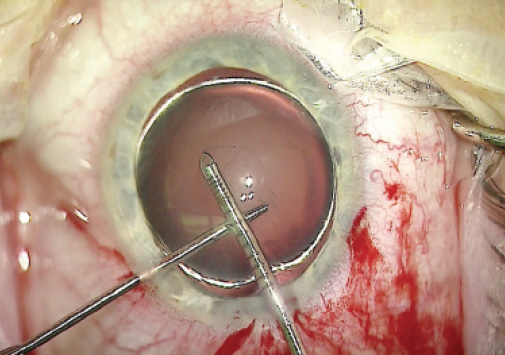
Figure 4. Use of atraumatic forceps and microrhexis forceps allowed completion of the CCC.
With a 25-gauge vitrector, I removed the lens, capsular bag, and anterior vitreous. The instability of the capsular bag was so severe that I did not attempt to use a capsular tension ring or sutured capsular tension segments (Figure 5).
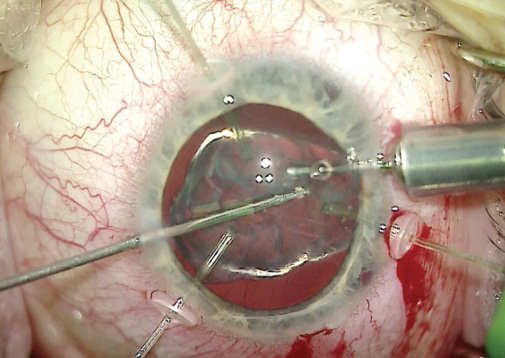
Figure 5. Capsular hooks held the lens in place, while a 25-gauge vitrector acted as an I/A unit to remove lenticular material and any prolapsed vitreous.
After completing the vitrectomy, I placed an 18.00 D CT Lucia IOL (model 602, Carl Zeiss Meditec) in the anterior chamber. I made marks 3 mm posterior to the limbus at the 6 and 12 clock positions. With thin-walled 30-gauge needles, I made scleral incisions at the 6 and 12 clock positions, and then I incarcerated each lens haptic into the lumen of a needle (Figure 6). Thereafter, both haptics were externalized on the conjunctival surface. I melted the terminal ends of the needles using battery-operated low-temperature cautery, as described by Yamane (Figure 7).
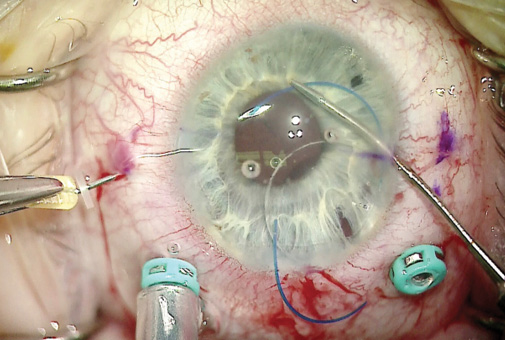
Figure 6. The surgeon placed a three-piece IOL in the anterior chamber and incarcerated the haptics in two 30-gauge thin-walled needles.
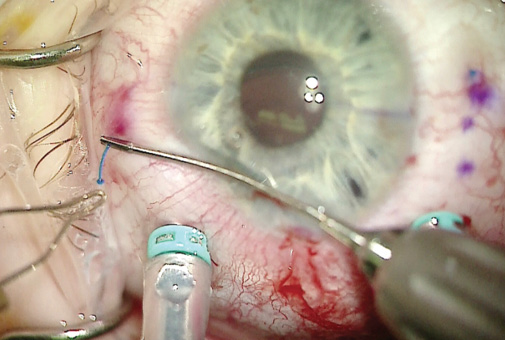
Figure 7. The terminal few millimeters of the three-piece IOL haptic were melted. The bulb prevented the haptic from prolapsing through the scleral channel.
At the conclusion of the case, I removed all OVD from the anterior chamber with the vitrector. I verified watertight closure of the corneal incisions and sutured the main wound with 10-0 nylon.
The patient tolerated the surgical procedure well. On postoperative day 1, his UCVA measured 20/20, and his IOP was 20 mm Hg. The postoperative course was uncomplicated, and he retained excellent distance UCVA.


