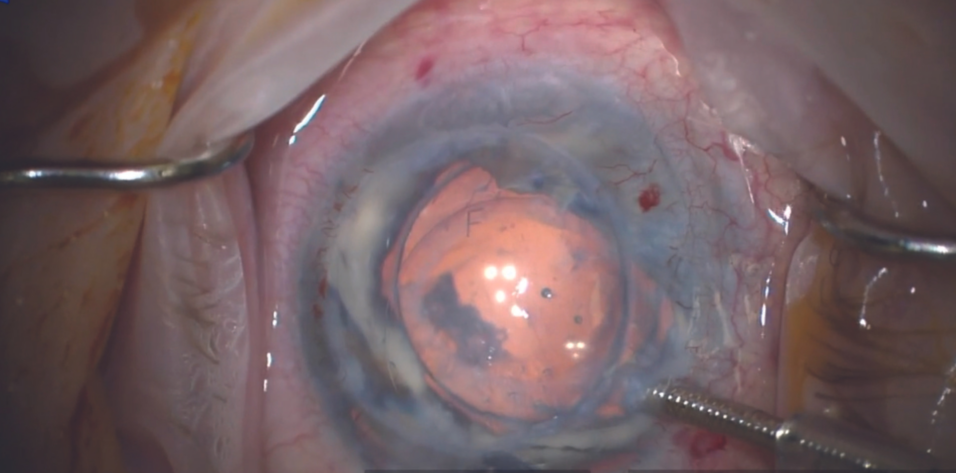
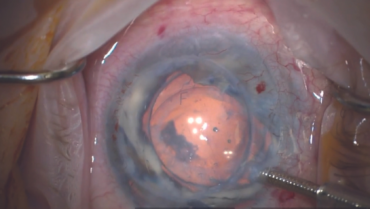
Corneal endothelial dysfunction is a primary cause of corneal edema that can lead to clouding and visual impairment for many patients.1,2 In advanced stages of the disease, a corneal endothelial transplantation procedure (ie, Descemet-stripping automated endothelial keratoplasty [DSAEK] or Descemet membrane endothelial keratoplasty [DMEK]) may be necessary to replace the diseased endothelium with healthy donor tissue. The complexity and invasiveness of DSAEK and DMEK procedures coupled with a global shortage of donor tissue,1 however, complicate the course of management. Researchers have been working on alternative treatments, and early results with an artificial endothelial layer technology are promising.3
WHAT IS ENDOART?
EndoArt (EyeYon Medical) is a thin (50 µm) and flexible artificial endothelial layer designed to match the posterior corneal curvature and act as a fluid barrier at the posterior stroma. The mechanism of action is shown in Figure 1. The optically clear, biocompatible device is implanted in the anterior chamber under a penetrating keratoplasty (PKP), positioned on the posterior stroma, and secured with an air-gas mixture. According to the company, EndoArt can lead to clear improvements in visual function and corneal thickness in eyes with even advanced corneal edema, such as those with multiple graft failures (Figure 2).
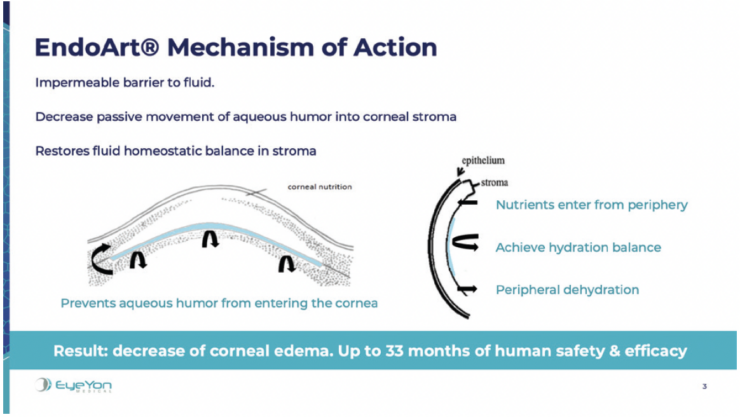
Figure 1. The mechanism of action of EndoArt, a fully synthetic artificial endothelial layer.
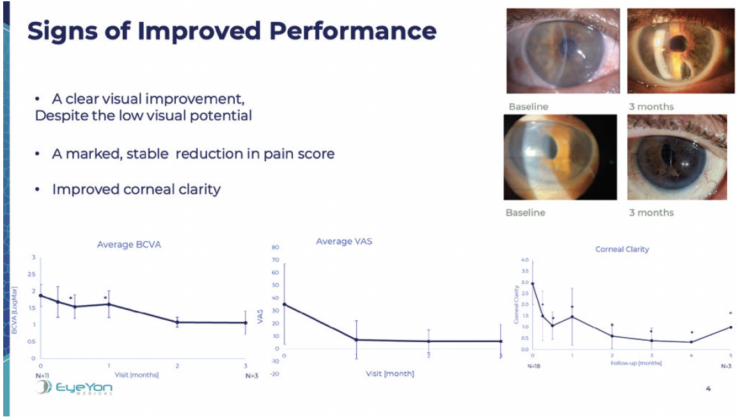
Figure 2. Signs of improved visual performance in eyes that received the EndoArt.
At the time I presented early results during the AECOS Europe meeting in Antwerp, Belgium,4 80 patients had been treated with the technology. I was the first in Belgium to perform the procedure. My results are described here.
CASE PRESENTATION
An 80-year-old woman presented with a history of ankylosing spondylitis and multiple episodes of uveitis. She underwent cataract surgery in 1993 and developed complications of bullous keratopathy in 2004. A PKP was performed in 2005, and graft rejection occurred in 2013. Despite prolonged corneal edema, a DSAEK was performed in 2017. Again, the graft was rejected. Rather than subject the patient to further risk and potential complications of a repeat endothelial keratoplasty procedure, I decided to implant an EndoArt under a PKP.
Some posterior capsular opacification was present preoperatively, and the corneal edema had progressed. Her visual acuity was worse than 20/200. Preoperative imaging is shown in Figure 3.
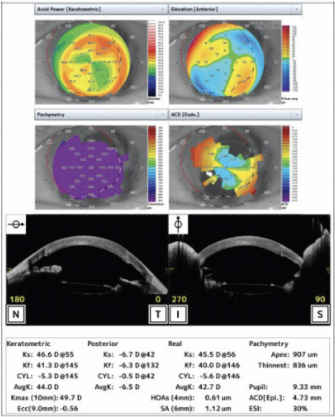
Figure 3. Preoperative imaging.
A video of the procedure may be viewed below. The case took me 16 minutes to perform, but I expect procedural time to decrease after a learning curve. Orienting the EndoArt precisely on the posterior stroma can take practice. An F stamp on the device is used to align it at the 12 clock position (Figure 4). The EndoArt is then tapped into place. The rest of the procedure is straightforward—much easier than DSAEK or DMEK. A little bubble of an air-gas mixture (octafluoropropane) is used to create an airtight fill, securing the device on the posterior cornea. Then, the main incision is sutured.
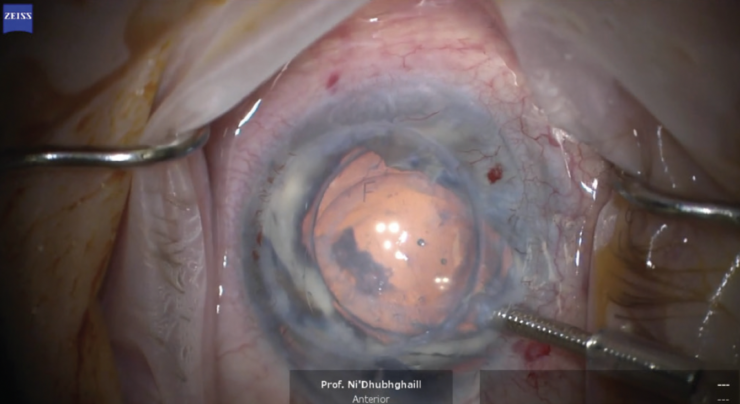
Figure 4. An F stamp on the EndoArt can be used to align the device at the 12 clock position.
Meet the AECOS Update Section Editors
CONCLUSION
The EndoArt procedure is evolving. Early results with the device are promising. A proven, reproducible implantation technique should help further refine the procedure. I expect the EndoArt to be particularly useful for eyes that have experienced a previous endothelial graft rejection.
1. Van den Bogerd B, Zakaria N, Adam B, Matthyssen S, Koppen C, Ni Dhubhghaill S. Corneal endothelial cells over the past decade: Are we missing the mark(er)? Trans Vis Sci Tech. 2019;8(13).
2. Bourne WM. Clinical estimation of corneal endothelial pump function. Trans Am Ophthalmol Soc. 1998;96:229-239; discussion 239-242.
3. Auffarth GU, Son HS, Koch M, et al. Implantation of an artificial endothelial layer for treatment of chronic corneal edema. Cornea. 2021;40(12):1633-1638.
4. Ni Dhubbghaill S. Premiere: EndoArt under PK. Paper presented at: 2022 AECOS Europe; June 10–12, 2022; Antwerp, Belgium.