CASE PRESENTATION
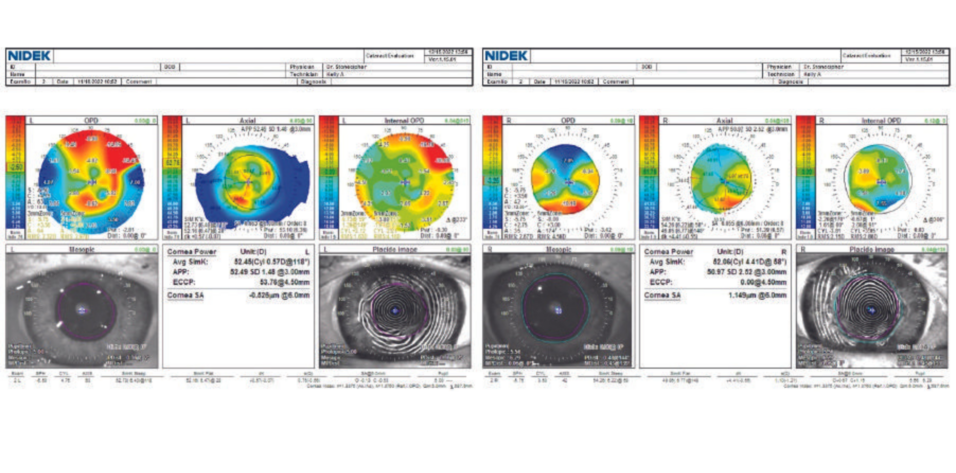
An 81-year-old woman presents for a refractive surgery evaluation. Her history is significant for bilateral hexagonal keratotomy more than 45 years ago. This early refractive surgery technique was used to correct hyperopia. Six linear incisions were made in the stroma of the peripheral cornea in a hexagonal pattern that encircled the central cornea. On examination, the patient’s BCVA is -4.25 +4.00 x 022º = 20/50 OD and -4.00 +4.25 x 069º = 20/70 OS. Each eye has moderate dry eye disease with lissamine green staining +2/+2/+2 and a tear breakup time of less than 4 seconds. Both eyes have 2+ nuclear sclerosis and 1+ to 2+ cortical changes. Potential acuity meter testing demonstrates 20/40 VA OU. The retinas are normal. Pachymetry readings are 565 µm OD and 560 µm OS. Findings with the OPD-Scan (Nidek) are shown in Figures 1 and 2.
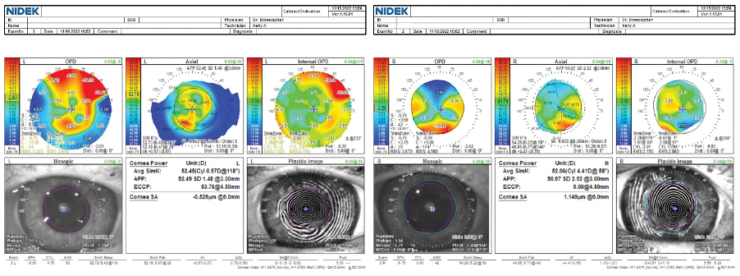
Figure 1. Topographic irregular astigmatism related to prior incisional surgery.
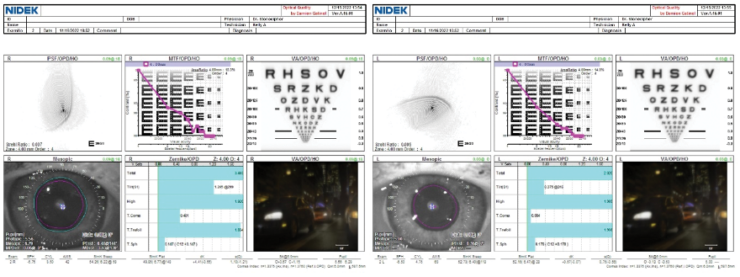
Figure 2. Gatinel Optical Quality Analysis (Visiometrics) indicates that both eyes have poor corneal quality.
The patient desires improved vision in both eyes. What surgery would you recommend, and would you stage it? If you recommend refractive surgery, how would you proceed? If cataract surgery, which IOL would you choose, and how would you perform biometry and calculate the IOL power? What preoperative steps would you pursue?
—Case prepared by Karl G. Stonecipher, MD

JACQUELINE BELTZ, BMEDSCI, MBBS, MSURGED, FRANZCO
Given the patient’s age, cataract surgery would give her the best chance of improved vision. During the preoperative discussion, I would emphasize that the aim is to improve her visual acuity, most likely with spectacles. I would operate on the left eye first followed a month or so later by the right eye. Based on the degree of corneal irregularity, I would consider an IC-8 Apthera IOL (Bausch + Lomb) if all the central media are clear and the mesopic pupil size is less than 5.5 mm. If necessary, the tear film would be optimized before biometry. Primary and secondary keratometry readings would be obtained with the IOLMaster (Carl Zeiss Meditec) and corneal tomography, respectively. I would also consider calculating an integrated keratometry reading with the Barrett True K toric calculator.
I would use the Barrett True K IOL formula for the IOL power calculation. Post–radial keratotomy or hyperopic LASIK modifications would not be appropriate.
To the best of my knowledge, hexagonal keratotomy was rarely performed in Australia. I suspect that the patient’s corneas are highly unstable, and she is likely to develop greater hyperopia and corneal irregularity over time. A myopic target (approximately 1.00 D) would therefore be used. I favor the off-label use of an Apthera versus a toric IOL in eyes with significant irregular astigmatism. I do not normally recommend implanting a small-aperture IOL bilaterally but sometimes make an exception for patients with bilateral corneal irregularity.
I would give the patient plenty of time and counseling between surgeries. I would want to ensure that she is happy with the outcome in her first eye before planning surgery on her second eye.

JOHN A. HOVANESIAN, MD
Patients who have a history of hexagonal keratotomy present a unique challenge because their corneal curvature can vary widely and irregular astigmatism is the rule rather than the exception.
The first step here is to determine the approximate corneal power and estimate the visual potential allowed by the cornea. Corneal topography can be helpful for the former. Axial maps with a calculation of average powers in the central 1 mm provide a fairly accurate estimate. Rigid contact lens overrefractions can also be helpful, but their accuracy depends on the density of the cataract. To determine corneal visual potential, the patient’s old records may be requested or she may simply be asked how satisfied she was with her BSCVA before she developed cataracts. Other metrics, such as total corneal higher-order aberrations in the central 6 mm of the cornea, can give a quantitative estimate of the extent of irregularity.
The Apthera IOL is the most suitable choice in the current situation. Another good option would be a Light Adjustable Lens (RxSight) if the patient’s corneal visual potential justifies its use. Both lenses require a pupil that dilates to at least 6 mm.

H. BURKHARD DICK, MD, PHD, FEBOS-CR
Cataract and refractive surgeons are likely to encounter an increasing number of this sort of patient soon—individuals who underwent the first generation of refractive surgery procedures. (Editor's note: CRST's April issue is devoted to this topic.)
The current patient is most likely aware that the visual prognosis for both eyes is limited due to the cornea. The cornea exhibits significant irregularity with differences in the center of up to 4.00 D. The Pentacam (Oculus Optikgeräte) measures 1.60 D of astigmatism that is almost completely within the photopic pupillary opening and two hot spots, the smaller of which is located toward the temporal side and the larger more nasally. Topography warns me away from implanting a toric IOL. The astigmatism does not extend beyond the pupil, the meridians are not orthogonal to each other, and the entire line-of-sight complex seems slightly dislocated. Further attention must be paid to the 0.23 chord mu on the Pentacam’s Holladay Report, which indicates that the patient is not looking through the geometric center of the cornea.
Moreover, with preexisting corneal incisions, the chance of ripping open the cornea intraoperatively cannot be disregarded. This could complicate surgery and necessitate the placement of sutures, which could induce astigmatism.
Assuming the formerly highly hyperopic patient is interested primarily in crisp distance visual acuity, the implantation of a multifocal or extended depth of focus lens is not advisable. This leads me to what I consider the most appropriate choice for the patient, which is an Apthera IOL. Its optical principle—the pinhole effect—can smooth out up to 1.50 D of astigmatism. The lens can provide her with a stable distance visual acuity irrespective of any residual astigmatism.
Preoperative measurements of angles alpha and kappa would be performed. The IOL calculation should not be based on measurements obtained with the IOLMaster 500 because the device extrapolates corneal radii and anterior chamber depth based on image analysis. Instead, the eye would be measured with the IOLMaster 700 (which comes with OCT) and Pentacam HD–based ray tracing. Using the physiologic pupil size as a setting is crucial. If the rule is not observed, ray tracing will lead the surgeon astray because it will measure the entire corneal area.
When choosing the refractive power of the Apthera IOL, I would target a mildly myopic outcome and thus shift the defocus curve slightly toward myopia. This should improve the patient's intermediate and near vision without compromising her distance vision.
When counseling the patient preoperatively, the mantra of underpromise and overdeliver should be observed.

WHAT I DID: KARL G. STONECIPHER, MD
After aggressive treatment of dry eye disease with punctal occlusion, low-level light therapy with intense pulsed light therapy (Espansione Group), topical cyclosporine, and Evotears (URSA Pharm), final corneal topography and tomography plus biometry with the IOLMaster 700 was performed. The ASCRS refractive IOL calculator, the ESCRS refractive IOL calculator, and the Barrett True K toric calculator on the IOLMaster 700 were used preoperatively to calculate the lens power. (For more on the ESCRS IOL calculator, see “Next-Generation Online IOL Resources.") Cataract surgery was performed with the LenSx femtosecond laser system and Centurion Vision System (both from Alcon), and the large sleeve was used on the latter to account for the 3-mm incision. Near-clear corneal incisions were created with 1- and 3-mm blades to account for the larger incision required for an IC-8 Apthera IOL. (Watch the case below.)
After surgery on the patient’s first eye, an Apthera IOL was also implanted in her second eye at her request. Two weeks after surgery on the second eye, her UCVA and BCVA was 20/40 OU and 20/30 OU, respectively. Postoperative care was routine. Aggressive treatment of her dry eye disease continues.