

Viral anterior uveitis accounts for 4% to 11% of all anterior uveitis cases.1 Herpes simplex virus (HSV) types 1 and 2 can cause anterior uveitis, but HSV1 is more prevalent. Herpes zoster ophthalmicus caused by varicella zoster virus (VZV), which may be the initial manifestation of an immunocompromised state, presents with dermatomal vesicular eruption along the first division of the fifth cranial nerve, preceding the anterior uveitis. VZV, however, can also cause anterior uveitis in the absence of a rash.2
AT A GLANCE
- A viral etiology should be suspected when a patient presents with unilateral, acute, recurrent or chronic hypertensive anterior uveitis with characteristic patterns of keratic precipitates and iris changes.
- Herpetic ocular disease causes significant ocular morbidity and can have a profound effect on patients’ vision and quality of life.
- The early initiation of appropriate oral antiviral therapy and treatment with topical corticosteroids is essential to minimize visual sequelae. Prophylactic maintenance treatment may be necessary to prevent recurrences.
A viral etiology is usually suspected when a patient presents with unilateral, acute, recurrent or chronic hypertensive anterior uveitis that is often associated with iris atrophy.3 The keratic precipitates (KPs) tend to be small to medium in size (Figures 1A and 2A) and may be pigmented.4 Corneal scars or corneal hypoesthesia also suggest a HSV or VZV infection. Because there is an overlap of clinical features, distinguishing between HSV and VZV clinically can be difficult. Further complicating the diagnosis, cytomegalovirus can also present as hypertensive anterior uveitis with iris manifestations. Because appropriate antiviral therapy differs depending on the etiology of the virus, obtaining a sample of aqueous for polymerase chain reaction or Goldmann-Witmer coefficient to provide a specific diagnosis is important.5
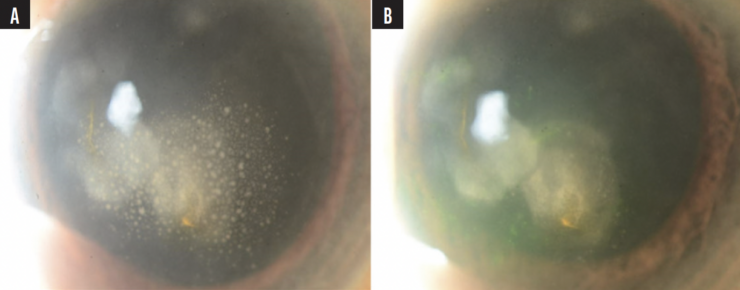
Figure 1. Slit-lamp photograph of HSV anterior uveitis presenting with small- to medium-sized KPs and associated corneal stromal scarring (A) and interval resolution of KPs with treatment (B).
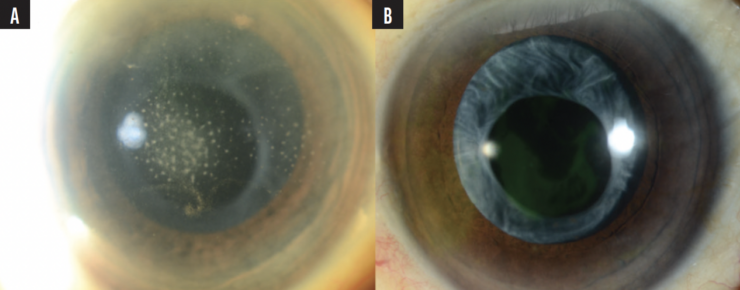
Figure 2. Slit-lamp photograph of VZV anterior uveitis presenting with small- to medium-sized KPs (A) with interval resolution of KPs with treatment (B).
PHARMACOLOGIC TREATMENT OF HSV AND VZV ANTERIOR UVEITIS
Herpetic ocular disease causes significant ocular morbidity. The recurrent nature of the disease and the inability of treatment to clear latent virus can profoundly affect patients’ vision and quality of life. The treatment of viral anterior uveitis depends on the causative agent and the host’s immune status. The mainstay is oral antiviral and topical antiinflammatory therapy. The former can inhibit viral replication and reduce the severity and number of recurrences, but it is not virucidal.6
During the acute disease phase, intensive topical corticosteroids are required to dampen the severe inflammatory response. The strength and duration of therapy can vary and is patient dependent. In general, treatment is tapered slowly over several months.7 In some situations, low-dose, long-term maintenance therapy with topical corticosteroids is essential to control inflammation and prevent recurrences.
Topical cycloplegics are administered to reduce pain and prevent the formation of posterior synechiae. Antiglaucoma medications are often necessary because many eyes demonstrate increased IOP at presentation. Prostaglandin analogues are largely avoided owing to concerns about reactivating a viral infection. There is little evidence, however, that this class of drug disrupts the blood-aqueous barrier.8
Oral acyclovir or valacyclovir are the most used antiviral agents for HSV and VZV acute anterior uveitis. Famciclovir is a third option. Brivudine is prescribed when a patient’s response to treatment is inadequate. Early initiation of therapy is recommended to limit the severity of the infection. Systemic resorption of oral acyclovir is unpredictable and varies according to the pH of gastric acid.9 The drug is administered five times per day during the induction phase. This frequency may affect patient compliance. Valacyclovir, a prodrug of acyclovir, has enhanced pharmacokinetics with more reliable intestinal absorption, resulting in more consistent systemic plasma levels.10 Additionally, the drug is administered three times daily during the induction phase, which may improve patient compliance. Valacyclovir is as effective as acyclovir in preventing ocular complications.11 Both systemic antiviral therapies have a good long-term safety profile and are well tolerated. Patients are also at low risk of developing acyclovir resistance.12 Thrombocytopenia and nephrotoxicity, however, can develop with the use of acyclovir and valacyclovir, especially in patients with underlying renal impairment.13
Prophylactic treatment becomes necessary if an individual experiences recurrent or chronic infection with HSV or VZV. Topical acyclovir may generate high aqueous humor levels, but it appears to have little prophylactic value against ocular complications compared to systemic acyclovir.14 Prophylaxis with long-term systemic antiviral therapy can reduce the rate of recurrence in HSV15 and VZV,16 but there is no consensus on the indication for or duration and dosage of maintenance treatment because published data are lacking.
TREATMENT APPROACHES AND CONSIDERATIONS
HSV anterior uveitis. In most cases, the use of oral antiviral agents with topical corticosteroids effectively controls inflammation in approximately 4 weeks (Figure 1). Oral acyclovir 400 mg administered five times per day17 or valacyclovir 500 mg administered three times per day does not decrease the recurrence risk after treatment ceases. Treatment duration and risk of recurrence can be reduced, however, with a maintenance dose of either acyclovir 400 mg two times per day or valacyclovir 500 mg once per day.
The duration of treatment at induction dose differs for each case but usually lasts several weeks until the inflammation is brought under control. The dose then may be reduced to a maintenance dose. We prefer to continue maintenance therapy for about 3 months, during which time topical corticosteroids are gradually tapered if possible. For recurrent disease, we continue maintenance therapy for at least 24 months while reducing topical corticosteroid therapy to a minimum and reviewing the possibility of halting antiviral therapy. Some patients may need lifelong antiviral therapy. A reduced dose of acyclovir is necessary for patients with reduced creatinine clearance.
VZV anterior uveitis. A regimen of oral acyclovir 800 mg administered five times daily for 10 days, given within 72 hours of the onset of skin lesions,16 is sufficient to treat the keratitis and anterior uveitis (Figure 2). Alternatively, valacyclovir 1 g or famciclovir 500 mg may be administered three times daily.
Chronic or recurrent VZV disease may develop, especially in an immunocompromised patient, and a maintenance dose of acyclovir 400 mg three times daily or valacyclovir 500 mg twice daily may be required.18 We usually try to discontinue the medication after 3 months. If the uveitis recurs, we continue maintenance therapy and assess the possibility of discontinuation after 12 months. If this fails, lifelong maintenance therapy may be indicated.
Topical corticosteroids administered during the acute phase are gradually tapered to a minimum dose.
Immunocompromised patients. The diagnosis and treatment of anterior uveitis in these patients can be challenging because of an atypical clinical course, severe presentation, and more frequent recurrences. The individual’s visual prognosis depends on early diagnosis and prompt treatment.
Pediatric herpetic disease. Aggressive treatment is necessary in children because they have a greater inflammatory response than adults. The former may therefore rapidly develop corneal scarring and opacification that induces irregular astigmatism and refractive amblyopia.19 Bilateral HSV ocular disease may also occur. About half of these patients experience recurrence without antiviral suppressive treatment.
CONCLUSION
A viral etiology should be suspected in patients who present with a unilateral, hypertensive anterior uveitis with characteristic KP patterns and iris changes. Aqueous humor analysis is recommended for a definitive diagnosis. The early initiation of appropriate oral antiviral therapy and treatment with topical corticosteroids are essential to minimize visual sequelae. Prophylactic maintenance treatment may be necessary to prevent recurrences.
1. Radosavljevic A, Agarwal M, Chee SP, Zierhut M. Epidemiology of viral induced anterior uveitis. Ocul Immunol Inflamm. 2022;30(2):297-309.
2. Van der Lelij A, Ooijman FM, Kijlstra A, Rothova A. Anterior uveitis with sectoral iris atrophy in the absence of keratitis: a distinct clinical entity among herpetic eye diseases. Ophthalmology. 2000;107(6):1164-1170.
3. Jap A, Chee SP. Viral anterior uveitis. Curr Opin Ophthalmol. 2011;22(6):483-488.
4. Chan NS, Chee SP. Demystifying viral anterior uveitis: a review. Clin Exp Ophthalmol. 2019;47(3):320-333.
5. Westeneng AC, Rothova A, de Boer JH, de Groot-Mijnes JD. Infectious uveitis in immunocompromised patients and the diagnostic value of polymerase chain reaction and Goldmann-Witmer coefficient in aqueous analysis. Am J Ophthalmol. 2007;144(5):781-785.
6. Pleyer U, Chee SP. Current aspects on the management of viral uveitis in immunocompetent individuals. Clin Ophthalmol. 2015;9:1017-1028.
7. Kardeş E, Bozkurt K, Sezgin Akçay Bİ, Ünlü C, Aydoğan Gezginaslan T, Ergin A. Clinical features and prognosis of herpetic anterior uveitis. Turk J Ophthalmol. 2016;46(3):109-113.
8. Horsley MB, Chen TC. The use of prostaglandin analogs in the uveitic patient. Semin Ophthalmol. 2011;26(4-5):285-289.
9. Wilson CG, Washington N, Hardy JG, Bond SW. The influence of food on the absorption of acyclovir: a pharmacokinetic and scintigraphic assessment. Int J Pharm. 1987;38(1):221-225.
10. MacDougall C, Guglielmo BJ. Pharmacokinetics of valaciclovir. J Antimicrob Chemother. 2004;53(6):899-901.
11. Colin J, Prisant O, Cochener B, Lescale O, Rolland B, Hoang-Xuan T. Comparison of the efficacy and safety of valaciclovir and acyclovir for the treatment of herpes zoster ophthalmicus. Ophthalmology. 2000;107(8):1507-1511.
12. Tyring SK, Baker D, Snowden W. Valacyclovir for herpes simplex virus infection: long-term safety and sustained efficacy after 20 years’ experience with acyclovir. J Infect Dis. 2002;186 suppl 1:S40-S46.
13. Izzedine H, Launay-Vacher V, Deray G. Antiviral drug-induced nephrotoxicity. Am J Kidney Dis. 2005;45(5):804-817.
14. Neoh C, Harding SP, Saunders D, et al. Comparison of topical and oral acyclovir in early herpes zoster ophthalmicus. Eye (Lond). 1994;8(pt 6):688-691.
15. Acyclovir for the Prevention of recurrent herpes simplex virus eye disease. Herpetic Eye Disease Study Group. N Engl J Med. 1998;339(5):300-306.
16. Hoang-Xuan T, Büchi ER, Herbort CP, et al. Oral acyclovir for herpes zoster ophthalmicus. Ophthalmology. 1992;99(7):1062-1070; discussion 1070-1071.
17. A controlled trial of oral acyclovir for iridocyclitis caused by herpes simplex virus. The Herpetic Eye Disease Study Group. Arch Ophthalmol. 1996;114(9):1065-1072.
18. Tugal-Tutkun I, Cimino L, Akova YA. Review for disease of the year: varicella zoster virus-induced anterior uveitis. Ocul Immunol Inflamm<. 2018;26(2):171-177.
19. Vadoothker S, Andrews L, Jeng BH, Levin MR. Management of herpes simplex virus keratitis in the pediatric population. Pediatr Infect Dis J. 2018;37(9):949-951.



