Fundus photography is a commonly used diagnostic procedure for detecting retinal disorders and systemic vascular diseases such as hypertension and diabetes. The manual interpretation of fundus images, however, can be time consuming and is prone to interobserver variability. Interest in the development of AI-based systems for the automated interpretation of fundus photographs is therefore growing.
THE FUNDUS AND SYSTEMIC VASCULAR DISEASE
AI-based fundus photography algorithms focus on diabetic retinopathy and glaucoma for the purposes of screening, diagnosis, and monitoring.1,2 Vascular features of the fundus, however, are associated not only with ocular disorders but also with systemic vascular disease. Small vessel or microvascular pathology influences the processes leading to the development of cardiovascular disease and its risk factors. Unfortunately, microcirculation is difficult to access, and robust microvascular biomarkers have yet to be developed. The retinal vasculature, in contrast, can be accessed noninvasively. It therefore represents a unique biologic model for the study of microvascular abnormalities and pathology associated with cardiovascular disease.
Numerous studies that have investigated the relationship between retinal microvascular abnormalities and systemic disease have shown that impaired retinal vascular network patterns were associated with an elevated cardiovascular risk assessment score, cardiovascular mortality, and an increase in cardiovascular risk factors.2-6 The field of research investigating ocular biomarkers of systemic disease is now referred to as oculomics,2 and the scientific literature on this topic is growing.3
APPROACHES
Various approaches are used to detect systemic vascular disorders with AI-based fundus photographs.
No. 1. Automated image processing software such as Quartz PCI (Quartz Imaging) can segment retinal vessel morphometry (Figure 1), differentiate arterioles and venules, and quantify retinal geometric features.4
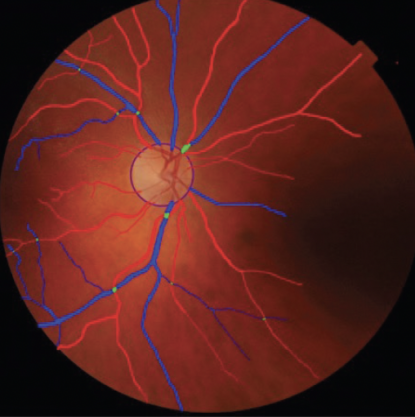
Figure 1. Retinal vessel segmentation using personal software (ISPED, team LEHA at Bordeaux Population Health Research Center, unpublished data, 2023).
No. 2. Deep learning algorithms were created to measure retinal-vessel caliber automatically from retinal photographs. The ground truth for these models was previously measured by a trained grader.5
No. 3. Deep learning algorithms based on a nonfeature-based approach can be used to classify systemic vascular disorders.
EARLY RESULTS
In a landmark study in 2018, Poplin et al predicted classic cardiovascular risk factors based on retinal fundus images alone with a convolutional neural network and UK Biobank and EyePACS datasets.6 Several factors had not previously been thought to be present and quantifiable in retinal images, including age (mean absolute error, 3.26 years; 95% CI [3.22, 3.31]), sex (area under the receiver operating characteristic curve = 0.97; 95% CI [0.96, 0.97]), smoking status (current smoker; area under the receiver operating characteristic curve = 0.71; 95% CI [0.70, 0.73]), and systolic blood pressure [mean absolute error, 11.35 mm Hg; 95% CI [11.18, 11.51]).
Cheung et al demonstrated a significant prediction rate of retinal arteriolar caliber for incident cardiovascular events (ie, stroke, myocardial infarction, and cardiovascular death). They showed that a narrower central retinal arteriolar equivalent (hazard ratio per standard deviation decrease, 1.13 [1.02–1.26]) was independently associated with incident cardiovascular events after adjusting for cardiovascular risk factors.5
The aforementioned studies and others indicate that AI-based algorithms can be used to interpret fundus photographs and have the potential to improve the early detection of systemic vascular disorders and facilitate screenings in clinical settings (Figure 2). This could revolutionize the field of ophthalmology and improve patients’ overall health outcomes.
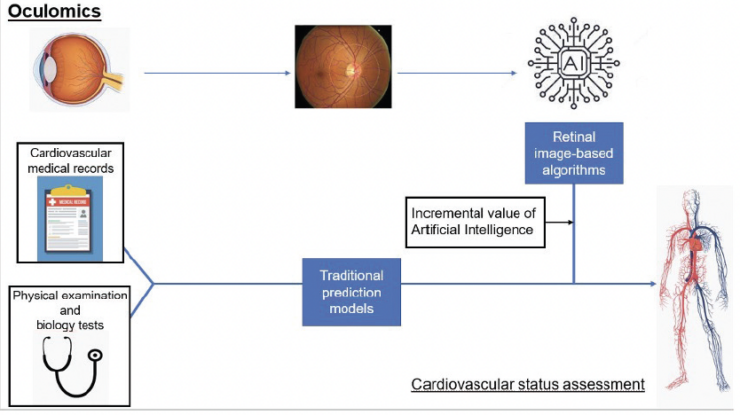
Figure 2. Incremental value of AI-based retinal biomarkers to detect systemic vascular disorders.
LIMITATIONS
Currently, there are two main obstacles to the widespread adoption of AI-based systems in clinical settings. First, the development of AI-based algorithms requires a significant amount of data for training and validation, which may be difficult to obtain in certain medical fields. Second, the lack of standardization in fundus photography and variations in image quality can affect the accuracy of AI-based interpretation. Standardized protocols for capturing and analyzing fundus photographs with AI-based algorithms therefore must be developed.
Further research is required to evaluate the reliability, accuracy, and cost-effectiveness of AI-based systems for the automated interpretation of fundus photographs in clinical settings. These efforts could improve the clinical relevance of AI-based systems and increase their adoption in medical practice.
1. Grzybowski A, Singhanetr P, Nanegrungsunk O, Ruamviboonsuk P. Artificial intelligence for diabetic retinopathy screening using color retinal photographs: from development to deployment. Ophthalmol Ther. Published online March 2, 2023. doi:10.1007/s40123-023-00691-3
2. Wagner SK, Fu DJ, Faes L, et al. Insights into systemic disease through retinal imaging-based oculomics. Transl Vis Sci Technol. 2020;9(2):6.
3. Arnould L, Meriaudeau F, Guenancia C, et al. Using artificial intelligence to analyse the retinal vascular network: the future of cardiovascular risk assessment based on oculomics? A narrative review. Ophthalmol Ther. 2023;12(2):657-674.
4. Tapp RJ, Owen CG, Barman SA, et al. Associations of retinal microvascular diameters and tortuosity with blood pressure and arterial stiffness: United Kingdom Biobank. Hypertension. 2019;74(6):1383-1390.
5. Cheung CY, Xu D, Cheng CY, et al. A deep-learning system for the assessment of cardiovascular disease risk via the measurement of retinal-vessel calibre. Nat Biomed Eng. 2021;5(6):498-508.
6. Poplin R, Varadarajan AV, Blumer K, et al. Prediction of cardiovascular risk factors from retinal fundus photographs via deep learning. Nat Biomed Eng. 2018;2(3):158-164.