The management of retinoblastoma has gradually advanced in recent decades from enucleation to globe conservation in most cases (Figure 1).1 Currently, use of external beam radiotherapy (EBRT) is restricted to large tumors that are resistant to chemoreduction or that have extraocular extensions.2 Cataract formation, one of the frequently documented complications of EBRT, typically occurs 1 to 3 years after irradiation.3,4 Rarely, cataract can be the initial manifestation in eyes with retinoblastoma (see Symptoms of Retinoblastoma).5,6
There is a lack of uniform guidelines for management of cataract in the setting of retinoblastoma. Unsettled questions include the best surgical approach, the optimal length of retinoblastoma quiescence prior to cataract surgery, whether to preserve the posterior capsule, and whether to avoid anterior vitrectomy due to concern for reactivation or metastasis of the tumor.2,4,7-15 The purpose of this article is to review the literature and to suggest guidelines for the management of pediatric cataracts in eyes treated for retinoblastoma (eyetube.net/?v=olusq).
Intraocular surgery after treatment for retinoblastoma poses distinctive challenges. Consequential cataract formation further confounds the management of retinoblastoma by precluding visualization of the tumor. Surgical intervention for cataract in these eyes increases concerns about the patient's systemic outcome because of the risk of viable tumor seeding.
Watch it Now
The surgical steps in pediatric cataract surgery in eyes with retinoblastoma are depicted.
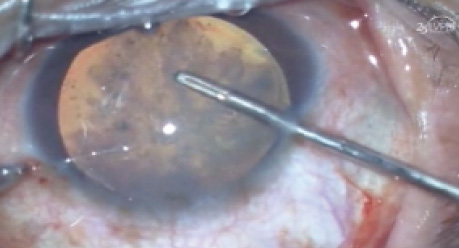
Nonetheless, visual rehabilitation for cataracts is justified in certain clinical settings, especially if the tumor is considered to be clinically stable or in regression. Quiescent interval is a key determinant of long-term success.16 It is crucial to weigh the expected benefits of visual rehabilitation against the risk of tumor dissemination or relapse.13,17
RECURRENCE, METASTASIS
Recurrence of retinoblastoma after intraocular surgery is a potentially serious problem (see Fast Facts). Tumor recurrence rate has been reported to range from 0% to 45% after various intraocular procedures.15
Payne and colleagues reviewed the results of 128 eyes that underwent cataract extraction for radiation-induced cataract following retinoblastoma treatment over a period of 22 years.15 They identified nine cases of retinoblastoma recurrence and no cases of systemic metastasis. In 42 eyes of 38 patients with retinoblastoma, Brooks and coworkers reported three cases of tumor recurrence.8 In each of these eyes, surgery was approached via a pars plana or pars plicata incision, and all three eyes had anterior tumors and vitreous seeding prior to EBRT with persistent vitreous haze present at the time of cataract surgery. Orbital exenteration was required in one case for subconjunctival retinoblastoma recurrence that developed at the site of the cataract incision. There was no systemic metastasis.
Reviewing results of all intraocular surgeries after retinoblastoma treatment, Honavar et al reported the recurrence of retinoblastoma in five of 34 eyes (21%) that underwent cataract surgery, all five of which underwent subsequent enucleation.13 None of the patients who underwent cataract surgery developed metastasis. Most of the recurrences occurred within the first year after surgery, with the longest interval in that series being 19 months. Patients needing a scleral buckling procedure or pars plana vitrectomy (PPV) seemed to be at greater risk for retinoblastoma recurrence compared with those needing cataract surgery. Of all patients in their series, including anterior segment and posterior segment surgeries, patients with a favorable outcome (26 eyes) had a median quiescent interval of 26 months, whereas patients with an unfavorable outcome (19 eyes) had a median quiescent interval of 6 months. An unfavorable outcome was defined as the presence of tumor recurrence, need for enucleation, or systemic metastasis.
Moshfeghi and colleagues reported one case of retinoblastoma recurrence after cataract surgery, ultimately requiring enucleation.14 Although no predisposing factors were identified in this patient, the quiescent interval was only 12 months prior to cataract surgery. Adequate tumor treatment and a sufficient quiescent interval appear to be crucial in preventing tumor recurrence and metastasis.
While the optimal quiescent interval is unknown, it is desirable to have at least 9 months of quiescence.15 Miller and colleagues reported that they observe for a minimum of 18 months after the conclusion of all tumor treatments before considering intraocular surgical management of radiation-induced cataract.2 Reported recurrence rates have varied, from 10% for Brooks et al,8 to 31% for Honovar et al,13 to 0% for Miller et al.2 The fact that Miller et al applied the longest interval of observation suggests a correlation between longer interval and lower rate of recurrence.
In a series reported by Osman and colleagues, one eye that experienced recurrence underwent cataract surgery 37 months after retinoblastoma treatment completion, but only 3 and 5 months elapsed in two other cases in which cataracts were removed for the sake of monitoring the tumor.18 This further suggests the importance of adequate tumor control before any surgical intervention. It also highlights the difficulty of weighing the risk of surgery in the presence of an undiagnosed recurrence against the risk of delay of diagnosing a recurrence due to an opaque lens causing insufficient tumor visualization. Despite the unknown optimal quiescent interval, a detailed review of the literature found no tumor recurrences when the quiescent interval was at least 16 months.15
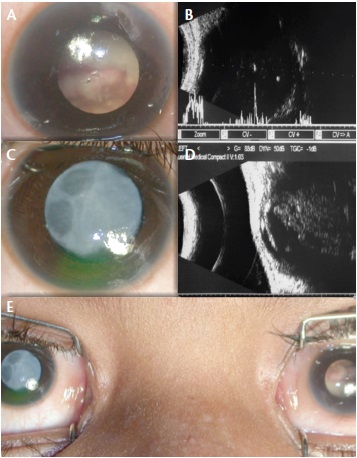
Figure 1. A child with bilateral retinoblastoma associated with cataract in the right eye: Left eye with leucocoria due to large tumor filling the globe and touching the posterior surface of the crystalline lens (A). Ultrasonography of the left eye showing intraocular mass with specks of calcification (B). Right eye showing total opacification of crystalline lens (C). Ultrasonography of the left eye showing intraocular mass with specks of calcification, touching the posterior surface of the crystalline lens (D). External photograph of both eyes revealing cataract in the right eye and leucocoria due to retinoblastoma in the left eye (E).
The risk of tumor recurrence and metastasis appears to be higher in patients who have undergone posterior segment surgeries such as PPV or scleral buckling. Honavar and colleagues reported the largest series,13 and they suspected that eyes that required a scleral buckling procedure or PPV were more likely to have advanced retinoblastoma and, therefore, a higher risk of recurrence. Moshfeghi and colleagues concluded that rhegmatogenous retinal detachments are an ominous sign for both visual acuity and eye salvage.14 They hypothesized that tumor reactivation could place mechanical stress on the retina, resulting in stretching of already damaged tissues and, thus, allowing microscopic breaks to form. Eyes that require posterior segment surgery may be associated with a higher risk of tumor reactivation.
SIGNS AND SYMPTOMS OF RETINOBLASTOMA1,2
• A white-yellow mass or glow seen through the pupil, often noticed when light is shone in the eye
• Complaints of poor vision
• Eyes that appear to be looking in different directions
• One or both eyes turning in- or outward
• Eye redness and swelling
• Pain from increased IOP as the tumor grows
1. Disease Information. Solid Tumor: Retinoblastoma. St Jude Children's Research Hospital website. www.stjude. org/retinoblastoma. Accessed February 23, 2015. 2. Diseases and Conditions: Retinoblastoma. Mayo Clinic website. www.mayoclinic.org/diseases-conditions/ retinoblastoma/basics/symptoms/con-20026228. Accessed February 23, 2015.
BARRIER PRESERVATION
Controversies in the surgical approach to cataract management after retinoblastoma treatment include management of the posterior capsule and the anterior vitreous: that is, whether to leave the posterior capsule intact or perform posterior capsulectomy with or without vitrectomy. These decisions take on great significance because of the attendant risk of tumor reactivation or metastasis.13
Theoretically, the posterior capsule can serve as a protective barrier against the spread of viable tumor cells from the eye. Therefore, the reasoning goes, the posterior capsule should be left intact whenever possible. However, it is often essential to accomplish a primary posterior capsulectomy with or without anterior vitrectomy in younger children, even in the setting of prior treatment for retinoblastoma.
Limbal and pars plana approaches to cataract removal have been used with good success in children after retinoblastoma treatment. Brooks et al advised against pars plana incisions based on their experience of tumor recurrence,8 but other series have not reported tumor recurrence with a pars plana approach. Miller and coworkers reported a series of 16 eyes, all of which underwent PPV in addition to cataract extraction, and they saw no evidence of tumor recurrence.2
We recommend avoiding opening the posterior capsule if there is no plaque and the age of the patient permits. However, because posterior capsular plaque is common after EBRT, it is sometimes necessary to perform either plaque peeling or posterior capsulectomy in order to clear the visual axis. In such cases, an attempt should be made to perform a manual posterior continuous curvilinear capsulorrhexis (PCCC), leaving intact the anterior vitreous face (AVF) and avoiding subsequent vitrectomy. The PCCC maneuver can be beneficial even in the absence of posterior capsule plaque. Integrity of the AVF can be checked using triamcinolone.
If retinoblastoma regression has been deemed stable for at least 6 to 12 months after cataract surgery, Nd:YAG laser posterior capsulotomy may be cautiously performed when required.
We believe the risks and benefits of primary posterior capsulectomy and anterior vitrectomy should be considered on a case-by-case basis, taking into account the location of the cataract, the age of the patient, the feasibility of performing secondary Nd:YAG laser, the length of the quiescent period, and the location and stage of the tumor.
Dissemination of retinoblastoma cells through the cataract surgery incision has been reported.8 A clear corneal incision (CCI) may reduce the risk of inadvertent conjunctival implantation of viable tumor cells. CCIs—unlike limbal or scleral incisions, which may be obscured by overlying conjunctiva—allow direct inspection of the incision site for tumor recurrence.13
Cytologic examination of vitrectomy fluid can provide direct intraoperative evidence of viable retinoblastoma cells. Prompt enucleation and adjuvant chemotherapy with or without orbital radiotherapy may be considered in the event the presence of cells is confirmed. Long-term follow-up is warranted to detect possible tumor recurrence and systemic metastasis after cataract surgery.
Miller and colleagues reported cystoid macular edema in five of 16 eyes (31%) and the development of iridocyclitis in three of 16 eyes (19%).2 These complications were transient and responded to topical antiinflammatory therapy. No local tumor recurrence, orbital tumors, or metastatic disease was detected during follow-up. No lens displacement, persistent inflammation, radiation vasculopathy or radiation optic neuropathy, or retinal detachment was seen.
POSTOPERATIVE VISION
Even after a successful surgical outcome, final visual acuity depends on several factors such as density of amblyopia, residual refraction, macular tumor location, complications of radiation (keratopathy and/or retinopathy), optic atrophy, retinal detachment, and secondary visual axis opacification. Final visual acuity outcome is generally correlated with the extent of preoperative macular involvement.
AT A GLANCE
- Visual rehabilitation for cataracts is justified in certain clinical settings, especially if the retinoblastoma is considered to be clinically stable or in regression.
- Modern cataract surgery—including a clear corneal approach, lens aspiration with or without posterior capsulectomy and anterior vitrectomy and with or without IOL implantation—is a safe and effective surgical approach in radiation-induced cataract in the setting of well-controlled retinoblastoma.
- Surgeons should have detailed conversations with families of retinoblastoma patients that reflect realistic prospects; this is especially important as there is a chance of tumor recurrence after intraocular procedures.
It is important to remember that eyes treated for retinoblastoma are poor candidates for contact lens wear due to dry eye associated with poor tear production from the irradiated lacrimal glands and ocular surface. Thus, in unilateral cases, IOL implantation is a reasonable option for the correction of aphakia. Implantation of an IOL may not by itself increase the risk of recurrence of retinoblastoma or systemic metastasis. Therefore, this option can be considered in order to provide optimal visual rehabilitation after cataract surgery.18 Portellos and Buckley reported the safety of extracapsular cataract extraction and posterior chamber IOL implantation in combination with pars plana posterior capsulectomy and anterior vitrectomy in a series of eight patients (11 eyes) with radiation-induced cataracts after retinoblastoma treatment.4
Due to radiation or early enucleation, the child may have a fellow eye that is significantly enophthalmic in appearance. If this is the case, a prescription of 6.00 or 7.00 D of sphere for an already poorly seeing eye may be considered. Another consideration is an anophthalmic socket with prosthesis, which will give the optical illusion of a larger and more symmetric appearance.19
If patching or atropine penalization is proposed for the patient with amblyopia after retinoblastoma treatment, the overall clinical picture should be considered. Is the family overwhelmed? Is the retina so distorted from residual tumor and treatment scars that vision may not improve? Remember that these are not normal amblyopic eyes. They often have extensive pathology that precludes improvement in vision even with the most rigorous patching regimen. Sometimes, especially in eyes of young children that have received EBRT, sensitivity to the patch adhesive can occur.19
CONCLUSION
Modern cataract surgery—including a clear corneal approach, lens aspiration with or without posterior capsulectomy and anterior vitrectomy and with or without IOL implantation—is a safe and effective surgical approach in radiation-induced cataract in the setting of well-controlled retinoblastoma. We recommend a minimum interval of 9 months' observation after the conclusion of all tumor treatments before considering intraocular surgical management of radiation-induced cataract.
Fast facts: Retinoblastoma by the numbers
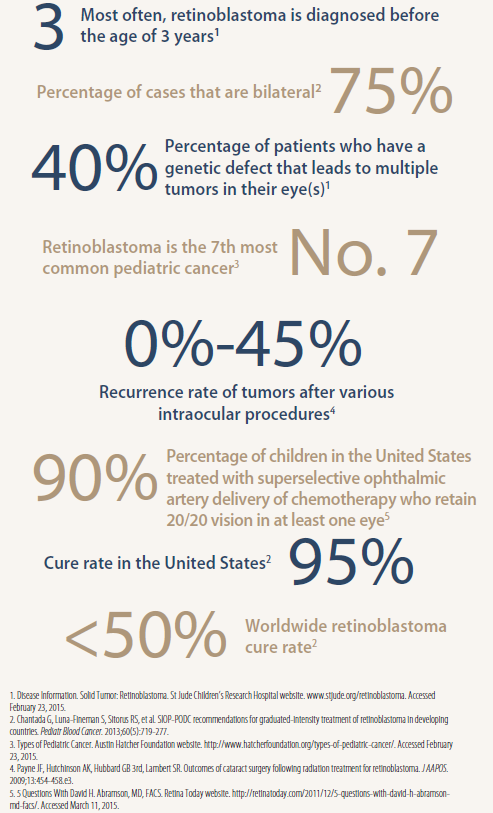
Irrespective of surgical strategy, it is important to have a detailed conversation with the family reflecting realistic prospects. As stated hitherto, amblyopia may be so dense, or retinal pathology so extensive, that there may not be an improvement in vision after cataract surgery. Although rare, tumor recurrence has been well documented in the literature and should be included in informed consent. n
1. Shields JA, Shields CL, Sivalingam V. Decreasing frequency of enucleation in patients with retinoblastoma. Am J Ophthalmol. 1989;108:185-188.
2. Miller DM, Murray TG, Cicciarelli NL, Capo H, Markoe AM. Pars plana lensectomy and intraocular lens implantation in pediatric radiation-induced cataracts in retinoblastoma. Ophthalmology. 2005;112:1620-1624.
3. Schipper J, Tan KE, van Peperzeel HA. Treatment of retinoblastoma by precision megavoltage radiation therapy. Radiother Oncol. 1985;3:117-132.
4. Portellos M, Buckley EG. Cataract surgery and intraocular lens implantation in patients with retinoblastoma. Arch Ophthalmol. 1998;116:449-452.
5. Hasan SJ, Brooks M, Ambati J, Kielar R, Stevens JL. Retinoblastoma with cataract and ectopia lentis. J AAPOS. 2003;7:425-427.
6. Brown GC, Shields JA, Oglesby RB. Anterior polar cataracts associated with bilateral retinoblastoma. Am J Ophthalmol. 1979;87:276.
7. Monge OR, Flage T, Hatlevoll R, Vermund H. Sightsaving therapy in retinoblastoma. Experience with external megavoltage radiotherapy. Acta Ophthalmol. 1986;64:414-420.
8. Brooks HL Jr, Meyer D, Shields JA, Balas AG, Nelson LB, Fontanesi J. Removal of radiation-induced cataracts in patients treated for retinoblastoma. Arch Ophthalmol. 1990;108:1701-1708.
9. Madreperla SA, Hungerford JL, Cooling RJ, Sullivan P, Gregor Z. Repair of late retinal detachment after successful treatment of retinoblastoma. Retina. 2000;20:28-32.
10. Bhattacharjee H, Bhattacharjee K, Chakraborty D, Talukdar M, Das D. Cataract surgery and intraocular lens implantation in a retinoblastoma case treated by external-beam radiation therapy. J Cataract Refract Surg. 2003;29:1837-1841.
11. Shanmugam MP, Rao SK, Khetan V, Kumar PJ. Cataract surgery and intraocular lens implantation in retinoblastoma. J Cataract Refract Surg. 2004;30:1825-1826.
12. Sinha R, Titiyal JS, Sharma N, Vajpayee RB. Management of radiotherapy-induced cataracts in eyes with retinoblastoma. J Cataract Refract Surg. 2004;30:1145-1146.
13. Honavar SG, Shields CL, Shields JA, Demirci H, Naduvilath TJ. Intraocular surgery after treatment of retinoblastoma. Arch Ophthalmol. 2001;119:1613-1621.
14. Moshfeghi DM, Wilson MW, Grizzard S, Haik BG. Intraocular surgery after treatment of germline retinoblastoma. Arch Ophthalmol. 2005;123:1008-1012.
15. Payne JF, Hutchinson AK, Hubbard GB 3rd, Lambert SR. Outcomes of cataract surgery following radiation treatment for retinoblastoma. J AAPOS. 2009;13:454-458.e3.
16. Shields JA, Shields CL, eds. Intraocular Tumours: A Text and Atlas. Philadelphia: WB Saunders; 1992:302-392.
17. Shields CL, Honavar S, Shields JA, Demirci H, Meadows AT. Vitrectomy in eyes with unsuspected retinoblastoma. Ophthalmology. 2000;107:2250-2255.
18. Osman IM, Abouzeid H, Balmer A, et al. Modern cataract surgery for radiation-induced cataracts in retinoblastoma. Br J Ophthalmol. 2011;95:227-230.
19. Hoehn ME, Irshad F, Kerr NC, Wilson MW. Outcomes after cataract extraction in young children with radiation-induced cataracts and retinoblastoma. J AAPOS. 2010;14:232-234.
Swathi Kaliki, MD
- Consultant, Institute for Eye Cancer, L.V. Prasad Eye Institute, Kallam Anji Reddy Campus, Hyderabad, India
- kalikiswathi@yahoo.com
- Financial disclosure: None
Ramesh Kekunnaya, FRCS
- Consultant and Head of Pediatric Services, Jasti V. Ramanamma Children's Eye Care Center, L.V. Prasad Eye Institute, Kallam Anji Reddy Campus, Hyderabad, India
- rameshak@lvpei.org; drrk123@gmail.com
- Financial disclosure: None
Muralidhar Ramappa, MD
- Consultant in Pediatric Cornea and Anterior Segment Services, L.V. Prasad Eye Institute, Kallam Anji Reddy Campus, Hyderabad, India
- muralidhar@lvpei.org
- Financial disclosure: None
Rupal H. Trivedi, MD, MSCR
- Associate Professor, Miles Center For Pediatric Ophthalmology, Storm Eye Institute, Department Of Ophthalmology, Medical University Of South Carolina, Charleston, South Carolina
- trivedi@musc.edu
- Financial disclosure: None

