Due to the increasing relative incidences of age-related cataract and age-related macular degeneration (AMD) and the increasing longevity of the population, ophthalmologists are now frequently faced with the management of patients with both visually significant cataract and AMD. Our article focuses on pertinent aspects of IOL selection in this group of patients.
Controversy exists regarding the appropriate timing of cataract surgery, methods of preoperative evaluation, and appropriate selection of IOLs in these patients. A complete understanding of the visual compromise in various stages of AMD is vital to maximize the patient's visual function postoperatively (see Visual Compromises According to the Stages of AMD).
IOL SELECTION
When the time arrives to counsel patients on IOL selection, the discussion will differ depending on the patient's clinical scenario. There are three broad categories of patients for whom there are subtle differences in IOL selection strategy, which are discussed below and depicted in the corresponding figures.
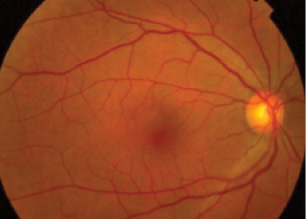
Figure 1. Fundus photograph of a patient without clinical evidence of macular disease but with a strong family history.
Category No. 1: Patients without clinical evidence of macular disease (no drusen or pigmentary changes) but with a strong family history (Figure 1). These patients are normal and, hence, candidates for all IOL technologies. Direct counseling on the potential pitfalls of multifocal IOLs should be carried out prior to cataract surgery.
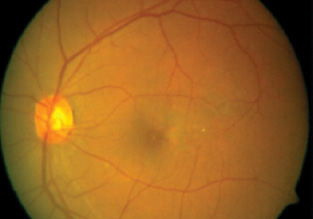
Figure 2. Fundus photograph of a patient with early maculopathy but relative sparing of Snellen visual acuity.
Category No. 2: Patients with early maculopathy (soft drusen and/or pigmentary changes) with relative sparing of Snellen visual acuity (Figure 2). These patients are difficult to counsel given their preserved visual acuity. Loss of contrast sensitivity at lower spatial frequencies is present even in mild forms of AMD. Therefore, implantation of a multifocal IOL is a relative contraindication.
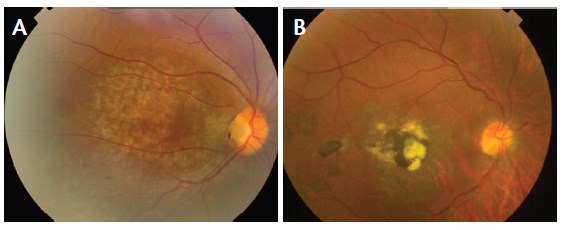
Figure 3. Fundus photographs of patients with moderate to severe AMD with geographic atrophy (A) and scarred exudative changes (B).
Category No. 3: Patients with moderate to severe AMD (presence of drusen, geographic atrophy, and/or exudative changes; Figure 3). These patients should receive monofocal or toric IOLs, as the benefits of an accommodating IOL would not be appreciated and a multifocal IOL would actually have deleterious effects on visual function because of the additional loss of contrast sensitivity. The use of an intraocular telescopic magnifier (discussed below) may be tried in this category of patients.
Implantable Magnification Devices
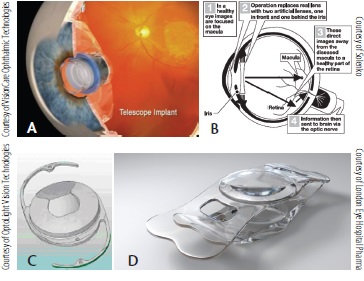
Low vision aids implanted in the anterior segment apply the concept of retinal image enlargement by using a combination of a minus and a plus lens as a magnifier. Examples of low vision aid implants include the Implantable Miniature Telescope (A); IOL-VIP System (B), Lipschitz Mirror Implant (C), and iolAMD lens (D).
ADVANTAGES, DISADVANTAGES OF IOL TYPES IN AMD
Aspheric IOLs. These IOLs negate the spherical aberration present in the cornea. Decreasing spherical aberration can improve contrast sensitivity, particularly in mesopic conditions, although depth of focus and distance corrected near acuity are often better with spherical IOLs. Thus, in theory, implantation of an aspheric design could be beneficial in AMD patients given the potential improvement in contrast sensitivity; however, functional visual benefits are difficult to demonstrate.
Blue-blocking IOLs. The advantages and disadvantages of blue-blocking IOLs have been a topic of debate for years. It has been conclusively shown that the macular protection afforded by violet- and blue-blocking IOLs is 20% less than that provided by a 53-year-old crystalline lens.1 Impaired dark adaption, which is seen with blue-blocking IOLs, has been associated with an increased risk of falling among the elderly.2 However, contrast sensitivity is not reduced by blue-blocking IOLs, and they have no reported deleterious effects in patients with maculopathy. The theoretical benefits of this technology in AMD patients remain to be elucidated.
Toric IOLs. IOLs with cylindrical correction have little impact on contrast sensitivity; they differ from monofocal IOLs only in their correction of corneal astigmatism. Thus, their use is not contraindicated in patients with AMD. If a patient has a cylindrical refraction of -4.00 D, a large central scotoma, and a cataract, he or she will always have a large scotoma that may limit the visual acuity to 20/200. Cataract surgery will improve this patient's quality of vision, and the implantation of a toric IOL will improve his or her overall visual acuity even more. Such patients will function better with toric correction.
Accommodating IOLs. IOLs that provide patients with some accommodative amplitude have little impact on contrast sensitivity and typically provide distance vision equivalent to monofocal IOLs. The level of maculopathy and the patient's visual potential dictate their use. Given the frequent need among patients with moderate to severe AMD for visual aids such as high-powered magnifiers, the relatively modest accommodation afforded by these IOLs in their present form has limited utility. An honest discussion with patients about the potential limitations of these technologies, particularly with respect to progression of maculopathy with time, is of utmost importance.
Multifocal IOLs. All multifocal IOLs, by the nature of their optical designs, reduce contrast sensitivity in mesopic conditions. In AMD patients, the additive effect of contrast sensitivity loss due to underlying maculopathy coupled with the image degradation inherent to current multifocal IOLs creates the potential for significant visual compromise.
Implantation of multifocal IOLs using a magnification strategy in cataractous eyes with AMD was assessed by Gayton et al.3 The investigators targeted a spherical equivalent of -2.00 D, yielding a 5.20 D near addition. They reported improvement of near UCVA in 90% of eyes and of distance BCVA in 70%. Although the use of diffractive multifocal IOLs in eyes with even early signs of maculopathy is questionable because of the associated contrast loss, the introduction of these IOLs as a potential low vision aid in advanced AMD opens a new chapter of potential applications.
IMPLANTable MAGNIFICATION DEVICES
A number of promising visual prosthetic devices designed for implantation in the anterior segment in patients with AMD have appeared in recent years (see Implantable Magnification Devices). These devices utilize the concept of enlarging the retinal image by using a combination of a minus and a plus lens as a magnifier. This is a concept that has been used in external visual rehabilitation devices for low-vision patients for many years. Implants that have shown promising results are detailed below.
Visual Compromises According to the Stage of AMDE
Early AMD
For patients with early AMD who have good Snellen visual acuity but a contrast sensitivity deficit, the appropriate selection of IOLs is a challenge. Although contrast sensitivity is not routinely measured, its loss has been shown to independently affect quality of life in patients with AMD. Appropriate selection of IOLs must take this loss of contrast sensitivity into account, and IOLs that would tend to decrease contrast sensitivity further might best be avoided.
Advanced AMD
Patients with advanced AMD have large central scotomas and poor macular function. In the past, because the visual prognosis for surgical intervention was poor in this group of patients, cataract surgery was often intentionally delayed until a dense cataract developed, precluding visualization of the posterior pole. However, despite their significant visual deficits, patients with advanced AMD frequently report subjective improvement in visual function after cataract surgery, often citing improved sensations of brightness, color saturation, and peripheral vision despite minimal gains in Snellen visual acuity. These modest improvements in visual function are greatly appreciated by most patients, and hence delaying surgery is not worthwhile given the potential risks of cataract surgery in a hard cataract, such as pseudophakic bullous keratopathy, posterior capsular tear, and dropped nucleus.
Implantable Miniature Telescope (IMT; VisionCare Ophthalmic Technologies). Developed by Isaac Lipshitz, MD, the IMT incorporates multiple wide-angle microoptics that function together with the cornea as a fixed-focus telephoto system.4 The device, with a glass cylinder housing the microoptics, is 4.4 mm long and 3.6 mm in diameter. Positioned in the capsular bag, this device protrudes through the pupil by 0.1 to 0.5 mm and allows clearance of about 2.5 mm from the anterior window of the device to the corneal endothelium. The rigid haptic loops are 13.5 mm in diameter. The prosthesis projects an enlarged image of the patient's central visual field onto the retina, thus reducing the size of the scotoma relative to the objects in the central field of vision. The implanted eye sees a 20° to 24° field of view due to the enlarged image projection.
Zonular stability and normal preoperative endothelial cell density are prerequisites for surgical implantation of this device following standard phacoemulsification.5 A 10- to 12-mm limbal or scleral incision accommodates the dimensions of the device. A large 7-mm capsulorrhexis allows easy implantation. Liberal use of a dispersive and a cohesive ophthalmic viscosurgical device is necessary to create space between ocular structures, to maintain the anterior chamber, and to coat the device and cornea for maximum protection of the endothelium.
Hudson et al evaluated the safety and efficacy of the IMT in 217 patients with bilateral end-stage AMD.6 At 1 year, 67% of implanted eyes achieved a 3-line or more improvement in distance BCVA, compared with 13% of fellow control eyes, and 53% achieved a 3-line or more improvement of distance and near BCVA. Mean distance and near BCVA improved by 3.5 and 3.2 lines, respectively. Endothelial cell density was reduced by 20% at 3 months and 25% at 1 year. There was no evidence that endothelial cell loss was accelerated by ongoing endothelial trauma after implantation. To achieve the desired retinal image, this device is implanted monocularly in the anterior segment for central vision, and the fellow eye provides peripheral vision for mobility and orientation.
IOL for Visually Impaired People (IOL VIP System; Solenko). This double IOL implant for visual rehabilitation of patients with macular disease consists of a biconcave high–minus-power IOL in the capsular bag and a biconvex high–plus-power IOL in the anterior chamber, creating an intraocular Galilean telescope with 1.3X magnification for distance. In a group of 40 eyes of 35 patients, this lens system was reported to be effective and well-tolerated and to improve patients' BCVA, reading magnification, and reading distance.7
Lipschitz Mirror Implant (LMI; OptoLight Vision Technologies). This conventional IOL incorporates two miniature mirrors in a Cassegrain reflecting telescope configuration to modify the image on the retina. The IOL has a dual optical system that ensures that light passing through the optic center is magnified by the Cassegrain telescope, whereas light passing through the periphery travels through the normal IOL configuration and is not magnified. The overall diameter is 13 mm, and the optic diameter is 6.5 mm. The anterior central mirror size is 1.4 mm, and the posterior donut-shaped mirror is 2.8 mm in diameter with a central hole diameter of 1.4 mm. The peripheral zone of the optic is akin to a normal IOL for undisturbed peripheral vision. The reflecting surfaces of the LMI are coated with multiple layers of dielectric coatings to create the mirror effect. The thickness of these mirrors is only 1 to 2 µm. The entire IOL is also coated with poly-para-xylylene polymer for biocompatibility.8
A new model of the mirror telescope, the OriLens, is a sulcus-fixated IOL that can be placed over an existing IOL in pseudophakic patients. This lens is in use in Europe and other markets not regulated by the FDA.
iolAMD lens (London Eye Hospital Pharma). Inspired by the spherical aberration of the Hubble telescope, the iolAMD was developed by Bobby Qureshi, FRCOphth, of the London Eye Hospital, and Pablo Artal, PhD, an optical physicist at the University of Murcia.9 The lens is based on the concept of an intraocular miniature telescope, combining two foldable lenses, one in the ciliary sulcus and the other in the capsular bag. The design enables two lenses, spaced 2 mm apart, to shift the focus of the image away from the damaged part of the macula and onto healthier retinal tissue. The commercially available platform consists of two hydrophobic acrylic plate haptic IOLs—a high–plus-powered lens (60.00 D) with a 5-mm optic and asymmetric haptics placed in the sulcus and a high–minus-powered lens (-50.00 to -60.00 D) with a 4-mm optic placed in the capsular bag. The asymmetric haptics of the sulcus IOL result in an offset of 0.85 mm between the two lenses. Together, they create a Galilean telescopic effect with about 1.3X magnification and 3º of foveal displacement.
AT A GLANCE
- Phacoemulsification with IOL implantation in patients with AMD, especially if the disease is advanced, produces only a modest improvement in visual function; however, this improvement is well appreciated by the patient.
- There are three broad categories of patients for whom there are subtle differences in IOL selection strategy: patients without clinical evidence of macular disease but with a strong family history, patients with early maculopathy, and patients with moderate to severe AMD.
The amount of magnification provided increases visual acuity without compromising visual field, therefore enabling binocular implantation. The lenses are made of a high refractive index, foldable, glistening-free material and can be implanted through a 3-mm incision. The first implantations in the United Kingdom took place in January 2014, and approximately 100 patients have been treated to date.
CONCLUSION
The modern cataract surgeon should be well aware of the various stages of AMD and its associated visual changes. Phacoemulsification with IOL implantation in these patients, especially if the disease is advanced, produces only a modest improvement in visual function. In a patient with advanced AMD, however, this modest improvement in visual function is well appreciated. Choosing a proper IOL in these compromised eyes is of paramount importance. n
1. Boettner E, Wolter J. Transmission of the ocular media. Invest Ophthalmol. 1962;1:776-783.
2. McMurdo ME, Gaskell A. Dark adaptation and falls in the elderly. Gerontology. 1991;37:221-224.
3. Gayton JL, Mackool RJ, Ernest PH, Seabolt RA, Dumont S. Implantation of multifocal intraocular lenses using a magnification strategy in cataractous eyes with age-related macular degeneration. J Cataract Refract Surg. 2012;38(3):415-418.
4. Lipshitz I, Loewenstein A, Reingewirtz M, Lazar M. An intraocular telescopic lens for macular degeneration. Ophthalmic Surg Lasers. 1997;28:513-517.
5. Lane SS, Kuppermann BD. The implantable miniature telescope for macular degeneration. Curr Opin Ophthalmol. 2006;17:94-98.
6. Hudson HL, Lane SS, Heier JS, et al; IMT-002 Study Group. Implantable miniature telescope for the treatment of visual acuity loss resulting from end-stage age-related macular degeneration: 1-year results. Ophthalmology. 2006;113(11):1987-2001.
7. Orzalesi N, Pierrottet CO, Zenoni S, Savaresi C. The IOL-Vip System: a double intraocular lens implant for visual rehabilitation of patients with macular disease. Ophthalmology. 2007;114; (5)860-865.
8. Agarwal A, Lipshitz I, Jacob S, et al. Mirror telescopic intraocular lens for age- related macular degeneration: design and preliminary clinical results of the Lipshitz macular implant. J Cataract Refract Surg. 2008;34(1):87-94.
9. iolAMD.com. http://iolamd.com/contact-us-patients.php. Accessed February 23, 2015.
Arup Chakrabarti, MS
- Chakrabarti Eye Care Centre, Kochulloor, Trivandrum, Kerala, India
- arupeye@gmail.com
- Financial disclosure: None
Meena Chakrabarti, MS
- Chakrabarti Eye Care Centre, Kochulloor, Trivandrum, Kerala, India
- drmeenachakrabarti@gmail.com
- Financial disclosure: None

