Pterygium is a triangular vascularized fibroelastic growth arising from the limbic conjunctiva that extends over the cornea, replacing the epithelium and Bowman membrane. A systematic review and meta-analysis of population-based studies estimated the prevalence of pterygium to be around 10.2% (range 6.3%–16.1%).1 The incidence increases with age and geographic latitude. Similarly, cataract is a common age-related condition; it is estimated that more than 100 million eyes have a visual acuity of less than 6/60 due to cataract and that perhaps three to four times that number of eyes have a visual acuity of less than 6/18 due to cataract.2
Pterygium has a causal relationship with exposure to UV-B radiation,3,4 and there is a high incidence of cataract in tropical countries such as India.5 With the incidence of both conditions so high in tropical countries, it is no wonder that many patients develop cataract and pterygium concomitantly. The question arises as to whether surgery for these concomitant conditions should be performed simultaneously or sequentially.
SURGical sequence
The case for simultaneous surgery. The susceptible population in tropical areas consists largely of daily wage earners without access to insurance, and loss of days of work on account of surgery amounts to loss of income. The proponents of simultaneous surgery argue that a single procedure results in reduced hospitalization, costs, morbidity, and loss of workdays compared with two procedures. For small pterygia, simultaneous surgery may not make a difference in the outcome; for larger, visually significant ones, however, sequential surgery can add significant morbidity and recovery time.
The case for sequential surgery. (See Three Cases for Sequential Surgery.) Large pterygia induce refractive astigmatism, which affects the outcome following cataract surgery.6 Hence, pterygia that interfere with keratometry (K) readings should be removed before proceeding with cataract surgery.
In eyes with pterygia, corneal topography is more valuable than keratometry because it provides a more detailed picture of the refractive status of the cornea, detecting curvature changes that induce astigmatism. Large, fleshy pterygia that are vascularized and inflamed change the shape of the cornea.
By contrast, small pterygia that have not changed the corneal curvature (Figure 1) are best left alone. In these eyes, cataract surgery can be done first, with pterygium excision planned later for refractive correction or cosmesis. Generally, only pterygia that are more than 3 mm in size cause topographical distortion (Figure 2).
Postoperative refractive surprises are prevented by ensuring accurate and stable keratometry. The K values used for IOL power calculation will not remain the same following excision of visually significant pterygia. Thus, when pterygium excision is combined with cataract surgery or performed after cataract surgery, the altered K values cause a refractive surprise.
AT A GLANCE
- For small pterygia, simultaneous surgery may not make a difference in the outcome; for larger, visually significant ones, however, sequential surgery can add significant morbidity and recovery time.
- Pterygia that interfere with K readings and those that cover the visual axis should be removed before proceeding with cataract surgery. Further, as a rule, any pterygium of 3 mm or greater should be excised before cataract surgery.
Three Cases of Sequential Surgery
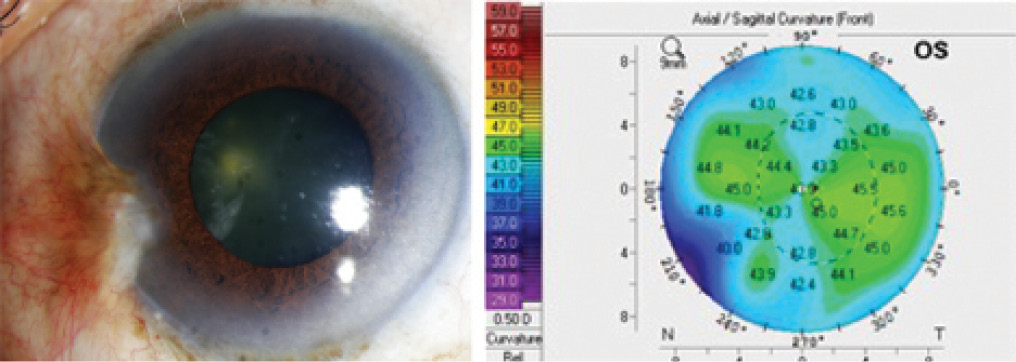
Figure 1. Case No. 1: Slit-lamp photo of regressing pterygium (A). Topography on Pentacam (Oculus Optikgeräte) shows regular astigmatism not induced by the pterygium, which is causing only peripheral changes in the topography (B). This patient can undergo cataract surgery without any prior surgical intervention for the pterygium.
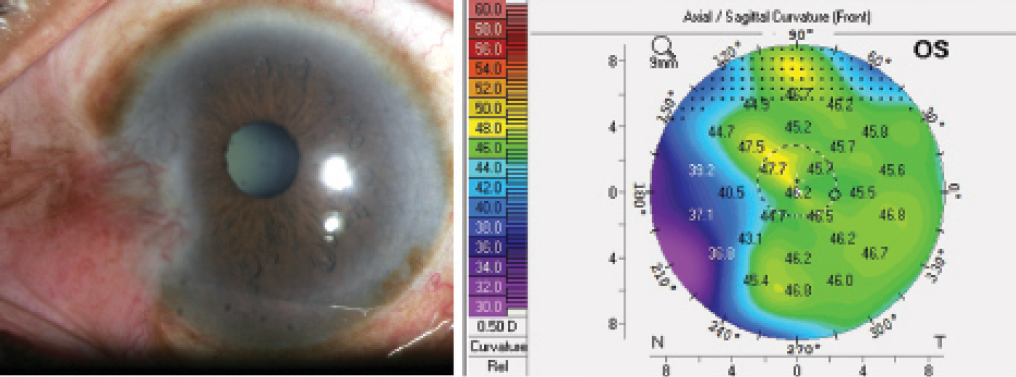
Figure 2. Case No. 2: Slit-lamp photo of a large, fleshy pterygium (A). Although it is difficult to detect the extension of the pterygium into the central cornea on the slit-lamp photo, the Pentacam shows irregular astigmatism of the central cornea induced by the pterygium (B). This patient would benefit from pterygium excision before cataract surgery.
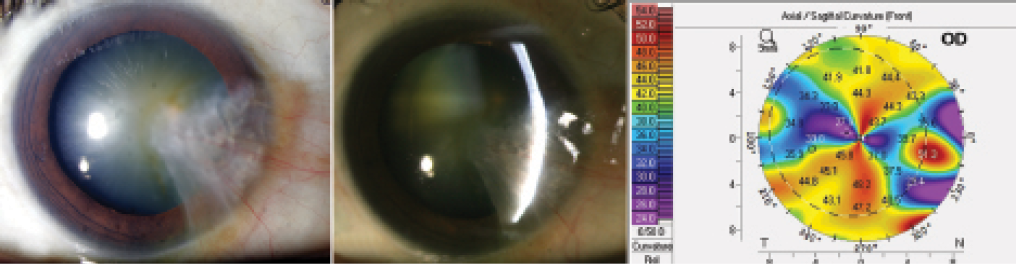
Figure 3. Case No. 3: Slit-lamp photos of an extensive pterygium covering the visual axis (A, B). Topography shows irregular mires and is not reliable (C). Topography is not required in this patient at this time, as pterygium excision is mandatory before cataract surgery.

Figure 4. Online toric IOL calculators from Alcon (A), Abbott Medical Optics (B), and Carl Zeiss Meditec (C); Hoya Toric Calculator on the iTrace aberrometer (D).
The corneal astigmatism induced by pterygium is often reversible on excision, unless it has caused corneal scarring, as is seen in atrophic ones of long standing. Large pterygia covering the visual axis (Figure 3) must be removed before the cataract surgery. It is impossible obtain a good corneal topography image before excision of these pterygia.
Present-day pterygium surgery involves excision of the pterygium followed by gluing of a conjunctival autograft over the bare sclera. Mitomycin C 0.02% is reserved almost exclusively for use with recurrent pterygia. Use of mitomycin C and glued conjunctival autografts may affect outcomes when cataract surgery is combined with pterygium excision. Thus, it may be better to perform sequential surgery and allow corneal healing to complete before scheduling cataract surgery.
As a rule of thumb, any pterygium of 3 mm or greater should be excised before cataract surgery. Patients with irregular mires on keratometry or irregular central curvature or high corneal astigmatism on topography are candidates for primary excision of the pterygium. Patients with small pterygia at the limbus with regular mires on keratometry, regular central curvature on topography, and low or absent astigmatism can have cataract surgery before pterygium excision.
Following pterygium excision, one should wait 6 to 8 weeks for corneal curvature to stabilize before assessing the patient for cataract surgery. Keratometry or topography should then be repeated 2 weeks later to ensure the stability of the cornea before these measurements are used for biometry.
TECHNIQUE AFTER PTERYGIUM EXCISION
Limbal stem cell deficiency has been implicated as a major cause of pterygium.7,8 Pterygium surgery itself may induce a further loss of stem cells.9 Hence, it is advisable to use a clear corneal cataract incision and opt for the smallest possible wound size, as with microcoaxial phacoemulsification. Any surgery that involves extensive dissection of the conjunctiva, such as extracapsular cataract extraction or manual small-incision cataract surgery, is best avoided in these patients.
In patients with scarred conjunctiva following excision of large pterygia, the quadrant opposite the pterygium should be the site of the phaco tunnel. Thus, a temporal approach is preferable in a nasal pterygium, as this would place the wound the furthest away from the site of the pterygium. In patients with corneal thinning and obvious flattening of the horizontal meridian, phacoemulsification can be performed through a superior tunnel to induce some flattening of the vertical meridian, negating to some extent the with-the-rule astigmatism. In patients who have previously undergone removal of a two-headed pterygium, the only approach for phacoemulsification would be superior or oblique.
Visibility through a scarred cornea during cataract surgery is poor. Staining the capsule with trypan blue dye can aid in visualizing the rhexis margin during surgery (eyetube.net/?v=ninif).
IOL CHOICE
The choice of IOL depends primarily on the presence or absence of corneal astigmatism. Keratometry or topography should be repeated 6 weeks after pterygium excision and preferably repeated 2 weeks later to determine whether the astigmatism is still changing. PTK can benefit patients with central superficial corneal opacities,10 and topography-guided PRK can help to regularize an irregular cornea.11
Watch it Now
Staining the capsule with trypan blue dye can aid in visualizing the capsulorrhexis margin during surgery.
In patients without significant corneal astigmatism or demonstrable higher-order aberrations (HOAs), any IOL of one's preference can be used. Patients who had large, scarred pterygia may be left with significant irregular astigmatism; toric IOLs are not of much use in these patients, but contact lenses may help to regularize the front refracting surface of the eye.
Toric IOLs can be used for regular corneal astigmatism of more than 1.00 D. Online toric IOL calculators (Figures 4A through 4C) or offline calculators using the built-in software in aberrometers such as the iTrace (Tracey Technologies; Figure 4D) can aid in choosing the appropriate toric IOL and the correct axis of placement. Unlike online calculators, software solutions allow a greater degree of manipulation of toric IOL models and can be used to check the effect on residual astigmatism. Multifocal IOLs are not a good option in patients with irregular astigmatism or high levels of HOAs or in any patient with high degrees of corneal aberration.
CONCLUSION
Simultaneous pterygium and cataract surgery is necessary only in patients who cannot easily access eye care facilities. In all other cases, sequential surgery may be preferred. Pterygium surgery should be sequenced before cataract surgery when the pterygium is large enough to induce corneal curvature changes. This order will significantly reduce the incidence of refractive surprise after cataract surgery. Small pterygia near the limbus, especially those that are atrophic, can be left alone, and one can proceed with cataract surgery only. Topography may detect subclinical extensions of the pterygium into the center of the cornea. n
1. Liu L, Wu J, Geng J, Yuan Z, Huang D. Geographical prevalence and risk factors for pterygium: a systematic review and meta-analysis. BMJ Open. 2013;3(11):e003787.
2. Foster A. Vision 2020: The Cataract Challenge. Community Eye Health. 2000; 13(34):17-19.
3. Saw SM, Tan D. Pterygium: prevalence, demography and risk factors. Ophthalmic Epidemiol. 1999;6(3):219-228.
4. Threlfall TJ, English DR. Sun exposure and pterygium of the eye: a dose-response curve. Am J Ophthalmol. 1999;128(3):280-287.
5. Chatterjee A, Milton RC, Tyles S. Aetiology of senile cataract. Br J Ophthal. 1982;66:35.
6. Maheshwari S. Effect of pterygium excision on pterygium induced astigmatism. Indian J Ophthalmol. 2003;51:187-188.
7. Kwok LS, Coroneo MT. A model for pterygium formation. Cornea. 1994;13(3):219-224.
8. Dushku N, Reid TW. Immunohistochemical evidence that human pterygia originate from an invasion of vimentin-expressing altered limbal epithelial basal cells. Curr Eye Res. 1994;13(7):473-481.
9. Sridhar MS, Vemuganti GK, Bansal AK, Rao GN. Impression cytology-proven corneal stem cell deficiency in patients after surgeries involving the limbus. Cornea. 2001;20(2):145-148.
10. Sher NA, Bowers RA, Zabel RW, et al. Clinical use of the 193-nm excimer laser in the treatment of corneal scars. Arch Ophthalmol. 1991;109(4):491-498.
Sudeep Das, MBBS, DO, DNB
- Senior Consultant, Cataract and Refractive Surgery, Narayana Nethralaya, Rajajinagar, Bangalore, India
- drsudeepdas@gmail.com
- Financial disclosure: None
Nikhil Negalur, MBBS, MS
- Fellow, Cataract and Refractive Surgery, Narayana Nethralaya, Rajajinagar, Bangalore, India
- Financial disclosure: None

