

Modern ophthalmology has seen a number of paradigm shifts in the treatment of corneal disease, but challenges remain. One of these is infectious keratitis.
Microbial keratitis, whether of bacterial or fungal origin, is a leading global cause of blindness, with an estimated several million new cases every year. The World Health Organization classified microbial keratitis as a silent epidemic in the 1990s, based on the alarming number of infections occurring on the Indian subcontinent.1-4 A broken epithelial barrier is usually required for microorganisms to penetrate into the corneal stroma and cause a corneal infection. The nature of the causative traumas differs by region: Insufficient hygiene and reduced compliance in contact lens wear are the main reasons in developed countries, whereas minor corneal trauma, combined with lack of funding and little to no access to ophthalmic care, is the main reason in emerging countries.5-9 (See A Tale of Two Scenarios for an example of each scenario.)
• PACK-CXL might represent a valuable alternative to conventional antimicrobial therapy in varying regions and geographic locations.
• Advantages of PACK-CXL over conventional treatments include independence from antibiotic resistance, freedom from the so-called diagnostic dilemma, and reduction of treatment-related costs.
• Currently, the beneficial effect of PACK-CXL is limited to treatment of bacterial, fungal, and mixed infections.
The nature of infectious keratitis largely depends on geographic location. Countries with cool climates report almost exclusively bacterial infections, whereas in subtropical countries mixed (bacterial/fungal) infections prevail, making therapy more difficult. Additionally, in emerging countries, the limiting factor is usually not the purchase price of therapy, which is often inexpensive and sold over the counter, but rather the doctor’s honorary fees, as keratitis often requires multiple follow-up visits. Not only are these visits expensive, but the loss of wages is unbearable for the individual and the individual’s family in emerging countries.10,11
A new treatment, called photoactivated chromophore for infectious keratitis CXL (PACK-CXL),12 might represent a valuable alternative to conventional antimicrobial therapy in varying regions and geographic locations.
CXL PRINCIPLES AND INITIAL USES
The first ophthalmic application of CXL in humans occurred in 1999, when a phase 1 clinical study was performed in Dresden, Germany.13 The technique, subsequently dubbed the Dresden protocol, consisted of applying the chromophore riboflavin to the cornea and using UV-A light to induce the formation of crosslinks between the side chains of the collagen fibers and proteoglycans in the extracellular matrix.14 The high molecular weight of riboflavin required manual removal of the corneal epithelium to allow the riboflavin to penetrate the corneal stroma.
Since 1999, CXL has become a gold standard of treatment of keratoconus and other corneal ectatic diseases in more than 120 countries.
NEW INDICATION: INFECTIOUS KERATITIS
Photoactivation of riboflavin has been used in the chemical industry for many decades to reduce the microbial load in liquids. The same technology is also used to reduce the microbial load in fresh-frozen plasma for blood transfusions, killing both bacteria and viruses.15-18
In 2003, the Swiss Federal Institute of Technology in Zurich (ETHZ) developed solar disinfection (SODIS), a technique in which riboflavin is added to contaminated water in polyethylene terephthalate bottles, and then the bottles are exposed to intense natural sunlight. After 8 hours of direct exposure, the resulting photochemical reaction kills bacteria, fungi, viruses, and even Acanthamoeba cysts; the water is then drinkable.19
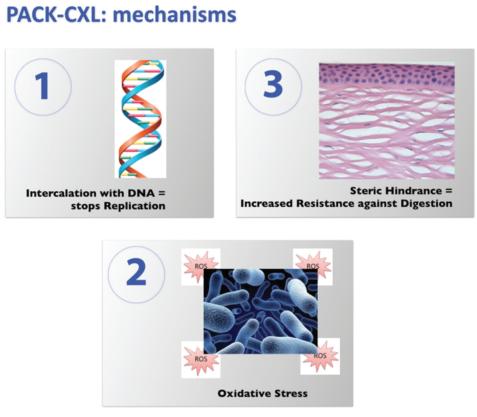
Figure 1. Presumed mechanisms of PACK-CXL action: (1) intercalation with DNA, which stops replication, (2) oxidative stress, and (3) steric hindrance, which increases resistance against digestion.
In 2006 and 2007, we tested the potential antimicrobial effect of CXL in corneal infections by treating five eyes of five patients with severe, therapy-resistant infectious keratitis. Our institutional review board allowed treatment only after conventional therapy had been tried for at least 4 weeks without positive effect. Four of the five eyes showed rapid improvement after CXL. The fifth required several additional weeks of conventional treatment.20
In light of these encouraging results, Makdoumi and collagues performed a nonrandomized clinical study in 2009-2010 to analyze the effects of CXL as a first-line therapy against infectious keratitis. In this setting, 16 eyes of 16 patients were treated without previous topical or systemic antimicrobial therapy. As in our pilot study, the technical parameters used were adapted from the Dresden protocol for keratoconus (3 mW/cm2 for 30 minutes). This study confirmed our hypothesis, and 14 of the 16 eyes required no further conventional antimicrobial therapy after CXL and showed complete reepithelialization within days. Two eyes required further antibiotic therapy.21-23
The study by Makdoumi and colleagues showed that PACK-CXL might be used not only as an adjuvant therapy but potentially as a first-line treatment for infectious keratitis. Advantages of PACK-CXL over conventional treatments include independence from antibiotic resistance, freedom from confirming the bacterial or fungal nature of the pathogen(s) causing infection (the so-called diagnostic dilemma), and simultaneous effectiveness against both methicillin-resistant and methicillin-sensitive bacteria and fungi.
HOW DOES IT WORK?
The antimicrobial effect of PACK-CXL is the direct result of an interaction between the energy delivered by the UV-A light and the riboflavin molecule.
We have learned that, besides biomechanical stiffening, a number of additional processes occur simultaneously during every CXL procedure. In addition to the photoactivated riboflavin irreversibly binding to the DNA of any microorganisms present in the cornea, stopping replication, crosslinking induces conformational changes in the 3-D structure of the cornea, thereby leading to an increased resistance against enzymatic digestion via steric hindrance. This minimizes tissue damage and the size of the resulting scar. Furthermore, high levels of oxidative stress disrupt the cellular membranes of any microorganisms present in the cornea (Figure 1).24
GLOBAL BENEFITS AND FUTURE OUTLOOK
The potential benefits of the PACK-CXL technique on a global level could be manifold.
• Potential benefit No. 1: The treating ophthalmologist would have an additional treatment modality in infectious keratitis;
• Potential benefit No. 2: The use of antimicrobial agents could be reduced;
• Potential benefit No. 3: The patient would save time and money;
• Potential benefit No. 4: The treatment is independent of antimicrobial resistance of bacteria; and
• Potential benefit No. 5: The treatment is less dependent on the accuracy of clinical assessment by the ophthalmologist and on patient compliance (Figure 2).
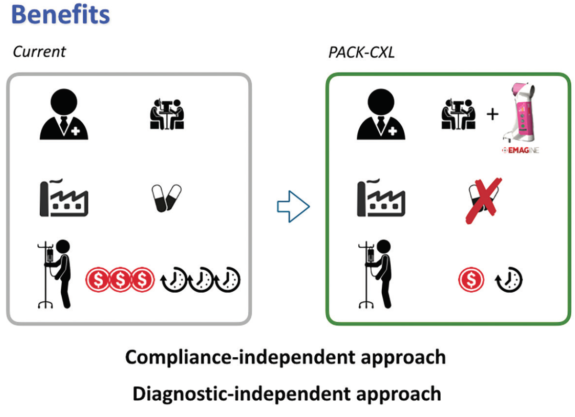
Figure 2. Potential benefits of PACK-CXL.
The epithelium-off Dresden protocol was originally developed for keratoconus, and it has demonstrated impressive efficacy and safety in long-term results, along with a low complication rate.25 However, it is time-consuming and it requires major infrastructure in the form of an operating room. If PACK-CXL is to become a global player in the treatment of bacterial keratitis, the technique must be simplified and accelerated.
A Tale of Two Scenarios
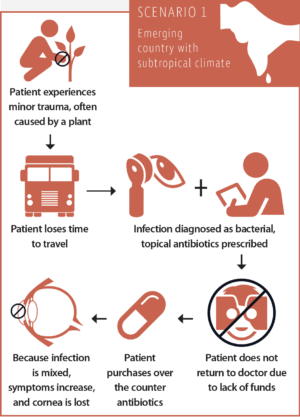
Scenario No. 1: The typical course of a corneal infection in an emerging country with a subtropical climate
After a minor trauma, often caused by a plant, the patient loses time by having to travel a great distance to see an ophthalmologist. The infection is diagnosed as bacterial, and the patient is advised to take topical antibiotics and revisit the doctor. The patient will take the antibiotics, but, due to lack of funds, he will not go back to see the doctor. The infection is not only bacterial but mixed (bacterial-fungal). Because the symptoms increase despite the antibiotics, the patient purchases steroids over the counter. The condition exacerbates, and the cornea is lost.
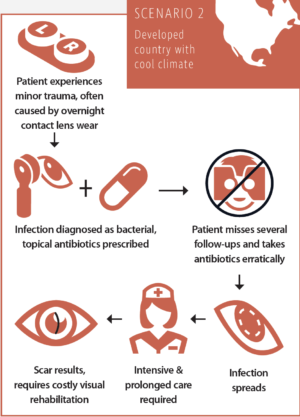
Scenario No. 2: The typical course of a corneal infection in a developed country with a cool climate
After a minor trauma, usually caused by excessive overnight contact lens wear, the patient sees an ophthalmologist. The infection is diagnosed as bacterial, and the patient is advised to take topical antibiotics and see the doctor for several follow-up visits. Due to a lack of compliance, the patient takes the antibiotics erratically and misses several follow-ups. The infection spreads and requires intensive and prolonged care, possibly even hospitalization. The resulting scar reduces CDVA and requires additional costly procedures at a later stage for visual rehabilitation.
Our research group has managed to reduce irradiation time by a factor of 10—from 30 to 3 minutes—and maintain the bacterial killing rate. Specifically, this reduction of treatment time opens alleys to kill bacteria, even methicillin-resistant Staphylococcus aureus, within only 3 to 5 minutes. This time span is short enough to allow a CXL procedure to be performed comfortably at the slit lamp.26
Currently, the beneficial effect of PACK-CXL is limited to treatment of bacterial, fungal, and mixed infections. SODIS, as noted previously, is capable of killing viruses and Acanthamoeba cysts as well. However, the total energy doses used in SODIS are significantly higher than the fluence used clinically for CXL in the cornea.19
Future research projects will aim to optimize irradiation doses and times for this new indication. Simultaneously, several randomized prospective clinical studies have been undertaken to investigate the efficacy of PACK-CXL in the treatment of infectious keratitis.
1. Whitcher JP, Srinivasan M. Corneal ulceration in the developing world--a silent epidemic. Br J Ophthalmol. 1997;81(8):622-623.
2. Srinivasan M, Gonzales CA, George C, et al. Epidemiology and aetiological diagnosis of corneal ulceration in Madurai, south India. Br J Ophthalmol. 1997;81(11):965-971.
3. Gonzales CA, Srinivasan M, Whitcher JP, Smolin G. Incidence of corneal ulceration in Madurai district, South India. Ophthalmic Epidemiol. 1996;3(3):159-166.
4. Upadhyay MP, Karmacharya PC, Koirala S, et al. Epidemiologic characteristics, predisposing factors, and etiologic diagnosis of corneal ulceration in Nepal. Am J Ophthalmol. 1991;111(1):92-99.
5. Carnt N, Willcox MD, Keay L, Flanagan J, Stapleton F. Pathogenesis of contact lens-associated microbial keratitis. Optom Vis Sci. 2010;87(8):612-613; author reply 613-614.
6. Dart JK, Radford CF, Minassian D, Verma S, Stapleton F. Risk factors for microbial keratitis with contemporary contact lenses: a case-control study. Ophthalmology. 2008;115(10):1647-1654, e1641-1643.
7. Stapleton F, Keay LJ, Sanfilippo PG, Katiyar S, Edwards KP, Naduvilath T. Relationship between climate, disease severity, and causative organism for contact lens-associated microbial keratitis in Australia. Am J Ophthalmol. 2007;144(5):690-698.
8. Keay L, Stapleton F, Schein O. Epidemiology of contact lens-related inflammation and microbial keratitis: a 20-year perspective. Eye Contact Lens. 2007;33(6 Pt 2):346-353, discussion 362-343.
9. Keay L, Edwards K, Naduvilath T, Taylor HR, Snibson GR, Forde K, Stapleton F. Microbial keratitis predisposing factors and morbidity. Ophthalmology. 2006;113(1):109-116.
10. Collier SA, Gronostaj MP, MacGurn AK, et al. Estimated burden of keratitis — United States, 2010. MMWR Morb Mort Wkly Rep. 2014;63(45):1027-1030.
11. Keay L, Edwards K, Brian G, Stapleton F. Surveillance of contact lens related microbial keratitis in Australia and New Zealand: multi-source case-capture and cost-effectiveness. Ophthalmic Epidemiol. 2007;14(6):343-350.
12. Hafezi F, Randleman JB. PACK-CXL: defining CXL for infectious keratitis. J Refract Surg. 2014;30(7):438-439.
13. Wollensak G, Spoerl E, Seiler T. Riboflavin/ultraviolet-A-induced collagen crosslinking for the treatment of keratoconus. Am J Ophthalmol. 2003;135(5):620-627.
14. Zhang Y, Conrad AH, Conrad GW. Effects of ultraviolet-A and riboflavin on the interaction of collagen and proteoglycans during corneal cross-linking. J Biol Chem. 2011;286(15):13011-13022.
15. Goodrich RP. The use of riboflavin for the inactivation of pathogens in blood products. Vox Sang. 2000;78 Suppl 2:211-215.
16. Corbin F 3rd. Pathogen inactivation of blood components: current status and introduction of an approach using riboflavin as a photosensitizer. Int J Hematol. 2002;76 Suppl 2:253-257.
17. Goodrich RP, Edrich RA, Li J, Seghatchian J. The Mirasol PRT system for pathogen reduction of platelets and plasma: an overview of current status and future trends. Transfus Apher Sci. 2006;35(1):5-17.
18. Grellier P, Benach J, Labaied M, et al. Photochemical inactivation with amotosalen and long-wavelength ultraviolet light of Plasmodium and Babesia in platelet and plasma components. Transfusion. 2008;48(8):1676-1684.
19. Heaselgrave W, Kilvington S. Antimicrobial activity of simulated solar disinfection against bacterial, fungal, and protozoan pathogens and its enhancement by riboflavin. Appl Environ Microbiol. 2010;76(17):6010-6012.
20. Iseli HP, Thiel MA, Hafezi F, Kampmeier J, Seiler T. Ultraviolet A/riboflavin corneal cross-linking for infectious keratitis associated with corneal melts. Cornea. 2008;27(5):590-594.
21. Makdoumi K, Backman A, Mortensen J, Crafoord S. Evaluation of antibacterial efficacy of photo-activated riboflavin using ultraviolet light (UVA). Graefes Arch Clin Exp Ophthalmol. 2010;248(2):207-212.
22. Makdoumi K, Mortensen J, Crafoord S. Infectious keratitis treated with corneal crosslinking. Cornea. 2010;29(12):1353-1358.
23. Makdoumi K. Ultraviolet light A (UVA) photoactivation of riboflavin as a potential therapy for infectious keratitis. 2011. Örebro University. https://www.diva-portal.org/smash/get/diva2:450387/FULLTEXT02.pdf. Accessed March 2, 2017.
24. Randleman JB, Khandelwal SS, Hafezi F. Corneal cross-linking. Surv Ophthalmol. 2015;60(6):509-523.
25. Koller T, Mrochen M, Seiler T. Complication and failure rates after corneal crosslinking. J Cataract Refract Surg. 2009;35(8):1358-1362.
26. Richoz O, Kling S, Hoogewoud F, et al. Antibacterial efficacy of accelerated photoactivated chromophore for keratitis-corneal collagen cross-linking (PACK-CXL). J Refract Surg. 2014;30(12):850-854.



