

The ocular surface plays a leading role in corneal refractive surgery. In fact, the recent seminal publication by the Tear Film & Ocular Surface Society (TFOS) of the results of the Dry Eye Workshop II (DEWS II) acknowledged the importance of paying attention to tear dysfunction and dry eye disease (DED) in patients undergoing refractive surgery.1 The revised global definition of DED and an overview of the DEWS II study are discussed in Dry Eye Disease Redefined .
AT A GLANCE
- A healthy corneal surface and tear film are essential for good, stable vision and a healthy wound-healing response after corneal refractive surgery.
- Despite technological advances, the induction or aggravation of DED and neurotrophic epitheliopathy are the most common complications after refractive surgery.
- Proactive identification and treatment of DED preoperatively and selection of the best surgical procedure for each patient should be considered optimal approaches.
Interestingly, a new subcommittee on iatrogenic DED was included in the DEWS II, with the aim of reviewing the most important mechanisms and providing an evidence-based review of the causes and effects of iatrogenic DED.2 The panel identified systemic drugs and topical drugs containing toxic preservatives such as benzalkonium chloride as important sources of iatrogenic DED, but it also noted that DED can be induced and aggravated by refractive corneal surgery, mainly due to neurosensory abnormalities.2
A healthy corneal surface and tear film are essential for good, stable vision and a healthy wound-healing response after corneal refractive surgery.3 Even the most sophisticated surgical techniques still depend on a stable tear film to provide good vision. Tear dysfunction and DED can be challenging and potentially serious problems after laser vision correction procedures.2,4,5 Thus, it is essential that refractive surgeons preoperatively identify patients at risk.4 A proactive detection strategy would allow practitioners to properly educate patients about DED and to prescribe measures to optimize the ocular surface before and after surgery.4,6
SIGNS AND SYMPTOMS
The prevalence of preexisting DED identified among laser vision correction candidates may vary depending on the diagnostic criteria used and on other factors such as age.4 Notably, contact lens intolerance—an early sign of DED—is a common reason that patients pursue laser vision correction, and it should be considered a major risk factor for DED after refractive surgery. Diagnosis of DED requires a thorough assessment of the patient’s ocular history and a careful ophthalmic examination, including vital staining. It is well known that the signs and symptoms of DED are poorly correlated, so patients may not themselves be aware that they have DED.
Of the traditional signs of DED, staining is the most significant. Classic fluorescein staining is often used, along with lissamine green and rose bengal.7 We have described the novel use of trypan blue for corneal staining (Figure 1),8 and we found that it has staining patterns similar to those of lissamine green and rose bengal.9
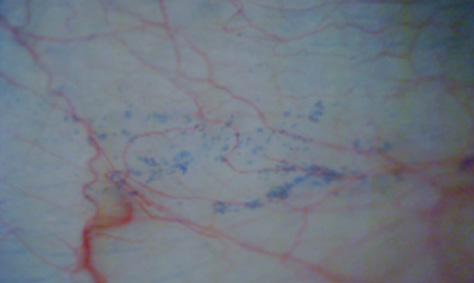
Figure 1. Slit-lamp photo of the conjunctiva stained with trypan blue dye in a patient presenting for LASIK due to contact lens intolerance. Along with a TBUT of 9 mm, this was the only clinical sign of DED detected.
Traditional tests such as Schirmer testing are of limited applicability outside of late-stage DED due to their limited sensitivity and specificity.4,10 Tear film stability, as measured by tear breakup time (TBUT), is commonly affected by ocular surface procedures and the use of postoperative medications. Recently developed point-of-care tests to assess tear health have improved sign-symptom correlation coefficients.
Alternatively, time-saving, noninvasive methods for measuring tear film quality can be used. Advanced Placido-disc–based imaging technologies such as the Keratograph 5M (Oculus) can evaluate the tear film with the aid of white or infrared illumination in a noninvasive fashion. This modality provides objective, repeatable, noninvasive measurements of the TBUT, tear meniscus height, lipid layer, and tear-film particle movement, all of which are useful in the diagnosis and monitoring of DED (Figure 2).
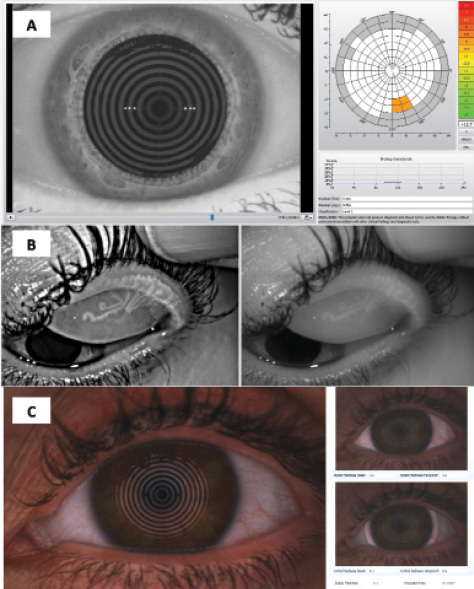
Figure 2. Keratograph 5M exam of a patient with evaporative DED. The scans revealed a reduced TBUT of approximately 9 seconds (A), generalized meibomian gland atrophy (B), and moderate redness of the ocular surface (C).
Tear osmolarity assessment is a sensitive and specific diagnostic test for dry eye. Scientific evidence suggests that tear hyperosmolarity is a primary mechanism in the initiation of DED.11-13 Interestingly, Lee et al found an increase in tear film osmolarity of approximately 35% in patients
6 months after LASIK.14
Matrix metalloproteinase-9 (MMP-9) is an enzyme that has multiple functions including remodeling and maintaining the protective epithelial cell tight junction barrier function and up-regulating quickly in response to injury. MMP-9 levels are elevated in chronic DED via a positive feedback loop with inflammation. MMP-9 is a nonspecific inflammatory marker implicated in a variety of immunologic processes including but not limited to DED. The MMP-9 analysis test InflammaDry (Rapid Pathogen Screening) is an affordable, efficient point-of-care diagnostic test that can help increase confidence in preoperative corneal topography readings and guide treatment strategies.15,16
DED AFTER LASER VISION CORRECTION
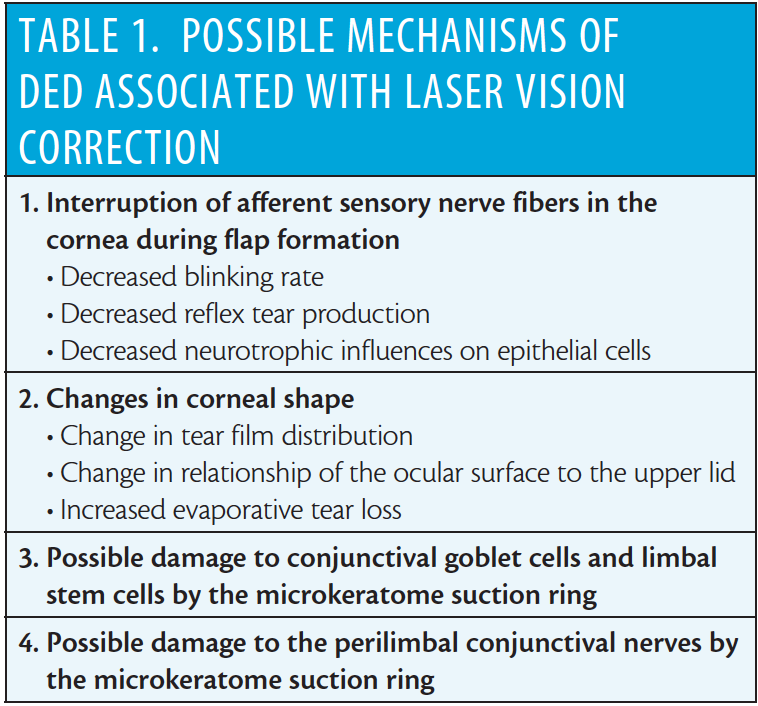
The possible mechanisms of DED associated with laser vision correction are listed in Table 1. During refractive surgery, the corneal nerves are transected (in LASIK) or pruned (in PRK), resulting in a loss of the crucial homeostasis-maintaining feedback loop of the lacrimal functional unit.17 The term LASIK-induced neurotrophic epitheliopathy (LNE) was coined by Steven E. Wilson, MD.18,19 The diagnosis of LNE is based on an overall constellation of symptoms and signs. There are a number of symptoms of ocular irritation that patients may experience, including ocular fatigue, burning, itching, foreign body sensation, fluctuating vision, dryness, soreness, heaviness of the lids, and photophobia. Exacerbation of an ocular surface condition typically improves as corneal reinnervation takes place, returning to preoperative baseline by about 9 months after LASIK.4-6
The method of LASIK flap creation can make a difference. In one study, the incidence of LNE and the need for aggressive postoperative treatments such as the use of cyclosporine ophthalmic emulsion 0.05% (Restasis; Allergan) was significantly lower in eyes that had femtosecond flaps compared with eyes that had microkeratome flaps.5 Small-incision lenticule extraction (SMILE) also reduces the neurotrophic impact on the cornea,20,21 decreasing the incidence of DED and possibly the resulting degradation in quality of life after refractive surgery.22
BEST STRATEGY IS PREVENTION
In current practice, the best strategy for managing complications is preventing them; we consider the health of the ocular surface to be a primary factor for the selection of the ideal laser vision correction procedure for each patient.
Once DED is diagnosed, patient education plays a major role. It is important to discuss the condition, the pathogenesis, the anticipated clinical course, and the treatment options with the patient. This applies to laser vision correction candidates with DED prior to surgery and to patients with DED after surgery. Surgeons should inform patients that the cornea will be hypesthetic for several months postoperative and will improve with healing; this may be reassuring for some patients who experience fluctuating vision.
Preservative-free artificial tears, nutrition supplementation with omega-3 essential fatty acids, punctal occlusion, and topical cyclosporine are among the most common approaches to ocular surface optimization, along with treatment of meibomian gland dysfunction. These steps can be taken preoperatively to improve a suboptimal ocular surface or used postoperatively in the event DED develops. Other postoperative therapeutic approaches may include protection of the cornea with a bandage soft contact lens, healing promotion with lacrimal-fluid surrogates such as 20% autologous serum (delivering epidermal growth factor and transforming growth factor beta 1), and tissue regeneration with self-retaining cryopreserved amniotic membrane.4,23-25
Corneal neuralgia with moderate to severe and debilitating symptoms of light sensitivity and difficulty with visual activities have been reported after LASIK or LASIK enhancement.26 These patients often have relatively good visual acuity and normal tear film measurements, but in vivo confocal microscopy demonstrates abnormal corneal nerves. Such cases typically show poor responses to standard DED treatments but may respond to more aggressive treatments such as use of autologous serum tears; prescription of a prosthetic device such as the Prosthetic Replacement of the Ocular Surface Ecosystem (PROSE; BostonSight); or use of systemic agents for pain, anxiety, or depression. Corneal neuralgia of this type should be considered a different condition from LNE, but distinguishing it from typical post-LASIK DED will remain a challenge until a better understanding of its risk factors and a proper prevention approach are established.26
CONCLUSION
Despite many technological advances in refractive surgery, the induction or aggravation of DED and neurotrophic epitheliopathy are still its most common postoperative complications. Proactive identification and treatment of DED preoperatively and selection of the best surgical procedure for each patient should be considered the optimal approaches.
1. Craig JP, Nichols KK, Akpek EK, et al. TFOS DEWS II definition and classification report. Ocul Surf. 2017;15:276-283.
2. Gomes JAP, Azar DT, Baudouin C, et al. TFOS DEWS II iatrogenic report. Ocul Surf. 2017;15:511-538.
3. Sweeney DF, Millar TJ, Raju SR. Tear film stability: a review. Exp Eye Res. 2013;117:28-38.
4. Ambrosio Jr R, Tervo T, Wilson SE. LASIK-associated dry eye and neurotrophic epitheliopathy: pathophysiology and strategies for prevention and treatment. J Refract Surg. 2008;24:396-407.
5. Salomao MQ, Ambrosio Jr R, Wilson SE. Dry eye associated with laser in situ keratomileusis: Mechanical microkeratome versus femtosecond laser. J Cataract Refract Surg. 2009;35:1756-1760.
6. Ambrosio Jr R, Wilson SE. Complications of laser in situ keratomileusis: etiology, prevention, and treatment. J Refract Surg. 2001;17:350-379.
7. Machado LM, Castro RS, Fontes BM. Staining patterns in dry eye syndrome: rose bengal versus lissamine green. Cornea. 2009;28:732-734.
8. Ambrósio Jr R, Ahmad H, Caldas D, et al. Novel use of trypan blue in ocular surface staining: redefining implications for this vital dye. Revista Brasileira de Oftalmologia. 2011;70:408-410.
9. Castro RS. Staining patterns in dry eye syndrome: rose bengal versus tripan blue. Invest Ophthalmol Vis Sci. 2012;53:545.
10. Wolffsohn JS, Arita R, Chalmers R, et al. TFOS DEWS II diagnostic methodology report. Ocul Surf. 2017;15:539-574.
11. Versura P, Campos EC. TearLab osmolarity system for diagnosing dry eye. Expert Rev Mol Diagn. 2013;13:119-129.
12. Potvin R, Makari S, Rapuano CJ. Tear film osmolarity and dry eye disease: a review of the literature. Clin Ophthalmol. 2015;9:2039-2047.
13. Versura P, Profazio V, Campos EC. Performance of tear osmolarity compared to previous diagnostic tests for dry eye diseases. Curr Eye Res. 2010;35:553-564.
14. Lee JB, Ryu CH, Kim J, Kim EK, Kim HB. Comparison of tear secretion and tear film instability after photorefractive keratectomy and laser in situ keratomileusis. J Cataract Refract Surg. 2000;26:1326-1331.
15. Pahuja N, Kumar NR, Francis M, et al. Correlation of clinical and biomechanical outcomes of accelerated crosslinking (9 mW/cm2 in 10 minutes) in keratoconus with molecular expression of ectasia-related genes. Curr Eye Res. 2016;41:1419-1423.
16. Shetty R, Ghosh A, Lim RR, et al. Elevated expression of matrix metalloproteinase-9 and inflammatory cytokines in keratoconus patients is inhibited by cyclosporine A. Invest Ophthalmol Vis Sci. 2015;56:738-750.
17. Chao C, Golebiowski B, Stapleton F. The role of corneal innervation in LASIK-induced neuropathic dry eye. Ocul Surf. 2014;12:32-45.
18. Wilson SE. Laser in situ keratomileusis-induced (presumed) neurotrophic epitheliopathy. Ophthalmology. 2001;108:1082-1087.
19. Wilson SE, Ambrosio R. Laser in situ keratomileusis-induced neurotrophic epitheliopathy. Am J Ophthalmol. 2001;132:405-406.
20. Reinstein DZ, Archer TJ, Gobbe M. Small incision lenticule extraction (SMILE) history, fundamentals of a new refractive surgery technique and clinical outcomes. Eye Vis (Lond). 2014;1:3.
21. Reinstein DZ, Archer TJ, Gobbe M, Bartoli E. Corneal sensitivity after small-incision lenticule extraction and laser in situ keratomileusis. J Cataract Refract Surg. 2015;41:1580-1587.
22. Denoyer A, Landman E, Trinh L, Faure JF, Auclin F, Baudouin C. Dry eye disease after refractive surgery: comparative outcomes of small incision lenticule extraction versus LASIK. Ophthalmology. 2015;122:669-676.
23. Torricelli AA, Santhiago MR, Wilson SE. Topical cyclosporine a treatment in corneal refractive surgery and patients with dry eye. J Refract Surg. 2014;30:558-564.
24. Lee HS, Jang JY, Lee SH, Im SK, Yoon KC. Clinical effectiveness of topical cyclosporine a 0.05% after laser epithelial keratomileusis. Cornea. 2013;32:e150-e155.
25. Jones L, Downie LE, Korb D, et al. TFOS DEWS II management and therapy report. Ocul Surf. 2017;15:575-628.
26. Theophanous C, Jacobs DS, Hamrah P. Corneal neuralgia after LASIK. Optom Vis Sci. 2015;92:e233-e240.



