

Pre-Descemet endothelial keratoplasty (PDEK),1 the latest development in the area of endothelial keratoplasty, has evolved based on the theory and description of the pre-Descemet layer (PDL), or Dua layer,2 originally defined by Harminder Dua, MBBS, DO, MS, MNAMS, FRCS, FRCOphth, FEBO, MD, PhD, of the University of Nottingham, United Kingdom (see The Dua Layer). Since we started performing PDEK in 2013, this technique has undergone multiple iterations, including improvements in the method of graft preparation, donor lenticule insertion, graft visualization, and adhesion.
In this article, we review how to create and insert the PDEK graft and outline the benefits of the procedure compared with Descemet membrane endothelial keratoplasty (DMEK).
AT A GLANCE
- The PDEK procedure allows the use of young and infant donor corneas, and the graft does not curl and scroll because of the splinting effect of the PDL.
- In aphakic eyes or eyes with malpositioned IOLs, PDEK with the glued IOL technique can help to optimize the patient’s visual potential.
- The double infusion cannula technique helps prevent globe collapse and facilitates graft adherence in the combined PDEK and glued IOL procedure.
GRAFT CREATION AND INSERTION
To create the graft for PDEK (Figure 1), we take a 30-gauge needle attached to a 5-mL air-filled syringe and insert it from the edge of the corneoscleral rim up to the midperiphery of the corneoscleral rim of a donor cornea lenticule with the endothelial side up. Air is then slowly injected into the donor lenticule, and a type 1 big bubble is created, which characteristically spreads from center to midperiphery. The bubble is dome-shaped and approximately 7 to 8 mm in diameter.
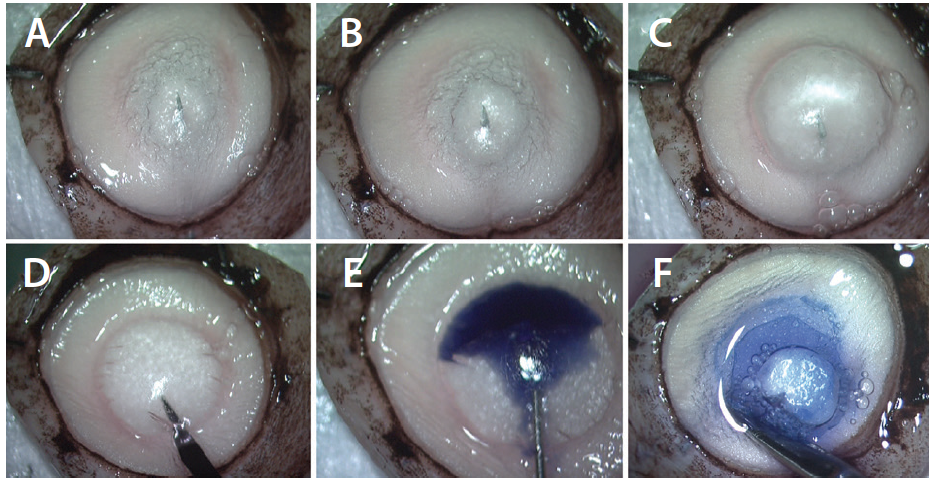
Figure 1. PDEK graft preparation. A 30-gauge needle attached to a 5-mL air-filled syringe is introduced from the corneoscleral rim to the corneal midperiphery, and air is slowly injected. Small bubbles are initially formed (A). A type 1 big bubble starts to form from the center, extending to the midperiphery (B), and then a type 1 big bubble with characteristic domeshaped appearance is seen (C). The extreme periphery of the bubble is punctured with a sideport blade (D). Trypan blue dye is injected to stain the bubble (E). The bubble is cut all around with corneoscleral scissors, and the graft is harvested (F).
If air is not injected into the correct plane by the needle, then often a type 2 big bubble is created,3 indicating that air is between the PDL and the Descemet membrane (DM)-endothelium complex. When a type 2 bubble is achieved, PDEK cannot be performed because the PDL is missing from the graft. However, this tissue can still be used for DMEK.
Once the bubble is created, the extreme periphery of the bubble is punctured, and trypan blue dye is injected to stain the graft. With the help of corneoscleral scissors, we then cut the graft along the peripheral edge, and the graft is harvested.
The recipient bed is prepared in exactly the same manner as in a DMEK procedure. A descemetorrhexis is performed, and the diseased DM-endothelium complex is removed. The donor lenticule is then loaded into a foldable IOL cartridge, and the graft is injected. After we confirm correct orientation of the graft, air is injected beneath the donor tissue. This pushes the donor tissue against the recipient bed and facilitates graft adherence.
APHAKIC EYES
In aphakic eyes, it is essential to perform secondary IOL fixation before the endothelial keratoplasty procedure; when this is done, the IOL helps to compartmentalize the anterior and posterior chambers. For us, the procedure of choice for secondary IOL fixation is the glued IOL technique.4,5 The intrascleral tuck of the haptics and sealing of the flaps with fibrin glue helps to achieve stable IOL fixation.
We often perform pupilloplasty, which prevents the escape of air into the vitreous cavity, helps to maintain the anterior chamber depth, and also acts as a positive factor by pushing the graft against the recipient bed. In all of these cases, we perform the single-pass, four-throw pupilloplasty technique (Figure 2).6 In this procedure, a 10-0 nylon suture threaded to the long arm of the needle is threaded through the iris leaflets that are to be apposed. Then a loop is withdrawn, and four throws are taken. The suture ends are pulled, and the loop slips inside the eye, apposing the iris tissue. In single-pass, four-throw pupilloplasty, because no knot is formed, the approximation loop of the suture lies in a parallel fashion to the iris. This has the additional advantage that there is virtually no chance of the knot rubbing against the donor graft. This is especially important in eyes with a shallow anterior chamber, in which the propensity of the suture to rub against the donor graft is increased.
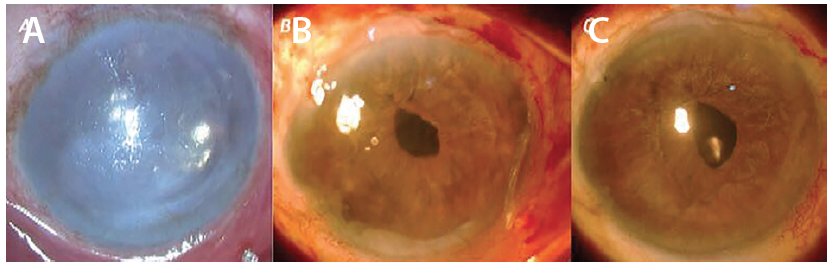
Figure 2. PDEK with glued IOL in scarred cornea with singlepass, four-throw pupilloplasty pupilloplasty. Preoperative scarred cornea (A). Two weeks after PDEK with glued IOL fixation and single-pass, four-throw pupilloplasty (B). At 2 months postoperative, the clear corneal graft is seen (C).
We have also started using a double infusion cannula technique, in which two infusion cannulas—one for air and one for infusion fluid—are placed to optimize the surgical outcomes of glued IOL fixation with PDEK. A video of the technique can be viewed below. Initially, the fluid infusion cannula is placed at the pars plana while the glued IOL technique is executed. After completion of the glued IOL procedure, we place another cannula through a trocar to act as an anterior chamber maintainer.7 The trocar–anterior chamber maintainer is connected to the Constellation Vision System (Alcon), and the cannula delivers pressurized air inside the anterior chamber to facilitate the PDEK procedure.
During descemetorrhexis, the air infusion is switched on, and the diseased DM-endothelium complex is removed. Meanwhile, the graft has been loaded, and it is then injected inside the anterior chamber. At this point, the air infusion is switched off, and the light wave of fluid seeping into the anterior chamber from the posteriorly placed infusion cannula helps to maintain the chamber depth and simultaneously facilitates the unrolling of the graft.
Once the graft is correctly unrolled, the pressurized infusion is switched on again, and the air pressure inside the anterior chamber helps the graft to adhere to the recipient bed. A steady pressure of 50 mm Hg is maintained for 30 to 45 seconds. This vaults and pushes the donor graft against the host cornea and promotes graft adherence.
THE DUA LAYER
- Also known as the pre-Descemet layer
- Serves as the demarcating line for the creation of a PDEK graft
- The graft typically involves the Dua layer, Descemet membrane, and the endothelium as a single entity
In aphakic and previously vitrectomized eyes, it can be a challenge to unfold the graft and have it achieve adherence to the recipient eye. This is in addition to the existing challenge of fixating an IOL and handling the risk of inflammation in the eye. Aphakic eyes are prone to globe collapse in the absence of posterior segment infusion, and this directly reflects the outcomes of the endothelial keratoplasty surgery. Placing the infusion fluid cannula into the vitreous cavity helps to prevent globe collapse.
YOUNG DONOR CORNEA
An advantage of PDEK over DMEK is that young donor corneas can be used in the former procedure. The youngest tissue that we have used came from a 9-month-old donor.8 The ability to use this young donor tissue expands the prospective pool of donors, which for DMEK is usually limited to those older than age 40 years. This is because grafts from younger donors tend to curl up, and, once the edges become scrolled, they can be difficult to unfold inside the eye. With the PDL serving as an additional layer in PDEK donor tissue, however, the graft does not scroll as much; the PDL acts as a splint. Due to the presence of strong adhesions between the DM and the PDL, chances of accidental creation of a type 2 bubble with the use of young donor tissue are minimized (see video below). The adhesions help to avoid the need to convert from a planned PDEK to a DMEK.
The average thickness of the graft in PDEK is approximately 30 to 35 µm, which is thinner than the grafts used in Descemet-stripping endothelial keratoplasty (DSEK) or ultrathin DSEK. This means that a smaller amount of donor stroma is transplanted, which theoretically translates to early clearance of stromal tissue. Moreover, the expensive microkeratomes that are essential to obtain a donor lenticule for ultrathin DSEK or Descemet-stripping automated endothelial keratoplasty are not needed for PDEK. In PDEK, the donor tissue can be obtained with minimal instrumentation.
CONCLUSION
Studies have shown that endothelial cell loss is comparable between DMEK and PDEK.9 Also, we have noticed that, in eyes with scarred corneas in which PDEK is performed with young donor tissue, the recipient eye tends to heal faster and better than it does with older donor tissue. We have observed this in many cases, including those that we felt preoperatively would be impossible to treat successfully with an endothelial keratoplasty procedure.10 We believe that the reason for this may be the presence of stem cells or some other factor in the stroma of these young donor corneas. Currently, however, the reason remains unclear.
1. Agarwal A, Dua HS, Narang P et al. Pre-Descemet’s endothelial keratoplasty (PDEK). Br J Ophthalmol. 2014;98(9):1181-1185.
2. Dua HS, Faraj LA, Said DG, Gray T, Lowe J. Human corneal anatomy redefined. A novel pre-Descemet’s layer (Dua’s layer). Ophthalmology. 2013;120(9):1778-1785.
3. Dua HS, Katamish T, Said DG, Faraj LA. Differentiating type 1 from type 2 big bubbles in deep anterior lamellar keratoplasty. Clin Ophthalmol. 2015;9:1155-1157.
4. Agarwal A, Kumar DA, Jacob S, et al. Fibrin glue-assisted sutureless posterior chamber intraocular lens implantation in eyes with deficient posterior capsules. J Cataract Refract Surg. 2008;34(9):1433-1438.
5. Narang P, Agarwal A, Dua HS, Kumar DA, Kumar DA, Jacob S, Agarwal A. Glued intrascleral fixation of intraocular lens with pupilloplasty and pre-Descemets endothelial keratoplasty: a triple procedure. Cornea. 2015;34(12):1627-1631.
6. Narang P, Agarwal A. Single-pass four-throw technique for pupilloplasty. Eur J Ophthalmol. 2017;27(4):506-508.
7. Agarwal A, Narang P, Kumar DA, Agarwal A. Trocar anterior chamber maintainer: improvised infusion technique. J Cataract Refract Surg. 2016;42(2):185-189.
8. Agarwal A, Agarwal A, Narang P, Kumar DA, Jacob S. Pre-Descemet endothelial keratoplasty with infant donor corneas: a prospective analysis. Cornea. 2015;34(8):859-865.
9. Altaan SL, Gupta A, Sidney LE, Elalfy MS, Agarwal A, Dua HS. Endothelial cell loss following tissue harvesting by pneumodissection for endothelial keratoplasty: an ex vivo study. Br J Ophthalmol. 2015;99(5):710-713.
10. Agarwal A, Narang P, Kumar DA, Agarwal A. Young donor–graft assisted endothelial keratoplasty (PDEK/DMEK) with epithelial debridement for chronic pseudophakic bullous keratopathy. Can J Ophthalmol. In press. doi: http://dx.doi.org/10.1016/j.jcjo.2017.03.004.


