

The main modifiable risk factor for progression of glaucoma is IOP. Available treatments include medical and surgical options. If topical medication therapy fails or is not well tolerated by the patient, a variety of surgical interventions with varying success rates are available. The choice of a surgical procedure should be evaluated critically.
Once the decision is made to treat glaucoma surgically, the most important factors that influence doctors’ choices of surgical procedure are:
- The procedure’s success rate in terms of short- and long-term IOP lowering;
- The rates of associated complications;
- The need for follow-up interventions; and
- The need to use antimetabolites adjunctively.
This overview article summarizes salient information on surgical options for glaucoma from the current literature. In Table 1, common procedures are displayed, comparing their pros and cons.

TRABECULECTOMY: Still the gold standard
Trabeculectomy is still considered the gold standard glaucoma surgical procedure, and it acts as reference point for most studies. Varying success rates have been reported, with generally good short-term results with regard to lowering IOP; however, long-term results are more controversial. Complete success after 5 years may be achieved in 40% to 71% of cases.1,2
The major disadvantage of trabeculectomy is the need for time-consuming and cost-intensive follow-up.3 The use of antimetabolites, techniques such as suture lysis or flap revision, and long-term continuation of topical medication are frequently necessary. Avascular blebs often also lack epithelial protection and may leak aqueous. Thus, severe complications such as blebitis, hypotony, and endophthalmitis can ensue.4,5
AT A GLANCE
- For decades, trabeculectomy was the gold standard of glaucoma surgery; now, canal-based approaches and MIGS procedures are taking center stage.
- The most important factors influencing doctors’ choices of surgical procedures include the procedure’s success rate, the rate of associated complications, and the need for follow-up interventions.
CANALOPLASTY
Canaloplasty is a nonpenetrating glaucoma surgery offering promising results similar to those achieved with nonpenetrating deep sclerectomy procedures. The 1-year results seem to be inferior to those for trabeculectomy,6 and complete long-term success rates range from 36% to 77.5% of cases.7-9
In contrast with trabeculectomy, postoperative management with canaloplasty is less intense. Patient satisfaction is higher,10 and further intervention is rarely needed. Severe complications, especially endophthalmitis, are rare. Occasional peripheral Descemet membrane detachment and, more commonly, transient hyphema have been reported, but the latter is considered to be a positive prognostic factor.6,11 The procedure as initially described was performed with an intracanalicular 10-0 polypropylene suture. Variations include implanting a Stegmann Canal Expander (Ophthalmos; Figure 1) and an ab-interno approach with no implant.
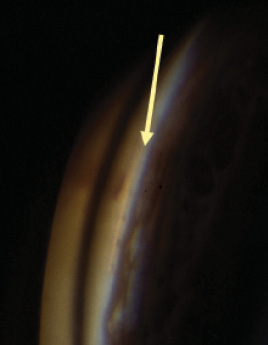
Figure 1. Gonioscopic view of Stegmann Canal Expander located in Schlemm canal following canaloplasty.
MICROINVASIVE GLAUCOMA SURGERY
Microinvasive glaucoma surgery (MIGS) is a development of relatively recent years. Therefore, for the most part, limited intermediate (1–3 year) follow-up data are available. The various devices, also outlined in Table 1, can be categorized into three groups:
1. Schlemm canal devices:
- iStent (Glaukos; Figure 2)
- Hydrus Microstent (Ivantis)
2. Suprachoroidal devices:
- CyPass Micro-Stent (Alcon)
3. Subconjunctival devices:
- Xen Gel Stent (Allergan; Figure 3)
- Ex-Press Miniglaucoma implant (Alcon).
Short-term results with all devices are in general very good.12-15 Intermediate-term results show higher variability. As MIGS devices are often implanted in conjunction with cataract surgery, many studies report results with combined procedures (implant plus phacoemulsification). The IOP-lowering effect of cataract surgery alone, 5 years postoperative, is reported to be 1.8 ±3.5 mm Hg.12-16 Results are also influenced by patients’ preoperative IOP levels, in that those with higher IOPs experience a greater effect of treatment.
For the iStent, initial success rates of up to 91% have been reported.13 Two-year data suggest success rates of 53% to 61%, depending on the definition of success.17 For the Hydrus, with success defined as a 20% reduction in IOP, success after 24 months was reported to be 80%.18 Complications are usually less frequent than with trabeculectomy; however, hyphema and IOP spikes are occasionally mentioned. In many cases, more than one iStent device is implanted to achieve an enhanced effect (Figure 2).
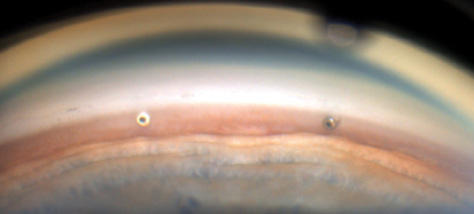
Figure 2. Two iStents implanted into Schlemm canal.
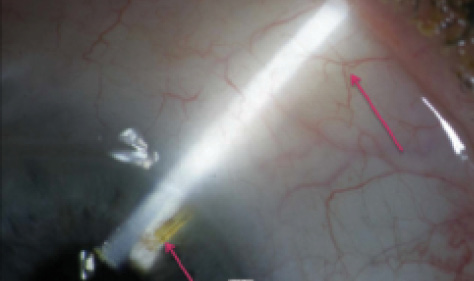
Figure 3. Xen Stent in situ after 2 years with subconjunctival filtering bleb.
In a randomized trial comparing the CyPass plus phacoemulsification with phacoemulsification alone, at 2 years postoperative, mean IOP reduction was 7.4 mm Hg in the microstent group and 5.4 mm Hg in the phaco alone group, and 85% of those receiving the device required no medications.19
At 1 year after Xen implantation, IOP reduction has been reported in the range of 23% to 30%.20,21 As with filtering surgery, there is a need for subconjunctival application of antimetabolites to secure success with this device. A review of the Ex-Press data showed complete success in 57% of patients after 5 years.22
TRABECULAR LASER
Argon laser trabeculoplasty (ALT) has been employed for many years as a way to increase aqueous outflow by opening portions of the trabecular meshwork. Recently, selective laser trabeculoplasty (SLT) has seemed to replace ALT, as it is as effective and can be repeated several times in one eye.
Short-term effects of SLT are reported to be good, but after 2 to 3 years the effect seems to fade. With follow-up of more than 3 years, IOP is successfully reduced in only 32% to 44% of eyes.23-25 Furthermore, the amount of IOP reduction achieved with SLT is relatively limited.
GLAUCOMA DRAINAGE DEVICES
Glaucoma drainage devices can be valved (eg, the Ahmed Glaucoma Valve; New World Medical) or nonvalved (eg, the Baerveldt Glaucoma Implant; Johnson & Johnson Vision).26-32 In short-term results, the success rate with these devices is comparable with those of other procedures.26,30 Long-term success with the Ahmed valve has been reported to be 8% to 50%,27 with a clinical annual failure rate of 10%.27 For the Baerveldt device, the 5-year results are similar, and success may vary between 14% and 71%.30,31 In general, data are limited, groups are heterogeneous, and definitions of success vary significantly.32
Complications similar to those of trabeculectomy have been reported with glaucoma drainage devices; additional disadvantages include corneal decompensation due to the placement of the tube and strabismus with diplopia.27,29
CYCLODESTRUCTION
Cyclophotocoagulation (CPC) is reserved mostly for refractory glaucoma when other procedures have failed. CPC can be performed transsclerally or with an endoscope. The indications are limited because of strong inflammatory reactions and a broad spectrum of complications such as cystoid macular edema, necrotising scleritis, phthisis, and sympathetic ophthalmia.33,34
Often, the procedure must be repeated to achieve satisfying IOP reduction. There are few published data on the long-term success of CPC; at 2 years, the success rate is 61% for transscleral CPC and 74% for endocyclophotocoagulation.35,36
The relatively new concept of micropulse laser CPC has shown promising results in terms of predictability and reduction of side effects, but long-term data are not yet available.34,37
CONCLUSION
For decades, trabeculectomy was the gold standard of glaucoma surgery. New canal-based approaches and MIGS procedures have improved outcomes by reducing complications and the need for postoperative care.
1. Broadway DC, Clark A. The Norwich Trabeculectomy Study: long-term outcomes of modern trabeculectomy with respect to risk factors for filtration failure. J Clin Exp Ophthalmol. 2014;5:371.
2. Nouri-Mahdavi K, Brigatti L, Weitzman M, Caprioli J. Outcomes of trabeculectomy for primary open-angle glaucoma. Ophthalmology. 1995;102(12):1760-1769.
3. Brüggemann A, Müller M. Trabeculectomy versus canaloplasty – utility and cost-effectiveness analysis [in German]. Klin Monbl Augenheilkd. 2012;229(11):1118-1123.
4. Zahid S, Musch DC, Niziol LM, Lichter PR. Risk of endophthalmitis and other long-term complications of trabeculectomy in the Collaborative Initial Glaucoma Treatment Study (CIGTS). Am J Ophthalmol. 2013;155(4):674-680.
5. Jampel HD, Solus JF, Tracey PA, et al. Outcomes and bleb-related complications of trabeculectomy. Ophthalmology. 2012;119(4):712-722.
6. Zhang B, Kang J, Chen X. A system review and meta-analysis of canaloplasty outcomes in glaucoma treatment in comparison with trabeculectomy. J Ophthalmol. 2017;2017:2723761.
7. Grieshaber MC, Pienaar A, Olivier J, Stegmann R. Canaloplasty for primary open-angle glaucoma: long-term outcome. Br J Ophthalmol. 2010;94(11):1478-1482.
8. Lewis RA, von Wolff K, Tetz M, et al. Canaloplasty: three-year results of circumferential viscodilation and tensioning of Schlemm canal using a microcatheter to treat open-angle glaucoma. J Cataract Refract Surg. 2011;37(4):682-690.
9. Bull H, Von Wolff K, Körber N, Tetz M. Three-year canaloplasty outcomes for the treatment of open-angle glaucoma: European study results. Graefes Arch Clin Exp Ophthalmol. 2011;249(10):1537-1545.
10. Klink T, Sauer J, Korber NJ, et al. Quality of life following glaucoma surgery: canaloplasty versus trabeculectomy. Clin Ophthalmol. 2015;9:7-16.
11. Koch JM, Heiligenhaus A, Heinz C. Canaloplasty and transient anterior chamber haemorrhage: a prognostic factor? Klin Monbl Augenheilkd. 2011;228(5):465-467.
12. Fea AM, Consolandi G, Zola M, et al. Micro-bypass implantation for primary open-angle glaucoma combined with phacoemulsification: 4-year follow-up. J Ophthalmol. 2015;2015:795357.
13. Vold SD, Voskanyan L, Tetz M, et al. Newly diagnosed primary open-angle glaucoma randomized to 2 trabecular bypass stents or prostaglandin: outcomes through 36 months. Ophthalmol Ther. 2016;5(2):161-172.
14. Lindstrom R, Lewis R, Hornbeak DM, et al. Outcomes following implantation of two second-generation trabecular micro-bypass stents in patients with open-angle glaucoma on one medication: 18-month follow-up. Adv Ther. 2016;33(11):2082-2090.
15. Samuelson TW, Katz LJ, Wells JM, Duh YJ, Giamporcaro JE. Randomized evaluation of the trabecular micro-bypass stent with phacoemulsification in patients with glaucoma and cataract. Ophthalmology. 2011;118(3):459-467.
16. Shingleton BJ, Pasternack JJ, Hung JW, O’Donoghue MW. Three and five year changes in intraocular pressures after clear corneal phacoemulsification in open angle glaucoma patients, glaucoma suspects, and normal patients. J Glaucoma. 2006;15(6):494-498.
17. Craven ER, Katz LJ, Wells JM, Giamporcaro JE. Cataract surgery with trabecular micro-bypass stent implantation in patients with mild-to-moderate open-angle glaucoma and cataract: two-year follow-up. J Cataract Refract Surg. 2012;38(8):1339-1345.
18. Pfeiffer N, Garcia-Feijoo J, Martinez-de-la-Casa JM, et al. A randomized trial of a Schlemm’s canal microstent with phacoemulsification for reducing intraocular pressure in open-angle glaucoma. Ophthalmology. 2015;122(7):1283-1293.
19. Vold S, Ahmed II, Craven ER, et al; CyPass Study Group. Two-year COMPASS trial results: supraciliary microstenting with phacoemulsification in patients with open-angle glaucoma and cataracts. Ophthalmology. 2016;123(10):2103-2112.
20. Galal A, Bilgic A, Eltanamly R, Osman A. Xen glaucoma implant with mitomycin c 1-year follow-up: result and complications. J Ophthalmol. 2017;2017:5457246.
21. Pérez-Torregrosa VT, Olate-Pérez Á, Cerdà-Ibáñez M, et al. Combined phacoemulsification and Xen45 surgery from temporal approach and 2 incisions. Arch Soc Esp Oftalmol. 2016;91(9):415-421.
22. Chan JE, Netland PA. Ex-Press glaucoma filtration device: efficacy, safety, and predictability. Med Devices (Aukl). 2015;8:381-388.
23. Patel V, El Hawy E, Waisbourd M, et al. Long-term outcomes in patients initially responsive to selective laser trabeculoplasty. Int J Ophthalmol. 2015;8(5):960-964.
24. Weinand FS, Althen F. Long-term clinical results of selective laser trabeculoplasty in the treatment of primary open angle glaucoma. Eur J Ophthalmol. 2006;16(1):100-104.
25. Damji KF, Bovell AM, Hodge WG, et al. Selective laser trabeculoplasty versus argon laser trabeculoplasty: results from a 1‐year randomised clinical trial. Br J Ophthalmol. 2006;90(12):1490-1494.
26. Sharpe RA, Kammerdiener LL, Wannamaker KW, Fan J, Sharpe ED. Comparison of outcomes of resident-performed Ahmed valve implantation vs trabeculectomy. J Curr Glaucoma Pract. 2016;10(2):60-67.
27. Patel S, Pasquale LR. Glaucoma drainage devices: a review of the past, present, and future. Semin Ophthalmol. 2010;25(5-6):265-270.
28. Minckler DS, Francis BA, Hodapp EA, et al. Aqueous shunts in glaucoma: a report by the American Academy of Ophthalmology. Ophthalmology. 2008;115(6):1089-1098.
29. Budenz DL, Feuer WJ, Barton K, et al. Postoperative complications in the Ahmed Baerveldt Comparison Study during five years of follow-up. Am J Ophthalmol. 2016;163:75-82.
30. Budenz DL, Barton K, Gedde SJ, et al. Five-year treatment outcomes in the Ahmed Baerveldt Comparison Study. Ophthalmology. 2015;122(2):308-316.
31. Hamanaka T, Otora K, Ono K, Ishida N. Long-term results of non-valved glaucoma drainage implant surgery and glaucoma drainage implant combined with trabeculectomy. Indian J Ophthalmol. 2014;62(9):911-916.
32. Wang Y-W, Wang P-B, Zeng C, Xia X-B. Comparison of the Ahmed glaucoma valve with the Baerveldt glaucoma implant: a meta-analysis. BMC Ophthalmol. 2015;15:132.
33. Bloom PA, Tsai JC, Sharma K, et al. “Cyclodiode”. Trans-scleral diode laser cyclophotocoagulation in the treatment of advanced refractory glaucoma. Ophthalmology. 1997;104(9):1508-1520.
34. Ekici F, Waisbourd M, Katz LJ. Current and future of laser therapy in the management of glaucoma. Open Ophthalmol J. 2016;10:56-67.
35. Feldman RM, et al. Histopathologic findings following contact transscleral semiconductor diode laser cyclophotocoagulation in a human eye. J Glaucoma. 1997;6(2):139-140.
36. Lima FE, et al. A prospective, comparative study between endoscopic cyclophotocoagulation and the Ahmed drainage implant in refractory glaucoma. J Glaucoma. 2004;13(3):233-237.
37. Aquino MC, Barton K, Tan AM, et al. Micropulse versus continuous wave transscleral diode cyclophotocoagulation in refractory glaucoma: a randomized exploratory study. Clin Exp Ophthalmol. 2015;43(1):40-46.



