

Residual and surgically induced astigmatism after corneal refractive surgery is a difficult and frustrating problem. Causes of surgically induced astigmatism include surgeon experience, instruments, the laser, and patient characteristics. Astigmatism after corneal refractive surgery may decrease patients’ UCVA and quality of vision. When the astigmatism is irregular, it can also decrease patients’ BCVA and cause monocular diplopia and ghosting of images. Management requires understanding the cause of the astigmatism and thoroughly evaluating the patient.
CAUSES OF POSTOPERATIVE ASTIGMATISM
Astigmatism may be measured by keratometry and refraction, whereas corneal topographic techniques help to define irregular astigmatism. One of most frequent causes of residual astigmatism is regression, a progressive loss of the achieved refractive correction over time. Epithelial hyperplasia and stromal remodeling are thought to contribute to regression.1
The other most common cause of residual astigmatism is a decentered ablation. Misalignment of the laser ablation pattern can result in an uneven corneal surface, irregular astigmatism, higher-order aberrations, and poor visual acuity. The latest laser platforms with advanced eye-tracking systems have reduced the incidence of decentered ablations, but significant misalignment can still occur. Patients whose ablation is decentered may experience reduced visual acuity, irregular astigmatism, glare, halos, and undercorrection. The problem typically reaches clinical significance if the deviation from the visual axis exceeds 0.5 mm.2
FIRST STEPS
Patient dissatisfaction with visual acuity is the best indication that the residual astigmatism requires treatment. It is prudent, however, to wait at least 3 to 6 months for their refraction to stabilize before intervening surgically.
The first step when considering enhancement surgery is a thorough patient evaluation, including a complete ocular examination, corneal topography and pachymetry, and an assessment of the individual’s UCVA and BCVA. The surgical records on the excimer laser treatment should also be reviewed.
OPTIONS FOR TREATMENT
One-step topography-guided transepithelial PRK. Laser treatment is customized based on morphologic and refractive data calculated by the tomographer and spherical error quantified during the visual acuity examination. The target refractive and ablation zones are defined by the projection of an ideal pupil, as determined by dynamic pupillometry, onto the corneal surface.
A transepithelial approach can address the epithelium’s refractive contribution. By using the epithelium as a masking agent, the irregular corneal curvature may be made smoother and more regular, epithelial and stromal defects may be corrected, the risk of posttreatment haze can be reduced, and a significant improvement in visual acuity can be achieved.3 Improvement in a patient’s visual acuity and quality of vision is due to the correction of lower- and higher-order aberrations (Figures 1 and 2).
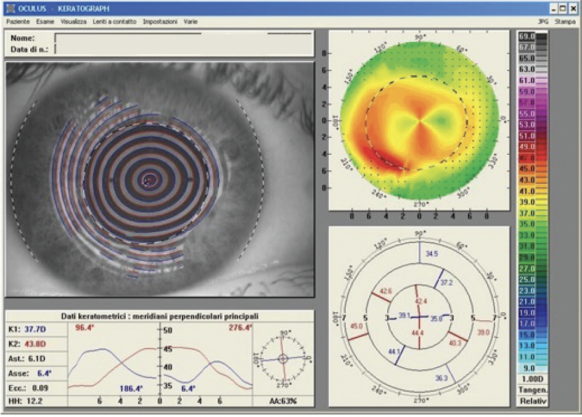
Figure 1. A computed tomography scan of the right eye of a 36-year-old man after PRK. The ablation for a high degree of astigmatism was performed on the wrong meridian and doubled his astigmatism value. After 3 months, his BCVA was 20/25 with a refraction of +0.50 +6.50 x 90º.
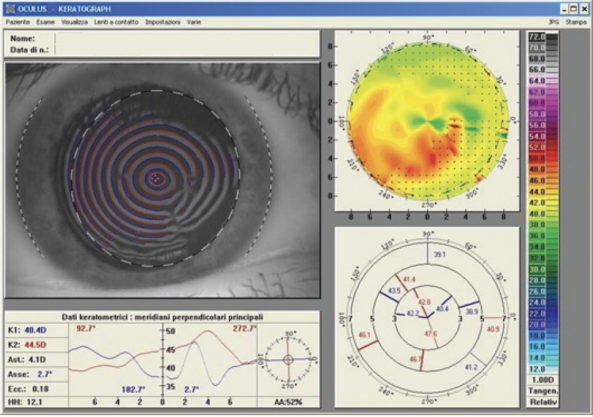
Figure 2. A computed tomography scan of the same eye 12 months after one-step topography-guided transepithelial PRK. The patient’s final corrected distance visual acuity was 20/20 with a refraction of +0.50 -1.50 x 155º.
Wavefront-guided PRK. Another option for treating an irregular cornea after unsuccessful keratorefractive surgery is wavefront-guided PRK. This procedure can minimize corneal higher-order aberrations. In highly irregular corneas, however, wavefront-guided PRK has some limitations, including a decreased reliability of the wavefront profiles, a lack of correlation between the corneal shape and the wavefront profile, a dependence on accommodation and pupil size, and a lack of information to customize the transition zone. In contrast, topography-guided ablations demonstrate good outcomes for the correction of astigmatism in highly aberrated corneas, many of which are poor candidates for wavefront-guided therapy.4
CONCLUSION
The options for treating residual astigmatism after corneal ablation must be considered carefully to optimize results and ensure patient satisfaction postoperatively.
1. Spadea L, Giammaria D, Trabucco P. Corneal wound healing after laser vision correction. Br J Ophthalmol. 2016;100(1):28-33.
2. Spadea L, Giovannetti F. Main complications of photorefractive keratectomy and their management. Clin Ophthalmol. 2019;13:2305-2315.
3. Spadea L, Visioli G, Mastromarino D, Alexander S, Pistella S. Topography-guided trans-epithelial no-touch photorefractive keratectomy for high irregular astigmatism after penetrating keratoplasty: a prospective 12-months follow-up. Ther Clin Risk Manag. 2021;17:1027-1035.
4. Pasquali T, Krueger R. Topography-guided laser refractive surgery. Curr Opin Ophthalmol. 2012;23(4):264-268.



