Corneal Topography Is a Critical Tool
Li Wang, MD, PhD

Corneal topography is a crucial part of the preoperative assessment of cataract surgery patients and toric IOL planning.
CORNEAL SURFACE DISEASE DETECTION
The presence of corneal surface disease can render corneal astigmatism measurements unreliable. In a study evaluating the incidence of ocular surface disease in patients presenting for cataract surgery, Gupta and colleagues found that 80% had at least one abnormal tear test.1
Corneal topography, especially Placido ring–based corneal topography, can be helpful for detecting corneal surface disease. The Placido mires are sensitive to irregularities of the precorneal tear film. In an eye with a dry ocular surface or anterior basement membrane dystrophy, the Placido mires are irregular (Figure), and data may be misleading or absent on the corneal topographic maps.
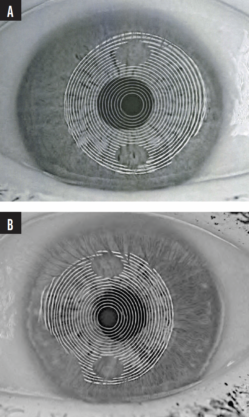
Figure. Irregular Placido mires on corneal tomography (Galilei, Ziemer Ophthalmic Systems) in an eye with a dry ocular surface (A) and an eye with anterior basement membrane dystrophy (B).
Courtesy of Li Wang, MD, PhD
CORNEAL ASTIGMATISM ASSESSMENT: REGULAR VERSUS IRREGULAR ASTIGMATISM
Corneal topography may be used to assess the pattern of corneal astigmatism and surface regularity. Specifically, the corneal topographic map is useful for qualitative assessments of whether a cornea has regular (ie, orthogonal and symmetric bow tie pattern) or irregular (ie, asymmetric or skewed bow tie pattern) astigmatism. Corneal topographic indices such as surface regularity index, surface asymmetry index, and corneal higher-order aberrations are helpful for quantitative evaluations of regular and irregular astigmatism.
It is difficult to correct asymmetric bow tie astigmatism or irregular astigmatism with a symmetric toric IOL. The prevalence of irregular astigmatism is approximately 40%.2 Corneal irregularities, especially in the central 4- to 5-mm zone, are a significant consideration when deciding whether to implant a toric IOL.
Li et al investigated the correction of asymmetric bow tie corneal astigmatism with toric IOLs and found that accuracy decreased as asymmetry increased.3 Gao and colleagues found that patients who had irregular astigmatism with a regular central component could be considered for toric IOL implantation.4 Other studies have found toric IOLs to be effective in patients with mild irregular corneal astigmatism, including keratoconus,5 pellucid marginal degeneration,6 and corneal transplantation.7
CORNEAL ASTIGMATISM MAGNITUDE AND STEEP MERIDIAN EVALUATION
Corneal astigmatism values obtained with a corneal topographer may be compared to those obtained with an ocular biometer. When the values from both devices are consistent, it can give surgeons greater confidence in toric IOL planning.
Correcting corneal astigmatism in eyes that have a history of radial keratotomy, PRK, and LASIK can be challenging owing to the presence of varying amounts of irregular astigmatism. My colleagues and I found that toric IOLs could effectively correct preexisting corneal astigmatism in these eyes if the following three criteria were met:
No. 1. Regular bow tie corneal astigmatism was present within the central 3-mm zone;
No. 2. The difference in corneal astigmatism magnitude between two devices (two biometers or one biometer and one corneal topographer) was 0.75 D or less; and
No. 3. The difference in the astigmatism meridians between two devices was 15º or less.
The percentages of eyes with 0.50 D or less of postoperative refractive astigmatism were 80%, 84%, and 76% in eyes that had undergone myopic LASIK/PRK, hyperopic LASIK/PRK, and radial keratotomy, respectively.8,9
SUMMARY
Corneal topography is a critical tool in toric IOL planning. When corneal surface disease is detected, it should be addressed before corneal astigmatism measurements are attempted.
1. Gupta PK, Drinkwater OJ, VanDusen KW, Brissette AR, Starr CE. Prevalence of ocular surface dysfunction in patients presenting for cataract surgery evaluation. J Cataract Refract Surg. 2018;44(9):1090-1096.
2. Oshika T, Tomidokoro A, Tsuji H. Regular and irregular refractive powers of the front and back surfaces of the cornea. Exp Eye Res. 1998;67(4):443-447.
3. Li H, He W, Guo D, et al. Correction of asymmetric bowtie corneal astigmatism with a toric intraocular lens: outcomes and accuracy of measurement modes. J Pers Med. 2023;13(3):401.
4. Gao Y, Ye Z, Chen W, Li J, Yan X, Li Z. Management of cataract in patients with irregular astigmatism with regular central component by phacoemulsification combined with toric intraocular lens implantation. J Ophthalmol. 2020;2020:3520856.
5. Alió JL, Peña-García P, Abdulla Guliyeva F, Soria FA, Zein G, Abu-Mustafa SK. MICS with toric intraocular lenses in keratoconus: outcomes and predictability analysis of postoperative refraction. Br J Ophthalmol. 2014;98(3):365-370.
6. Luck J. Customized ultra-high-power toric intraocular lens implantation for pellucid marginal degeneration and cataract. J Cataract Refract Surg. 2010;36(7):1235-1238.
7. Devebacak A, Degirmenci C, Barut Selver O, Palamar M, Egrilmez S. Correction of high astigmatism with toric intraocular lens in eyes with corneal transplant. Eur J Ophthalmol. Published online August 31, 2022. doi:10.1177/11206721221123885
8. Cao D, Wang L, Koch DD. Outcome of toric intraocular lens implanted in eyes with previous corneal refractive surgery. J Cataract Refract Surg. 2020;46(4):534-539.
9. Canedo ALC, Wang L, Koch DD, Al-Mohtaseb Z. Accuracy of astigmatism correction with toric intraocular lens implantation in eyes with prior radial keratotomy. J Cataract Refract Surg. 2022;48(4):417-420.
Exploring Opportunities and Challenges in Ray Tracing
An interview with Douglas D. Koch, MD

CRST Global: Numerical ray tracing has been proposed as an alternative to Gaussian optics for IOL power calculation. Can you explain the basic concept of ray tracing and the factors that are considered in the context of IOL power calculation?
Douglas D. Koch, MD: Ray tracing, as the name suggests, traces the path of light rays as they traverse the different refracting surfaces of the eye. The method produces an image on the retina, and the power of the IOL is determined by estimating the sharpest image. A key strength of ray tracing is its ability to incorporate higher-order aberrations of the front and back of the cornea and of various IOL designs.
Despite the advantages of ray tracing, the technology has not consistently demonstrated superiority over standard Gaussian optics. This is likely due to the persisting problem of determining the effective lens position (ELP)—an issue for which ray tracing does not have a unique solution. The PhacoOptics IOL calculation software (IOL Innovations ApS), developed by Thomas Olsen, MD, uses the concept of the C constant to represent a numerical value for predicting the ELP. Although not strictly a ray tracing solution, the C constant is an excellent formula that attempts to address one key challenge in IOL power calculation.
CRST Global: What, in your opinion, is the potential advantage of using ray tracing for IOL power calculation?
Dr. Koch: I think the main strength of ray tracing is evident in patients with irregular corneas. Determining the optical impact of an irregular cornea with Gaussian optics can pose challenges. Ray tracing may provide a more accurate assessment for eyes that have keratoconus or a history of corneal refractive surgery and penetrating keratoplasty. Identifying which part of these corneas significantly affects the final visual acuity is particularly challenging. Ray tracing is ideal to better handle this task.
CRST Global: What evidence is available that ray tracing is comparable to or even surpasses modern IOL calculation formulas? Are there any particular situations where ray tracing proves especially beneficial?
Dr. Koch: My colleagues and I have conducted research using Okulix (Panopsis), ray tracing software developed by Paul-Rolf Preussner, MD, PhD, and the Anterion (Heidelberg Engineering) in post-LASIK eyes. With small numbers, we have found that Okulix was comparable to the Barrett True K formula for IOL power calculation but with a tighter range, which is a promising development.
I am not aware of any studies that have unequivocally demonstrated the benefit of ray tracing. Jaime Aramberri Agesta, MD, in Spain, however, uses OpticStudio (Zemax) for ray tracing calculations in post-LASIK patients. He has reported that it achieves superior results in these eyes compared to those obtained using current IOL calculation methods (personal communication).
CRST Global: What improvements or challenges need to be addressed for ray tracing to be integrated effectively into routine IOL power calculation?
Dr. Koch: For ray tracing to be established as a superior method for IOL power calculation, we need to further improve our means of determining the ELP, as it is a significant source of error. This issue can sometimes overshadow any potential benefits of ray tracing. If newer methods could be used to determine the ELP, such as anterior segment OCT measurements to identify the scleral spur and predict the IOL position, we might begin to see some advantages of ray tracing.
An interesting application of ray tracing is present in the Zeiss AI IOL calculator (Carl Zeiss Meditec). This novel formula was designed by conducting ray tracing for 500,000 IOLs to train the dataset and then using AI and biometric data to perform the IOL calculation. It’s not strictly a ray tracing formula, but it leverages ray tracing to enhance the output from AI.
CRST Global: As technology advances, what developments or enhancements do you anticipate in the field of ray tracing for IOL power calculations?
Dr. Koch: It remains uncertain whether we will see significant advancements, particularly in solving the ELP conundrum. If we can do a better job of predicting ELP, ray tracing should be the optimal approach, aided perhaps by AI. In general, I am somewhat skeptical about how accurate we can be—surpassing 90% within ±0.50 D of the intended refractive correction may not be feasible. I anticipate that some form of postoperative modification will eventually become standard practice.


