The goal of refractive surgery in eyes that have not undergone previous ocular surgery is to achieve postoperative distance UCVA equal to the patient’s preoperative distance BCVA. Hyperopic excimer laser ablations use a ring profile in the paracentral-to-peripheral cornea. The hyperopic ablation algorithm steepens the central corneal profile to achieve the desired correction. This is in contrast to myopic corrections, which flatten the central cornea.
Historically, hyperopic corrections have been more of a challenge for refractive surgeons than myopic corrections. This article reviews some of the reasons for that difference and offers some guidelines for achieving accurate refractive surgical corrections in hyperopic patients.
Refractions in hyperopia are typically underestimated
Clinical subjective refractions in hyperopia are typically underestimated due to the accommodative response of hyperopic patients, who are used to accommodating for far distance refraction to bring the hyperfocal distance into focus. This response partly masks the patient’s real hyperopic defect.
A reasonable approach to achieving an accurate refraction, at the expense of investing chair time, is to begin by determining the distance BCVA from the patient’s subjective refraction under photopic conditions (ie, manifest refraction). The maximum positive sphere that the patient accepts should be attained. In clinical practice, this requires the accommodation of the crystalline lens to be paralyzed, usually through use of an anticholinergic agent. This results in both ciliary body paralysis and pupil mydriasis. A subjective refraction is then conducted in this condition with the aid of test lenses and phoropter. Room lights should be dimmed to avoid blinding glare. The refraction obtained is called the cycloplegic refraction.
Often, the patient will not accept this full refractive correction; therefore, a subsequent refraction should be ascertained under photopic conditions. Due to the dilation, this measurement (ie, pull-down refraction) should be taken at least 1 day after the initial refraction. Again, the target is the maximum accepted positive sphere, but starting this time from the hyperopic value obtained under cycloplegic conditions. The determined value is tested for a few minutes with a test lens. The accepted value can be used to determine the laser input for refractive surgical correction.
For example, a patient is best corrected with 2.00 D (manifest refraction), but he accepts 4.00 D in subjective mydriasis (cycloplegic refraction). If the patient accepts a value of 3.50 D with a normal pupil under photopic conditions (pull-down refraction) sustained for a few minutes, this value should be used as the basis for laser settings.
The value of the laser settings should not differ more than 0.75 D from the cycloplegic refraction. If the difference is greater, glasses with that correction should first be prescribed for a few weeks or months to let the patient relax into the accommodation, thus preventing latent hyperopia that would emerge after laser treatment. Particularly in young farsighted patients, in whom there can be a big difference between the optical correction they wear and the cycloplegic refraction, wearing a higher optical correction for a few months can reduce the accommodative spasm and allow better correction of the true latent hyperopia.
Typically, because of their short axial lengths, hyperopic eyes have an angle between pupillary and visual axes (ie, angle kappa) greater that 5.5°, manifesting a relatively large pupillary offset from the corneal vertex.1 Hyperopic patients fixate predominantly nasally from the pupil center, so a nasal offset of the treatment makes sense in these eyes.
Modern corneal topographers and tomographers display the distance between the pupil center and the corneal vertex, which is normally between 200 and 700 µm. Using this value to determine treatment centration helps to reduce the induction of coma and trefoil aberrations.2 Taking into account the often significant offset seen in highly hyperopic patients, or at least a large fraction of the measured offset, will shift the optical axis of the ablation map onto, or closer to, the visual axis of the patient.3
The opticAL ZONE SHOULD BE AS LARGE AS POSSIBLE
The optical zone (OZ) for the treatment of hyperopia should be as large as possible, but the size of the LASIK flap limits the diameter of the OZ. The treatment zone must fit within the flap size; a larger zone cannot be selected. The OZ for hyperopic correction should not be less than 6.5 mm, and preferably should be greater than 6.7 or even 7 mm, further including a transition zone that is wide and smooth and that optically blends the OZ to the untreated cornea.4
To ensure good preservation of the patient’s distance BCVA, a reasonable approach is the routine use of an OZ of at least 6.7 mm and a total treatment zone of at least 9.0 mm (Figure).5 For such large treatment zones to be possible in LASIK, a large flap leaving at least a 9.5-mm diameter of real stromal bed is needed, approaching the boundaries of the limbus and maximizing the exposed stroma.6 Temporal or nasal hinges allow the exposure of a larger corneal surface compared with superior hinges because the corneal diameter is greater horizontally than vertically. Furthermore, temporal or nasal hinges may be less affected by rolling movements of the eye, which can be pronounced in hyperopic patients during photoablation.7
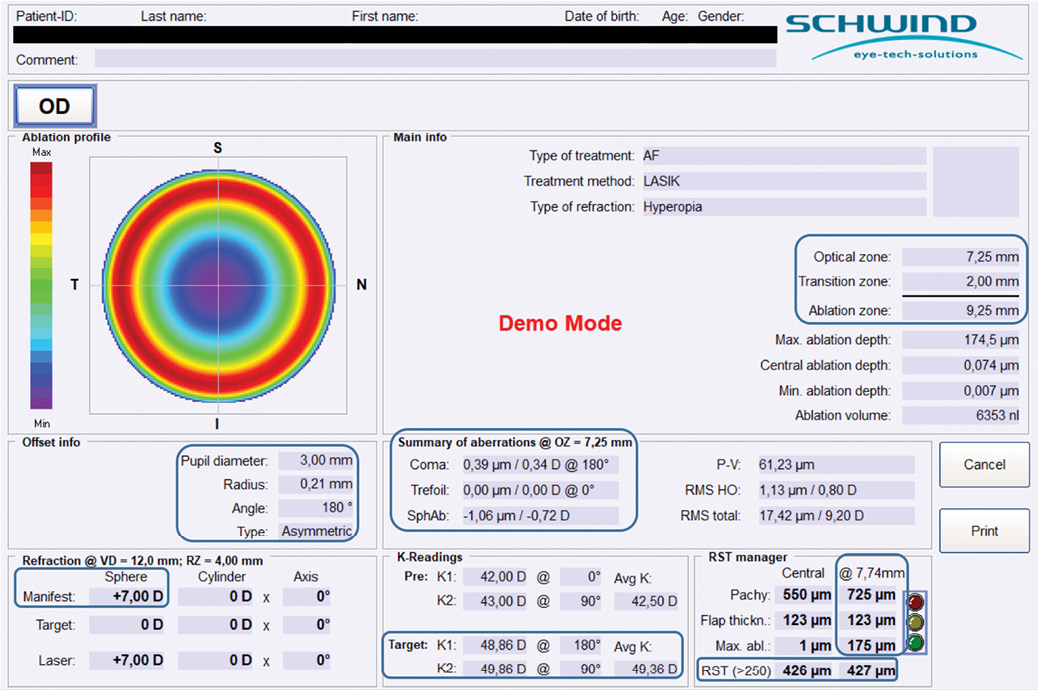
Figure. For this 7.00 D hyperopic correction, the 9.25-mm ablation zone includes a 2.0-mm transition zone.
A very large treatment zone is the key to success in hyperopic laser treatments. In eyes with small corneal limbal diameters, highly hyperopic treatments may be challenging. The flap cut can cause bleeding, especially in contact lens wearers. These small corneal diameters are found most often in eyes with high hyperopia and steep keratometry (K) measurements.
Another challenge in high hyperopic corrections is that the LASIK flaps are often too small to cover the exposed stromal surface, which becomes larger during treatment due to the corneal steepening introduced by the ablation profile. This has been shown to increase the risk for epithelial ingrowth.8
Repositioning the LASIK flap after such treatment may induce regression or undercorrection because the flap cannot follow the deep, steep doughnut shape of the ablation. It can also induce vertical coma because the flap cannot cover the ablation completely.9
Considering these implications of high hyperopic corrections, extremely large flaps and OZ sizes (with large transition zones) must be targeted for LASIK treatments. Surface ablation techniques such as PRK also require large OZ sizes.10 A low curvature gradient between the optical and transition zones is thought to reduce regression.11
CONsider ABLATION DESIGN
Treatment of high hyperopia in photoablative surgery has always been a challenge. The results of published series are controversial, as the results are directly dependent on the technology used in the surgery.
We use lasers in the Amaris laser family (Schwind eye-tech-solutions), which can treat high hyperopia efficaciously under certain conditions thanks to a specific ablation profile in which the size of the transition zone to the peripheral cornea varies according to the magnitude of the hyperopia to be treated.12 For example, for a treatment of 2.00 D with an OZ of 6.7 mm, the total treatment zone would be 7.8 mm; for a treatment of 5.00 D with the same OZ, the total treatment zone would be as large as 8.7 mm.
The higher the amount of photoablation in the corneal periphery, the more important it is to optimize optical quality. Only a laser with a very high frequency can achieve such large photoablations without increasing the time of the treatment, which is associated with stromal dehydration, loss of fixation, and patient discomfort. A distinct initial overcorrection is systematic for hyperopia and astigmatism. The patient should be warned about the possibility of visual discomfort in distance vision during the early postoperative period.13
Know The LIMITS OF HYPEROPIC CORRECTION
LASIK in hyperopic eyes has been shown to induce more higher-order aberrations per corrected diopter than LASIK in a similar group of myopic eyes. Because of this, the upper limit for hyperopic corrections is generally lower than that for myopic corrections (up to 6.00 or 7.00 D vs -12.00 to -14.00 D).12
Several considerations factor into determining the upper limit for hyperopic refractive corrections. First, the cornea is thinner at the center compared with the periphery. In any refractive treatment mode, the cornea must be manipulated in such a way that the natural curvature and form of the cornea is retained as much as possible. Respecting this limit avoids making the cornea thinner in the periphery compared with the center.12
Assuming an average corneal thickness (central pachymetry ~550 µm and peripheral ~700 µm at 8 mm diameter, with variations),5 and expressing the limit to retain the natural corneal form in diopters, the upper limit for hyperopic correction is 7.00 D. Interestingly, this upper limit is irrespective of OZ size and relates only to the center-to-OZ-edge variations.10
It is our personal impression that the limit of 7.00 D that is imposed by center-to-periphery pachymetry cannot be used for a large population, but rather is subject to interindividual variability. Considering that the average K in human eyes is 43.70 D in the vertical meridian and 42.70 D in the horizontal meridian, with a standard deviation of 2.00 D, 97% of the population has a natural K of 48.00 D in the vertical meridian. Targeting a postoperative K of 48.00 D, therefore, can definitely be considered safe. Adding three standard deviations to this average value renders an extreme limit of 50.00 D for postoperative target K. Beyond this, safety limits are exceeded.
These values suggest a treatment limit of 7.00 D in extreme situations and 5.00 D with a conservative approach. It must be noted that these values are based on a statistical average of the human population and exclude interindividual differences.
Human eyes tend to have a natural spherical aberration of 0.1 µm. It has been recently evaluated using adaptive optics that the human eye is capable of comfortably tolerating up to 0.56 µm of positive or negative spherical aberration at a 6.0 mm diameter.14 This suggests that the average human eye can safely tolerate up to -0.66 µm of spherical aberration induced through hyperopic corrections. Most modern laser systems induce slightly less than 0.1 µm of spherical aberration per diopter of correction, again suggesting a similar upper limit for hyperopic correction of approximately 7.00 D.
Patients with high levels of hyperopia must be warned that they may lose lines of distance BCVA following any correction for high hyperopia to due to minification of the image on the retina compared with the image created on the retina with spectacles. The same effect is seen with IOLs correcting high levels of hyperopia.13
Understand the implications of prior proceDures and conditions
One must also consider the effect of postoperative reepithelialization in treatment modes that require epithelial removal and regrowth. Reepithelialization can lead to thinner epithelium over steepened areas of the cornea. LASIK may also affect epithelial thickness and cause the epithelium to be thinnest over the steepest part of the cornea. In some cases, this may lead to apical syndrome, in which corneal scarring can occur,15 and, in very rare cases, to diurnal variations in refraction and vision that are consistent with a diurnal fluctuation in epithelial thickness. Different treatment modes may have different implications for the upper limit of hyperopic correction. Surface ablation methods such as PRK may be detrimental because the epithelium may react more aggressively, leading to greater regression.10,16
The size of the effective OZ is more important in hyperopic refractive corrections than in myopic refractive corrections of the same magnitude. A high postoperative K value has been shown to affect distance BCVA in many studies, limiting the indications for highly hyperopic corneal refractive surgery to patients with low initial K values. The magnitude of the keratometric change is the main factor affecting visual performance. The bad reputation of highly hyperopic corrections is related to the use of large photoablations in small OZs, resulting in poor postoperative optical quality. Furthermore, a steep initial K correlates with a smaller corneal diameter. This can further limit creation of the required flap diameter. All of these factors may result in small effective OZ sizes with high degrees of aberration postoperatively, further emphasizing the need for targeting large treatment zones in highly hyperopic treatments.2,7,17,18
Dry eye after LASIK. One of the most common postoperative sequelae of LASIK is dry eye disease (DED), with a majority of patients developing some degree of intermittent or transient DED.19 The afferent sensory nerves that mediate the sensitivity levels of the ocular surface come from the ophthalmic division of the trigeminal nerve. These innervations penetrate the cornea peripherally and form a dense subbasal nerve plexus from which terminal nerve endings enter into the epithelium.20,21
Corneal denervation is accredited to be the most significant cause of post-LASIK DED.19,20,22 Hyperopic laser treatments seem to have a more deleterious impact upon the ocular surface compared with myopic treatments,23 which may be due to a greater impact upon corneal sensitivity due to differences in the spatial treatment profile, which is more peripherally placed within the corneal stroma in hyperopes compared with myopes, resulting in reduced corneal sensitivity postoperatively after hyperopic laser treatments.23 Because post-LASIK DED is extremely common and can have a deleterious effect upon a patient’s vision and quality of life, hyperopic patients should be examined carefully preoperatively for either exclusion from treatment or active ocular surface management if any signs of ocular surface disease are seen prior to hyperopic LASIK.24
CXL. The use of CXL after hyperopic treatments has been postulated to increase the stability of the correction, thereby reducing regression over time.25
SMILE. In the relatively new laser vision correction technique of small-incision lenticule excision (SMILE), only the femtosecond laser is used. The procedure has mainly been used to date for the correction of myopia and astigmatism, but some surgeons have presented their experiences with the correction of hyperopia using SMILE.26
The evolution of the SMILE technique for hyperopia essentially shares the primary elements discussed above: treatment of maximum positive sphere; shift of the optical axis of the correction onto the visual axis of the patient; use of the largest OZ possible; and use of a wide, smooth, and optically blended transition zone to the untreated cornea with a low curvature gradient, with compensation of the induction of spherical aberration.
CONCLUSION
The correction of high hyperopia with corneal refractive surgery is possible, but it is important to follow much stricter protocols compared with myopic corrections. An imperfect realization of the intended hyperopic correction may have far greater consequences in a hyperopic eye than comparable myopic corrections. By following the advice given here and using modern technologies, hyperopic corrections can be accomplished with greater levels of success than previously demonstrated, enabling potential treatment levels in LASIK up to 8.00 D. Potential postoperative issues still limit hyperopic corrections in PRK to a maximum of 4.00 to 5.00 D.
1. Artal P, Benito A, Tabernero J. The human eye is an example of robust optical design. J Vis. 2006;6(1):1-7.
2. Arba Mosquera S, Ewering T. New asymmetric centration strategy combining pupil and corneal vertex information for ablation procedures in refractive surgery: theoretical background. J Refract Surg. 2012;28(8):567-573.
3. de Ortueta D, Schreyger FD. Centration on the cornea vertex normal during hyperopic refractive photoablation using videokeratoscopy. J Refract Surg. 2007;23(2):198-200.
4. Carones F, Vigo L, Scandola E. Laser in situ keratomileusis for hyperopia and hyperopic and mixed astigmatism with LADARVision using 7- to 10-mm ablation diameters. J Refract Surg. 2003;19(5):548-554.
5. Arba Mosquera S, de Ortueta D. Analysis of optimized profiles for ‘aberration-free’ refractive surgery. Ophthalmic Physiol Opt. 2009;29:535-548. 6. Antonios R, Arba Mosquera S, Awwad ST. Hyperopic laser in situ keratomileusis: comparison of femtosecond laser and mechanical microkeratome flap creation. J Cataract Refract Surg. 2015;41:1602-1609.
7. Arbelaez MC, Vidal C, Arba Mosquera S. Hyperopia and hyperopic astigmatism correction with the Schwind Esiris: 1-year results. J Emmetropia. 2011;2:131-135.
8. Friehmann A, Mimouni M, Nemet A, et al. Risk factors for epithelial ingrowth following microkeratome-assisted LASIK. J Refract Surg. 2018;34(2):100-105.
9. de Ortueta D, Arba Mosquera S, Baatz H. Topographic changes after hyperopic LASIK with the ESIRIS laser platform. J Refract Surg. 2008;24:137-144.
10. Arba Mosquera S, Awwad ST. Theoretical analyses of the refractive implications of trans-epithelial PRK ablations. Br J Ophthalmol. 2013;97:905-911.
11. Vinciguerra P, Roberts CJ, Albé E, et al. Corneal curvature gradient map: a new corneal topography map to predict the corneal healing process. J Refract Surg. 2014;30(3):202-207.
12. de Ortueta D, Arba-Mosquera S. Laser in situ keratomileusis for high hyperopia with corneal vertex centration and asymmetric offset. Eur J Ophthalmol. 2017;27(2):141-152.
13. Plaza-Puche AB, Aswad AE, Arba-Mosquera S, et al. Optical profile following high hyperopia correction with a 500-hz excimer laser system. J Refract Surg. 2016;32(1):6-13.
14. Rocha KM, Vabre L, Chateau N, Krueger RR. Expanding depth of focus by modifying higher-order aberrations induced by an adaptive optics visual simulator. J Cataract Refract Surg. 2009;35(11):1885-1892.
15. Vinciguerri P, Camesasca FI, Morenghi E, et al. Corneal apical scar after hyperopic excimer laser refractive surgery: long-term follow-up of treatment with sequential customized therapeutic keratectomy. J Refract Surg. 2018;34(2):113-120.
16. Friehmann A, Mimouni M, Nemet AY, et al. Risk factors for epithelial ingrowth following microkeratome-assisted LASIK. J Refract Surg. 2018; 34(2):100-105.
17. Arbelaez MC, Vidal C, Arba-Mosquera S. Six-month experience hyperopic correction with the Schwind Amaris Total-Tech Laser: clinical outcomes. J Optom. 2010;3:198-205.
18. Arba Mosquera S, de Ortueta D. Multifocality changes after LASIK. J Optom. 2012;05:202-208.
19. Solomon R, Donnenfeld ED, Perry HD. The effects of LASIK on the ocular surface. Ocul Surf. 2004;2(1):34-44.
20. Ambrósio R, Tervo T, Wilson SE. LASIK-associated dry eye and neurotrophic epitheliopathy: pathophysiology and strategies for prevention and treatment. J Refract Surg. 2008;24(4):396-407.
21. Rosenfeld SI. Evaluation and management of post-LASIK dry eye syndrome. Int Ophthalmol Clin. 2010;50(3):191-199.
22. Stern M, Beuerman R, Fox R, Gao J. The pathology of dry eye: the interaction between the ocular surface and lacrimal glands. Cornea. 1998;17(6):584-558.
23. Bragheeth MA, Dua HS. Corneal sensation after myopic and hyperopic LASIK: clinical and confocal microscopic study. Br J Ophthalmol. 2005;89:580-585.
24. Albietz JM, Lenton LM.Management of the ocular surface and tear film before, during, and after laser in situ keratomileusis. J Refract Surg. 2004;20(1):62-71.
25. Kanellopoulos AJ, Kahn J. Topography-guided hyperopic LASIK with and without high irradiance collagen cross-linking: initial comparative clinical findings in a contralateral eye study of 34 consecutive patients. J Refract Surg. 2012;28(11 Suppl):S837-S840.
26. Reinstein DZ, Pradhan KR, Carp GI, et al. Small incision lenticule extraction (SMILE) for hyperopia: optical zone centration. J Refract Surg. 2017;33(3):150-156.




