Laser vision correction (LVC) has been the most popular mode of refractive correction for decades. Some surgeons have reported seeing their LASIK volumes declining; however, this is likely due to economic and personal circumstances or preferences, rather than a reflection of what LVC can achieve. For patients with appropriate prescriptions and in appropriate age brackets—including, in many cases, presbyopes—LVC can be a fantastic choice to decrease spectacle dependence. In some situations, however, refractive lens exchange (RLE) is preferable. This article explores some of the situations in which RLE might be the best refractive surgical option for appropriate patients and outlines my decision tree for RLE.
POPULARITY OF RLE
In my practice, the popularity of RLE with our patients appears to be increasing, and this may be occurring for a number of reasons. First, we have newer, better designed IOL technologies and surgical platforms that allow lens surgery to be carried out more precisely and with fewer risks than in the past. Second, we also have better diagnostics and improved IOL power calculation formulas that allow us to hit our refractive targets with greater accuracy and reliability. Finally, we are treating more patients in their 60s and beyond than we have in the past several years.
For patients now in their 60s, and even to some extent those in their late or early 50s, many surgeons have begun to think more carefully about RLE as an option for refractive correction. When patients of this age enter our practice, should we be talking to them about lens- or cornea-based correction? If we select RLE, how do we decide what kind of lens we might offer? This is where the RLE decision tree comes into play.
THE DECISION TREE
The RLE decision tree considers a combination of factors, including age, prescription, degree of presbyopia, quality of the ocular surface, and other factors that might exclude a patient from LVC, such as an irregular cornea. Age is one of the most important factors, and I generally would not perform RLE in a patient who is younger than 50. In those patients, LVC and phakic IOLs are both better options.
There are exceptions to the rule, however, such as patients between the ages of 45 and 50 who are very highly hyperopic (7.00–9.00 D). In this group, the ocular anatomy cannot safely accommodate a phakic IOL. Further, the magnitude of their hyperopia produces earlier and more significant presbyopic symptoms than eyes with myopic experience. So, for instance, the eyes of a 47-year-old high hyperope behave similarly to the eyes of a myope or emmetrope in his or her late 50s. In that case, therefore, RLE with a presbyopia-correcting lens can be a good option.
In patients with established presbyopia, lower prescriptions, and good ocular surface condition, I feel that laser blended vision (LBV) can provide patients with more natural vision, but for those who are entering their 50s and have higher prescriptions, I am more likely to recommend RLE. A good example would be a 63- or 64-year-old with -10.00 D of myopia. Although this is a perfectly acceptable and safe age to have LVC, in my opinion, RLE is the better choice because he or she is close to the age at which cataracts develop.
HYPEROPIC CONSIDERATIONS
There are additional considerations for patients with hyperopia. If I treat a myopic presbyope with LBV, I typically slightly undercorrect one of the eyes. For a hyperope, however, I will overcorrect to provide myopia in the nondominant eye.
For example, a presbyopic patient with a prescription of 3.00 D for distance and an add of another 2.00 D might actually need a 5.00 D hyperopic treatment in the nondominant eye. This might be a larger correction than some surgeons are comfortable making with LVC. In such cases, I might be more likely to recommend RLE because high hyperopic LVC treatments are associated with some regression. Furthermore, we know that with time hyperopes will become more presbyopic, losing some of their near-vision effect over 5 to 10 years. RLE gives these patients a more permanent option.
The other issue in hyperopes is that they tend to have smaller eyes and more crowded angles, which increases the risk of angle-closure glaucoma. In many cases, removing the natural lens in RLE eliminates that risk.
OTHER PATIENT FACTORS
When it comes to procedure selection, I am strongly guided by a few things. First and foremost, what is the patient’s goal? If it is complete and permanent spectacle independence, RLE is a more likely choice. On the other hand, if the patient is happy wearing reading glasses or occupational bifocals, I may opt for LVC. If a patient has some form of macular degeneration, corneal irregularity, or a very poor ocular surface that I don’t feel I can treat and improve, I advise him or her to pursue distance correction only, as a presbyopia-correcting IOL will not likely be tolerated.
Most patients want to achieve spectacle independence. I try to determine their near and intermediate vision requirements by using the following line of questioning and observation:
- Would the patient be happy putting glasses on for small print reading, or does he or she want to be spectacle independent for everything?
- Can he or she understand the information you provide?
- Will he or she accept the pros and cons of various technology options?
- Is he or she motivated to be independent of glasses and contact lenses? (The more motivated the more likely I will recommend RLE.)
- How much time does the patient spend driving at night? (This information helps me determine what technology would be appropriate.)
- How demanding is the patient likely to be?
- Is the patient rational?
Any corneal astigmatism must be treated at the time of surgery. If a patient has more than 2.50 D of corneal astigmatism, I prefer a monovision or blended vision strategy with a standard toric IOL. This is because I worry about the combination of coma and toric presbyopia-correcting IOLs with significant residual astigmatism, which could be difficult to treat even with a laser enhancement. If the patient has less than 2.50 D but more than 1.25 D of corneal astigmatism, a regular cornea, a normal macula, and a good ocular surface, I will consider a presbyopia-correcting toric IOL. For less than 1.25 D of corneal astigmatism or less than 1.00 D if against-the-rule, patients can have on-axis presbyopia-correcting IOL surgery with or without a limbal relaxing incision. It is also important that, in these situations, patients are accepting of the implications of presbyopic-lens technology.
PRESBYOPIa-correcting LENS
CHOICES
The next decision is which type of presbyopia-correcting lens technology to use. I prefer a trifocal IOL because the various focal points that these lenses generate more closely simulates natural vision. Specifically, I tend to implant the RayOne Trifocal (Rayner). With this lens, half of the light is allocated for distance vision and the other half is divided between near and intermediate vision (Figure 1); the light energy is split at the 3-mm pupil as follows: 52% distance, 22% intermediate, and 26% near. The RayOne Trifocal offers high-quality distance, reading, and intermediate vision, and, because it has fewer rings on the optic surface than other trifocal IOL designs, patients experience a little less glare and improved night vision. This lens is also developed to be less dependent on pupil size or lighting conditions.
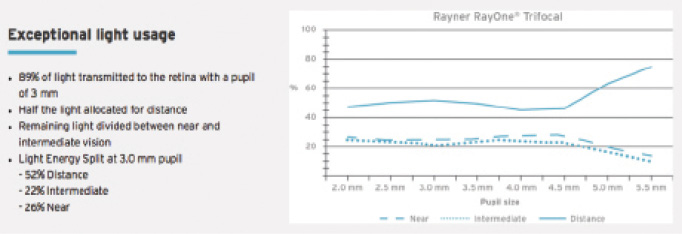
Figure 1. Allocation of light energy by the RayOne Trifocal.
Patients who do a lot of night driving, wear glasses for reading small print, or have slight corneal irregularities might benefit from an extended depth of focus (EDOF) lens. EDOF lenses can provide an extended range of vision in patients who have a slightly aberrated cornea but who are not candidates for LVC. These lenses, such as the Symfony (Johnson & Johnson Vision), provide much more depth of focus in near vision than one might expect. Patients also tend to experience less prominent halos with EDOF lenses than with trifocals. However, the amount of near add is approximately 1.75 D in the IOL plane, so these lenses tend to provide distance and intermediate vision rather than small print reading. Therefore, patients often do not achieve complete spectacle independence.
Lastly, in patients who have very irregular corneas or who have had previous RK or PKP, for instance, I implant the IC-8 small-aperture lens (AcuFocus) in the nondominant eye. I find that this lens gives about 2.00 D increased depth of focus, which is enough to neutralize the variation in vision that these patients otherwise get and to compensate for their corneal irregularity.
CONCLUSION
We have an abundance of surgical options to correct refractive errors (Figure 2), and deciding what is the most appropriate choice for each patient can sometimes be daunting. Presbyopic patients often have high expectations; therefore, understanding what they want and communicating to them a realistic account of what they are likely to experience in the early, intermediate, and long term is crucial.
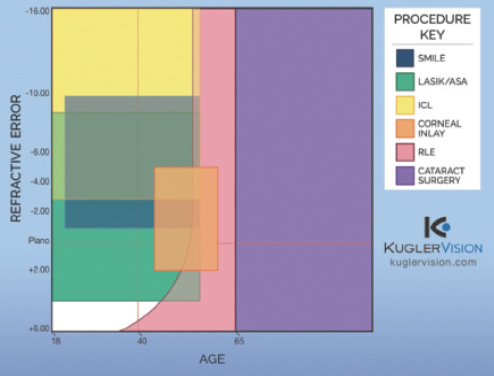
Figure 2. Available procedures in the refractive surgery spectrum. (Abbreviations: SMILE, small-incision lenticule extraction; ASA, advanced surface ablation; RLE, refractive lens exchange)
When used in the right subset of patients, RLE is a precise and positive surgical option that can provide patients with excellent postoperative outcomes in near, intermediate, and distance vision. In my practice, more than 97% of patients with trifocal lenses achieve complete independence from glasses for all activities. The best part is that RLE eliminates the potential need for future cataract surgery and does not disrupt the corneal surface.
Whichever approach to presbyopia correction is chosen, it is important to help patients understand that every available option requires some compromise on their part. Patients need to see the procedure as a journey rather than a one-off intervention, and helping them to set realistic expectations is mandatory. When patients know what to expect, they are more understanding and accepting of the compromise that they will be agreeing to when it comes to surgery. For most presbyopic patients, these compromises are acceptable compared with the alternative of spectacle dependence.




