In recent years, CXL has significantly reduced the number of keratoplasties required for patients with corneal ectasia. The origin of keratoconus is unclear, but its incidence in those older than 40 years is much lower than in those younger, probably because of age-related corneal stiffening. CXL can be considered a technique that accelerates natural aging so that progression of keratoconus can be stopped.
AT A GLANCE
- The standard CXL Dresden protocol is limited to use in corneas with stromal thickness of greater 400 μm, but corneas with advanced keratoconus often fall below this threshold and require use of a modified protocol.
- Most currently used modified protocols for thin corneas aim to increase overall stromal thickness, therefore also increasing the complexity of CXL in thin corneas.
- The authors’ new approach tries to simplify the protocol and make it more precise: It reduces UV energy by shortening irradiation time based on individual stromal thickness, as measured at the end of riboflavin imbibition, thus lowering total irradiance.
The standard Dresden protocol for CXL1 is limited to use in corneas with stromal thickness of greater 400 µm. However, corneas with advanced keratoconus often fall below this thickness threshold and require the use of modified protocols. Most currently used modified protocols for thin corneas aim to increase overall stromal thickness, either by swelling the cornea with hypoosmolar riboflavin solution to increase absorption throughout the corneal stroma or by applying a contact lens on top of the deepithelialized cornea. These protocols increase the complexity of CXL in thin corneas.
In this article, we describe a new approach we have devised, which aims to simplify the protocol while making it even more precise. We reduce UV energy by shortening the irradiation time based on the eye’s individual stromal thickness, as measured at the end of riboflavin imbibition, thus lowering total irradiance. This technique should not be confused with customized CXL, in which different but fixed areas of irradiance are used.
PRINCIPAL FACTORS OF CXL
CXL relies on a photochemical reaction that is initiated by the absorption of UV energy by the photosensitizer riboflavin. As a consequence, the highly reactive triplet state of riboflavin is excited, generating reactive radicals and reactive oxygen species by type 1 and type 2 photochemical reactions.2 These highly reactive species then oxidize the extracellular matrix of the cornea.
To date, the underlying chemical reactions responsible for the observed mechanical stiffening effect of CXL are not fully understood. The molecular weight of both collagens and proteoglycans have been reported to increase after CXL. Interestingly, the stiffening effect of CXL does not strictly correlate with the overall applied UV dosage; it also depends on irradiation time and oxygen availability.3
To better understand the interaction of the three principal factors of CXL—UV energy, riboflavin concentration (which determines absorption), and oxygen availability (which depends on oxygen pressure and irradiation time)—we recently developed a theoretical model and performed experimental validation with multiple CXL protocols.4 This model allows us to predict how the stiffening effect of CXL is altered if a single treatment parameter is changed. By applying this model, we found that the standard CXL protocol is limited by oxygen diffusion, and hence the UV fluence can be reduced by half, at no cost to the stiffening effect, if the irradiation time remains the same.5
The ability to predict the overall stiffening effect, and, more important, to predict the depth of corneal stiffening also opens the way for individualized CXL parameter calculation, as is further discussed later in this article.
SAFETY CONSIDERATIONS IN CXL
If the UV energy absorbed by a single cell surpasses a certain threshold, apoptosis is induced.6 With standard CXL, and if corneal thickness is greater than 400 µm, keratocytes go into apoptosis up to a depth of approximately 300 µm; endothelial cells remain unaffected and therefore safe.7 In thinner corneas, more UV energy is absorbed by the endothelium, increasing the risk of endothelial cell loss.
Although keratocyte apoptosis and repopulation occur naturally in corneal wound healing, endothelial cells cannot regenerate. Therefore, endothelial cell loss or UV-induced endothelial mutations are the major safety concerns of CXL in thin corneas.
POTENTIAL FOR INDIVIDUALIZED CXL
Previous attempts to increase corneal thickness during CXL treatment have included the use of hypoosmolar riboflavin solution,8 increased dosing frequency, and interplay with the eyelid speculum. Nevertheless, the induction of excessive corneal swelling is not recommended, as it separates the extracellular matrix. Therefore, reducing UV irradiation time in thin corneas is a promising and potentially more precise tool to individualize CXL while keeping corneal thickness constant.
Given the predictions of our theoretical model, we devised a nomogram for a UV intensity of 3 mW/cm2. With the help of our model, the UV irradiation time that is required to reach a stiffening effect up to a depth of 70 µm distance from the endothelium can be determined for an individual corneal thickness during surgery. A decisive factor for determining the individualized irradiation time is the corneal thickness directly before UV irradiation. The distance of 70 µm to the endothelium is comparable to the demarcation line in standard Dresden protocol CXL.
MORE WORK NEEDED
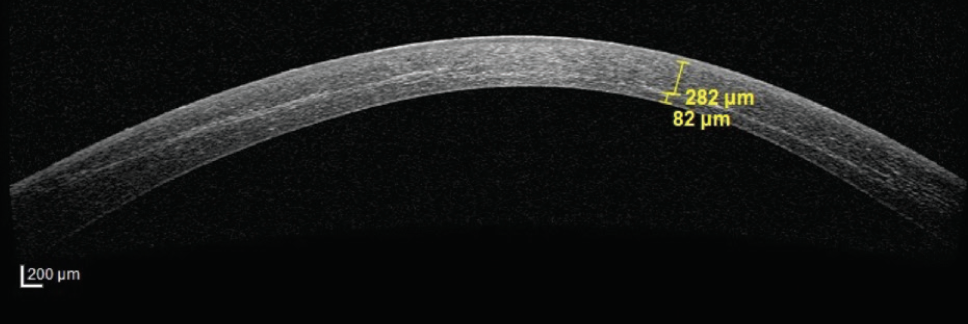
To date, we have performed individualized CXL in eyes with corneal thickness as low as 312 µm (Figure). As expected, the demarcation line was much shallower in thinner corneas being irradiated for shorter times, and the endothelium did not show any sign of damage. Despite these promising preliminary outcomes, it remains yet to be investigated whether CXL with reduced UV dosage can effectively halt keratoconus progression in the long term.
1. Wollensak G, Spoerl E, Seiler T. Riboflavin/ultraviolet-A–induced collagen crosslinking for the treatment of keratoconus. Am J Ophthalmol. 2003;135(5):620-627.
2. Kamaev P, Friedman MD, Sherr E, Muller D. Photochemical kinetics of corneal cross-linking with riboflavin. Invest Ophthalmol Vis Sci. 2012;53(4):2360-2367.
3. Kling S, Richoz O, Hammer A, et al. Increased biomechanical efficacy of corneal cross-linking in thin corneas due to higher oxygen availability. J Refract Surg. 2015;31(12):840-846.
4. Kling S, Hafezi F. An algorithm to predict the biomechanical stiffening effect in corneal cross-linking. J Refract Surg. 2017;33(2):128-136.
5. Kling S, Hafezi F. Biomechanical stiffening: slow low-irradiance corneal crosslinking versus the standard Dresden protocol. J Cataract Refract Surg. 2017;43(7):975-979.
6. Spoerl E, Mrochen M, Sliney D, Trokel S, Seiler T. Safety of UVA-riboflavin cross-linking of the cornea. Cornea. 2007:26(4):385-389.
7. Wollensak G, Spoerl E, Wilsch M, Seiler T. Endothelial cell damage after riboflavin–ultraviolet-A treatment in the rabbit. J Cataract Refract Surg. 2007;29(9):1786-1790.
8. Hafezi F, Mrochen M, Iseli HP, Seiler T. Collagen crosslinking with ultraviolet-A and hypoosmolar riboflavin solution in thin corneas. J Cataract Refract Surg. 2009;35(4):621-624.





